Clear Sky Science · en
The Ontology of Adverse Events in 2025
Why tracking medical side effects matters
Whenever we take a medicine, receive a vaccine, or undergo surgery, there is a chance of unwanted side effects. Doctors, regulators, and scientists try to watch these "adverse events" closely so that dangerous patterns are caught early. But today’s reports are scattered across many databases, written in slightly different ways, and hard for computers to analyze. This article introduces an evolving global effort called the Ontology of Adverse Events (OAE), which aims to give the medical world a shared, precise language for describing side effects so that safety questions can be answered faster and more reliably.
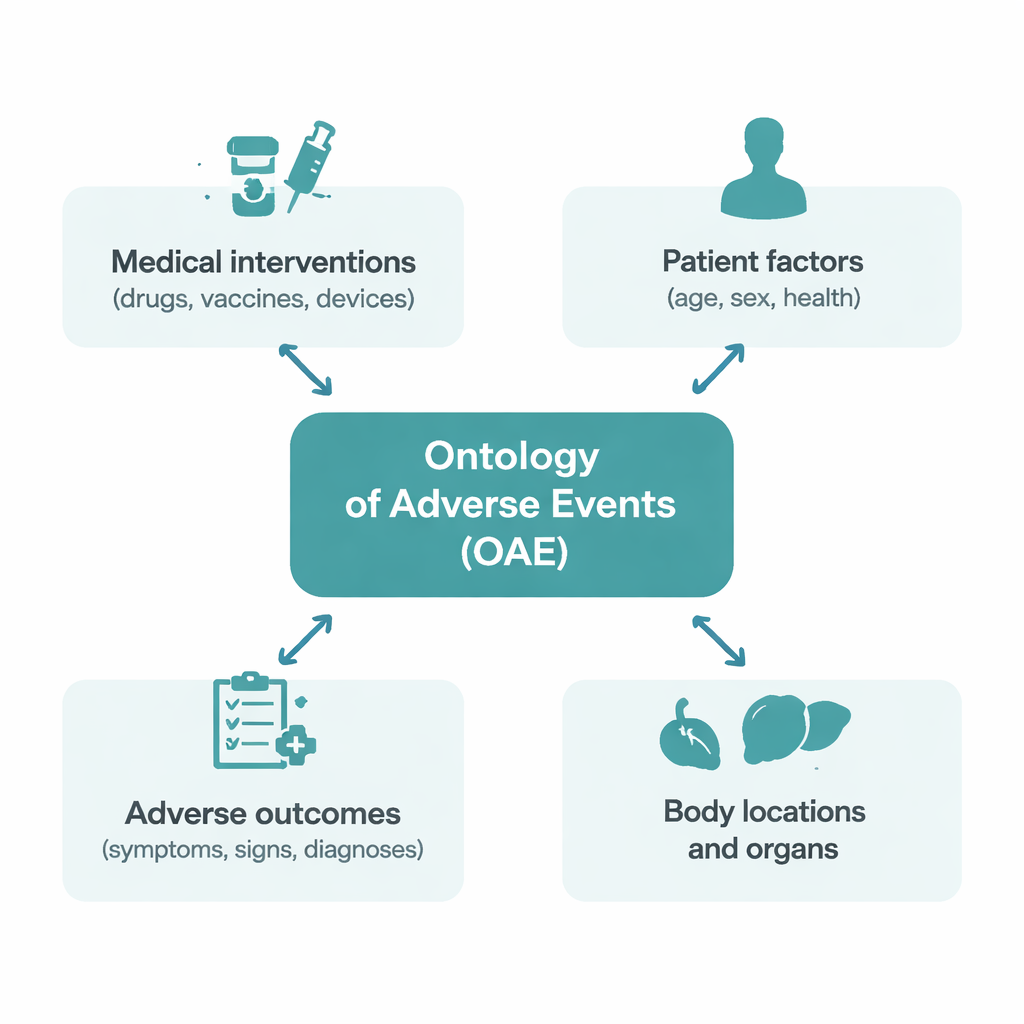
A common map for medical side effects
At its heart, the OAE is like a detailed map of how medical side effects are described. Instead of just listing names of problems, it organizes them into logical families: where in the body they occur, what they look like in the clinic (such as symptoms patients feel, signs doctors can measure, or underlying disease processes), and how they relate to medical interventions like drugs, vaccines, and devices. The project began in 2011 to reduce confusion among competing medical vocabularies. A key decision was to distinguish carefully between any event that happens after treatment (an adverse event) and those events where there is actual evidence the treatment caused the problem (a causal adverse event). That separation makes safety analyses less biased and more transparent.
From a small catalog to a rich encyclopedia
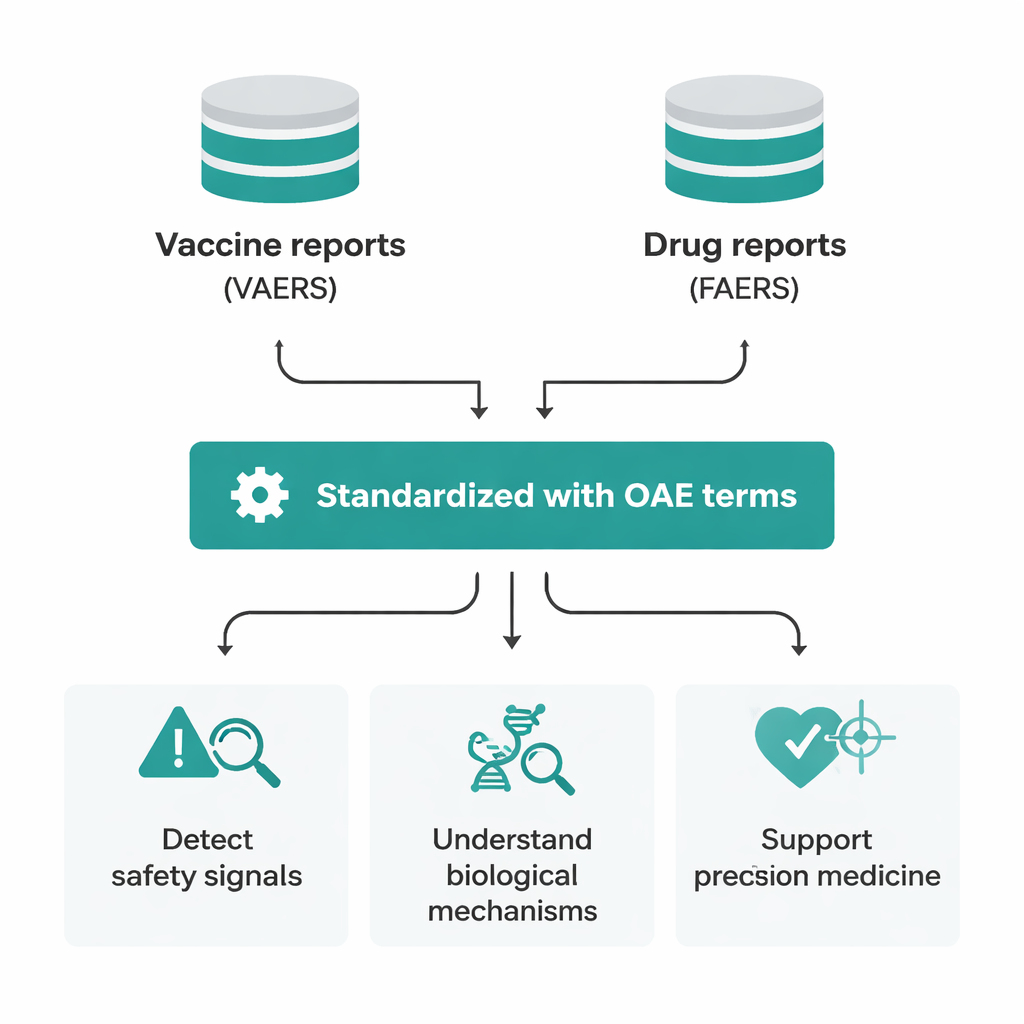
Over the past decade, OAE has expanded from just over 3,000 terms to more than 10,800 unique entries, a rise of about 250% that reflects both finer detail and broader clinical coverage. The team grew the ontology in several ways. They mined major safety databases such as the Vaccine Adverse Event Reporting System (VAERS) and the FDA’s drug safety database (FAERS) to identify real-world side effects. They cross-linked with widely used regulatory vocabularies like MedDRA, and they carefully curated new terms emerging from research on vaccine and drug safety. Each new term carries a human-readable label, clear definition, links to source references, its location in the body, and editing history, making OAE both traceable and easy for computers to process.
Connecting scattered data for better safety insights
Because OAE follows common standards for biomedical ontologies, it can plug into many other specialized knowledge systems. Researchers have already built spin-off ontologies for side effects of particular diseases, nerve-damaging drugs, and heart medicines. By aligning OAE terms with anatomy, gene, and symptom ontologies, scientists can trace paths from a drug through its molecular targets and affected organs all the way to the side effects seen in patients. This has helped uncover previously under-reported reactions in case reports, compare safety profiles of different vaccines, and explore why some drug classes tend to harm multiple body systems at once.
Supporting precision medicine around the world
The article also highlights how OAE fits into the broader shift toward precision medicine—the idea of tailoring treatments to individual patients based on their biology and life context. A precise, computer-readable description of side effects is essential if we want to match drugs and vaccines to the people most likely to benefit and least likely to be harmed. OAE’s structured language allows large datasets to be combined and searched in consistent ways, enabling more refined grouping of patients and clearer views of risk. In China, where existing vaccine side-effect systems rely on broad categories and an overused "other" label, researchers are translating OAE into Chinese and integrating it with national data platforms to bring the same level of detail and interoperability to domestic surveillance.
What this means for patients and clinicians
For a lay reader, the main message is that better words lead to better safety. By turning a messy collection of side-effect descriptions into a carefully organized, multilingual, and computer-friendly reference, the OAE helps scientists detect safety signals earlier, regulators compare products more fairly, and clinicians understand how a treatment’s risks vary across age, sex, disease, and other factors. The authors plan to keep extending OAE into more specialized areas and tightening its links with other medical knowledge systems. Over time, this shared vocabulary could make the complex world of vaccine and drug safety more transparent, more responsive, and ultimately safer for patients everywhere.
Citation: Pan, C., Yang, Q., Zhang, X. et al. The Ontology of Adverse Events in 2025. Sci Data 13, 259 (2026). https://doi.org/10.1038/s41597-026-06584-x
Keywords: adverse events, drug safety, vaccine safety, medical ontology, precision medicine