Clear Sky Science · en
Stress adaptation of mitochondrial protein import by OMA1-mediated degradation of DNAJC15
How Our Cells Cope When Their Powerhouses Are Under Stress
Every cell in your body depends on tiny structures called mitochondria to turn food into usable energy. Like miniature factories, mitochondria constantly import fresh parts—new proteins made elsewhere in the cell—to keep their machinery running. This paper reveals how, when mitochondria are stressed or damaged, cells deliberately dial back this protein import pipeline. By doing so, they protect themselves from overload and coordinate a wider stress response that involves not only mitochondria, but also another key compartment, the endoplasmic reticulum.
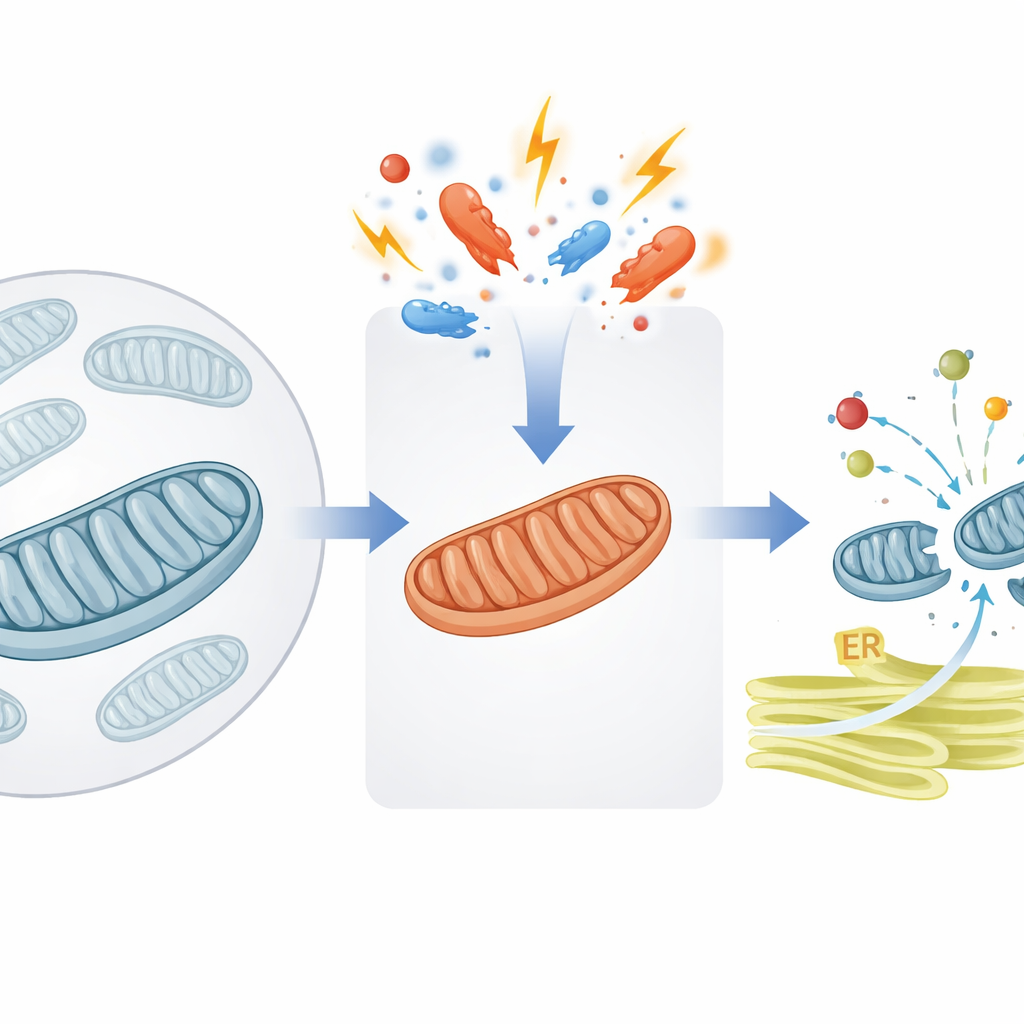
A Safety Valve for Mitochondrial Protein Traffic
Mitochondria rely on thousands of proteins that are first built in the cell’s main compartment and then shipped in. The authors focus on a crucial gateway in the mitochondrial inner membrane, supported by a helper protein called DNAJC15. Under normal conditions, DNAJC15 helps pull new protein “packages” through the import channel and into the mitochondrial interior, where many of them become part of the energy-producing machinery known as oxidative phosphorylation (OXPHOS). The study shows that when mitochondria experience stress—such as impaired energy production or changes in membrane properties—the cell flips a molecular switch that changes how much protein the organelle is willing to import.
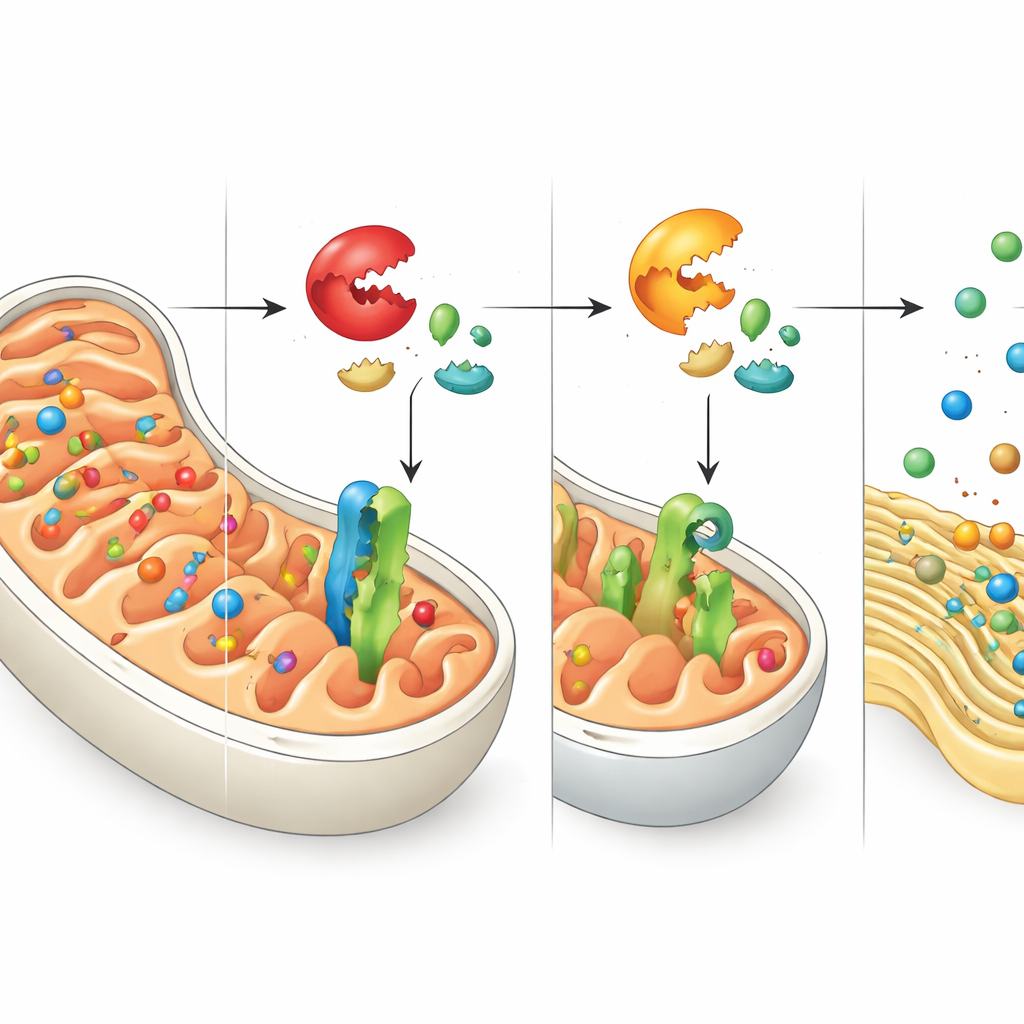
OMA1: The Stress-Activated Cutter
At the heart of this switch is a stress-sensitive enzyme called OMA1, lodged in the inner mitochondrial membrane. When mitochondria run into trouble, OMA1 becomes active and clips DNAJC15 at a specific position near its beginning. This cut produces a shorter version of DNAJC15 that is quickly chewed up by another mitochondrial enzyme complex called the m-AAA protease. As a result, the full-length, import-competent form of DNAJC15 is lost. The authors show that cells lacking DNAJC15, or in which DNAJC15 is rapidly destroyed after OMA1 activation, have a reduced capacity to bring new proteins into mitochondria, especially those needed for building and maintaining OXPHOS complexes.
Slowing Energy Assembly to Match Mitochondrial Health
Using large-scale protein measurements and import assays, the team found that DNAJC15 is particularly important for supplying the mitochondrial interior with proteins involved in gene expression and assembly of the respiratory chain. When DNAJC15 is missing, these proteins accumulate more slowly inside mitochondria, and the organelles’ ability to consume oxygen and fuel energy production—as measured in isolated mitochondria—declines, especially for complex I of the respiratory chain. Another component of the import channel, TIMM17A, works hand in hand with DNAJC15: losing both causes especially strong defects in the levels of mitochondrial ribosomal proteins and OXPHOS components. Together, these findings suggest that OMA1-mediated destruction of DNAJC15 is a way to temporarily curb construction of new energy machinery until the organelle recovers.
Misdelivered Packages and a Call for Help from the ER
Shutting down DNAJC15 does not stop the cell from making mitochondrial proteins—it mainly stops them from arriving at their intended destination. The authors traced where these stranded proteins go and found that many end up at the endoplasmic reticulum (ER), a network of membranes that normally helps fold and process different sets of proteins. These wayward mitochondrial proteins become embedded in ER membranes, disturbing the ER’s own quality-control balance. In response, the cell activates a protective program known as the unfolded protein response, specifically through a branch controlled by the sensor ATF6. This response boosts the ER’s capacity to handle misfolded or misplaced proteins, revealing a tight coordination between mitochondrial and ER stress systems.
A Coordinated Slowdown to Protect the Cell
Taken together, the study paints a picture of a built-in braking system for mitochondrial biogenesis. When mitochondria are stressed, OMA1 trims and indirectly destroys DNAJC15, reducing the flow of new, energy-related proteins into compromised organelles. Some of these proteins are temporarily buffered at the ER, which mounts its own protective response. By coupling mitochondrial import capacity to organelle health, and by engaging the ER as a backup site for excess proteins, cells can avoid clogging damaged mitochondria and gain time either to repair them or to remove them entirely. For a lay observer, this work shows how deeply integrated our cellular quality-control systems are: even the tiniest missteps in one compartment are sensed and counterbalanced by others to keep energy production—and life itself—on track.
Citation: Kroczek, L., Nolte, H., Lasarzewski, Y. et al. Stress adaptation of mitochondrial protein import by OMA1-mediated degradation of DNAJC15. Nat Struct Mol Biol 33, 499–511 (2026). https://doi.org/10.1038/s41594-026-01756-0
Keywords: mitochondrial stress, protein import, cellular quality control, oxidative phosphorylation, endoplasmic reticulum