Clear Sky Science · en
Structure and mechanism of antiphage retron Eco2
How Bacteria Use a Built-In "Self-Destruct" to Fight Viruses
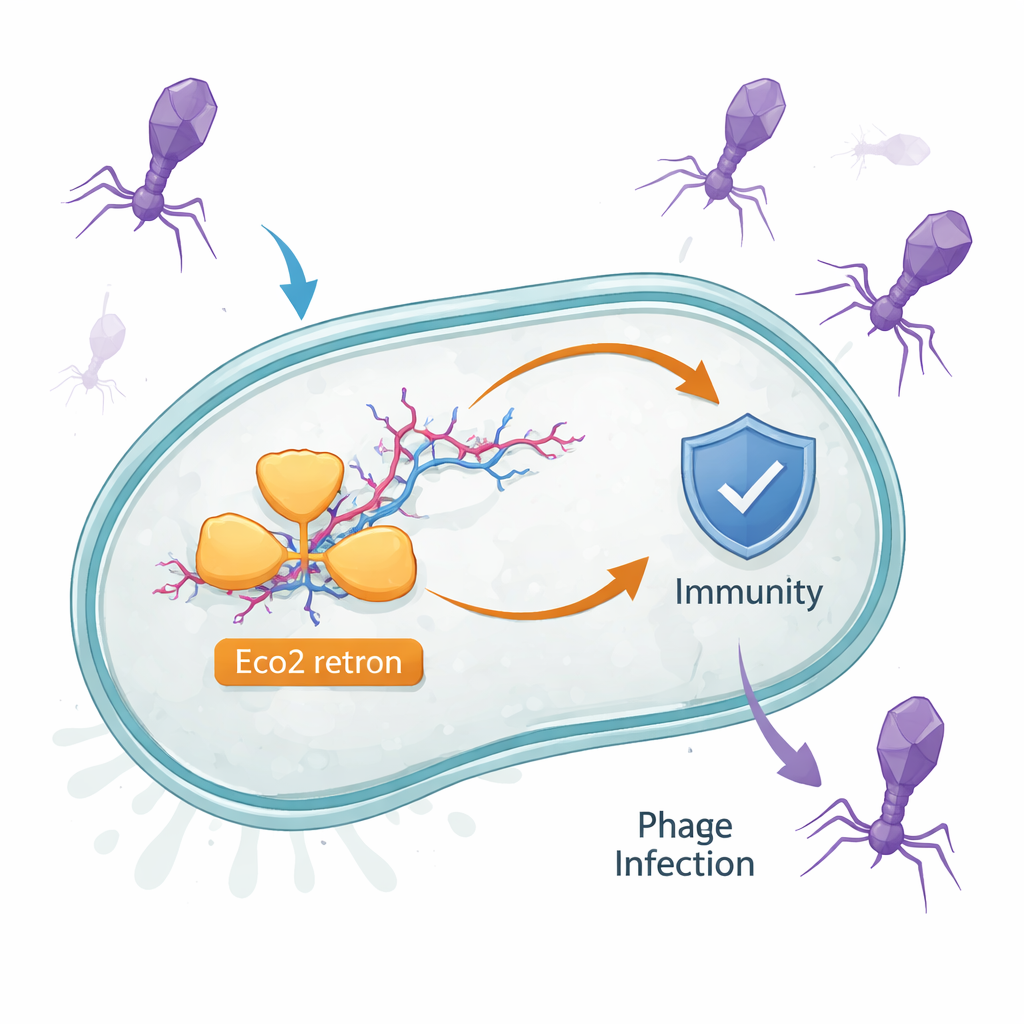
Viruses that infect bacteria, called phages, are everywhere—from oceans to our own gut. This study uncovers how a tiny genetic gadget inside some bacteria, known as the Eco2 retron, helps them survive phage attack. By revealing Eco2’s 3D structure and how it turns on, the work explains a new kind of emergency defense: when infection is detected, the cell deliberately shuts down its own protein production to stop the virus from spreading.
A Hidden DNA–RNA Gadget in Bacterial Cells
Bacteria carry many small defense systems that only spring into action during infection. Eco2 is one of these. It lives in Escherichia coli and consists of a single protein that both copies RNA into DNA and can cut nucleic acids. This protein works together with a strange molecule called msDNA, a hybrid of RNA and single-stranded DNA that branches like a tiny forked twig. Earlier hints suggested that retrons like Eco2 defend against phages, but how a single protein and its msDNA partner sense infection and respond was not understood.
Broad Protection Against Many Viruses
The researchers first asked how well Eco2 protects bacteria from a wide range of phages. By challenging Eco2-bearing cells with a large, diverse collection of viruses, they found that Eco2 blocks many phage families but not all of them. When some phages managed to escape, their genomes showed mutations in genes that encode viral DNA-cutting enzymes, especially one called DenB. This pattern suggested that Eco2 is triggered by specific viral nucleases: when those enzymes are inactivated by mutation, the virus can slip past the defense.
A Three-Armed Molecular Shield
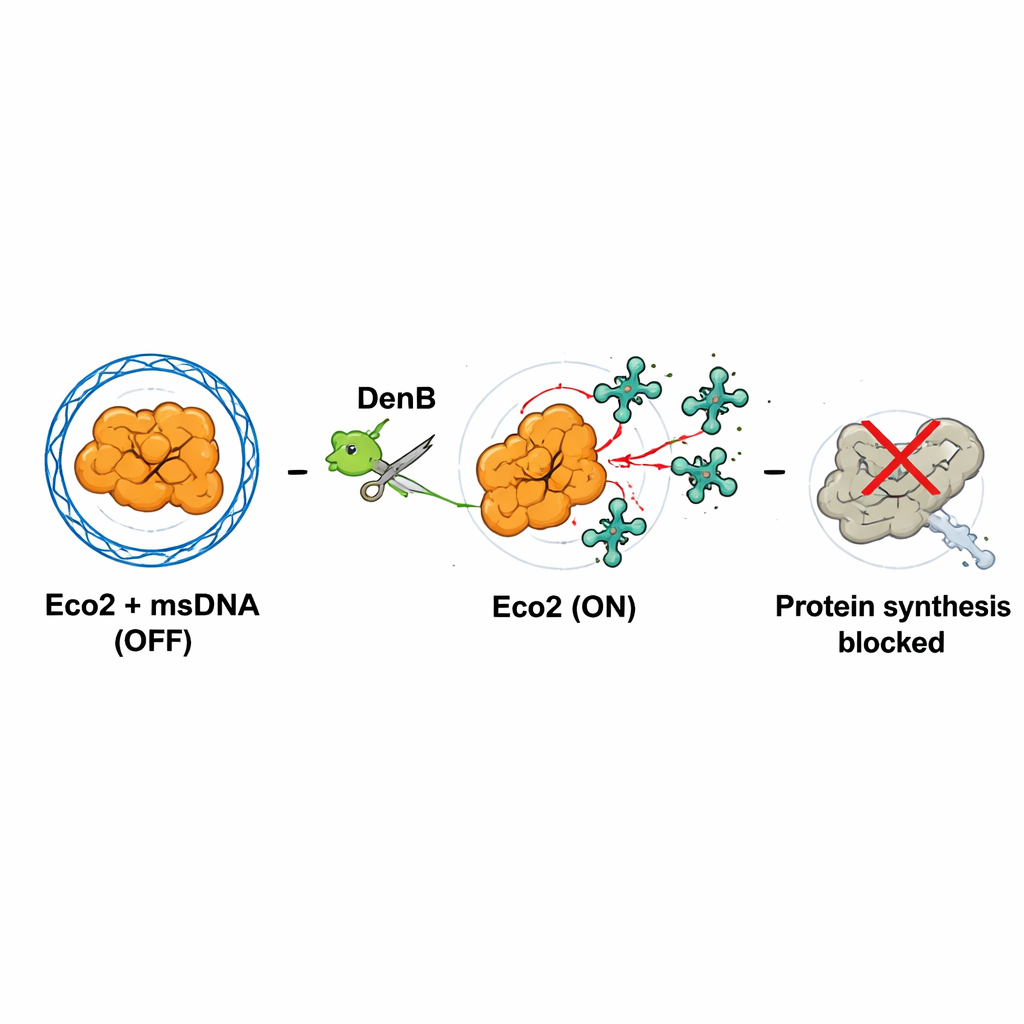
To see how Eco2 is built, the team used cryogenic electron microscopy to visualize the complex at near-atomic detail. Eco2 does not act alone: three copies of the Eco2 protein assemble into a three-armed, shuriken-like structure. The branched msDNA threads through and around these three proteins, tying them together. In doing so, msDNA forms a three-way junction at the center and physically “cages” the nuclease part of Eco2, blocking its cutting site. Hydrogen–deuterium exchange experiments, which track how flexible different regions are, showed that msDNA binding stiffens and stabilizes the complex. In this resting state, Eco2 is effectively locked in the OFF position.
Viral Attack Turns the System On
The key to activation is msDNA destruction. The viral enzyme DenB, which normally helps the phage process DNA, also cuts within the msDNA that cages Eco2. The authors mimicked this process using both purified DenB and a standard DNA-cutting enzyme and saw that once msDNA is sliced, Eco2’s nuclease site becomes exposed and rearranges into an active configuration. High-resolution structures of this “ON state” showed that a critical amino acid in the nuclease pocket moves into a position where it can properly hold a magnesium ion, which is essential for cutting RNA. Thus, viral nuclease activity, meant to remodel the host’s DNA, unintentionally pulls the safety pin on Eco2.
Cutting the Cell’s Own tRNAs to Stop the Virus
What does activated Eco2 cut? Using cell-free systems, radioactive labeling, and RNA sequencing during real infections, the team discovered that Eco2 primarily nicks transfer RNAs (tRNAs), the adaptor molecules that feed amino acids into the ribosome. Eco2 clips these tRNAs near their tail ends, especially at sites rich in the base cytosine. When many tRNAs are damaged, the cell’s ribosomes can no longer make proteins efficiently. In the presence of DenB and active Eco2, green fluorescent protein production dropped sharply, and both bacterial and phage RNAs were chewed up. This shutdown likely represents an “abortive infection”: the infected bacterial cell sacrifices its own growth so that the virus cannot successfully replicate and spread.
Why This Matters Beyond Bacteria
This work shows how a minimal retron can sense a viral enzyme, flip from an inactive to an active form, and then cripple protein synthesis by targeting tRNAs. For a non-specialist, the key idea is that bacteria sometimes beat viruses not by precision targeting, but by pulling the plug on their own cellular machinery at just the right moment. Beyond deepening our understanding of microbial immunity, the detailed structures of Eco2 and its msDNA scaffold provide a blueprint for engineers hoping to repurpose retrons as tools for genome editing and synthetic biology.
Citation: Jasnauskaitė, M., Juozapaitis, J., Liegutė, T. et al. Structure and mechanism of antiphage retron Eco2. Nat Struct Mol Biol 33, 330–340 (2026). https://doi.org/10.1038/s41594-026-01754-2
Keywords: bacteriophage immunity, retron Eco2, bacterial antiviral defense, tRNA cleavage, msDNA