Clear Sky Science · en
The filamentous ultrastructure of the PopZ condensate is required for its cellular function
How cells use soft droplets to stay organized
Inside every living cell, chemistry is crowded and chaotic. Yet cells still manage to keep crucial molecules in the right place at the right time. One way they do this is by forming tiny liquid-like droplets, or condensates, that gather certain proteins while excluding others. This study looks closely at one such droplet-building protein in bacteria, called PopZ, and shows that its internal scaffold of tiny filaments is essential for keeping the cell’s life-or-death processes on track.
A microscopic organizer at the cell’s poles
In the bacterium Caulobacter crescentus, PopZ collects at the two ends, or poles, of the cell where it forms concentrated compartments. These PopZ-rich “microdomains” help control the cell cycle by attracting specific partner proteins and anchoring the chromosome during division. When PopZ is deleted, cells divide poorly, lose their normal shape, and mis-handle their DNA. Earlier work showed that if PopZ droplets are too runny or too stiff, cells also struggle. What was unknown is how the detailed structure inside these droplets — the way PopZ molecules assemble into larger shapes — gives rise to their material properties and to proper cellular behavior.
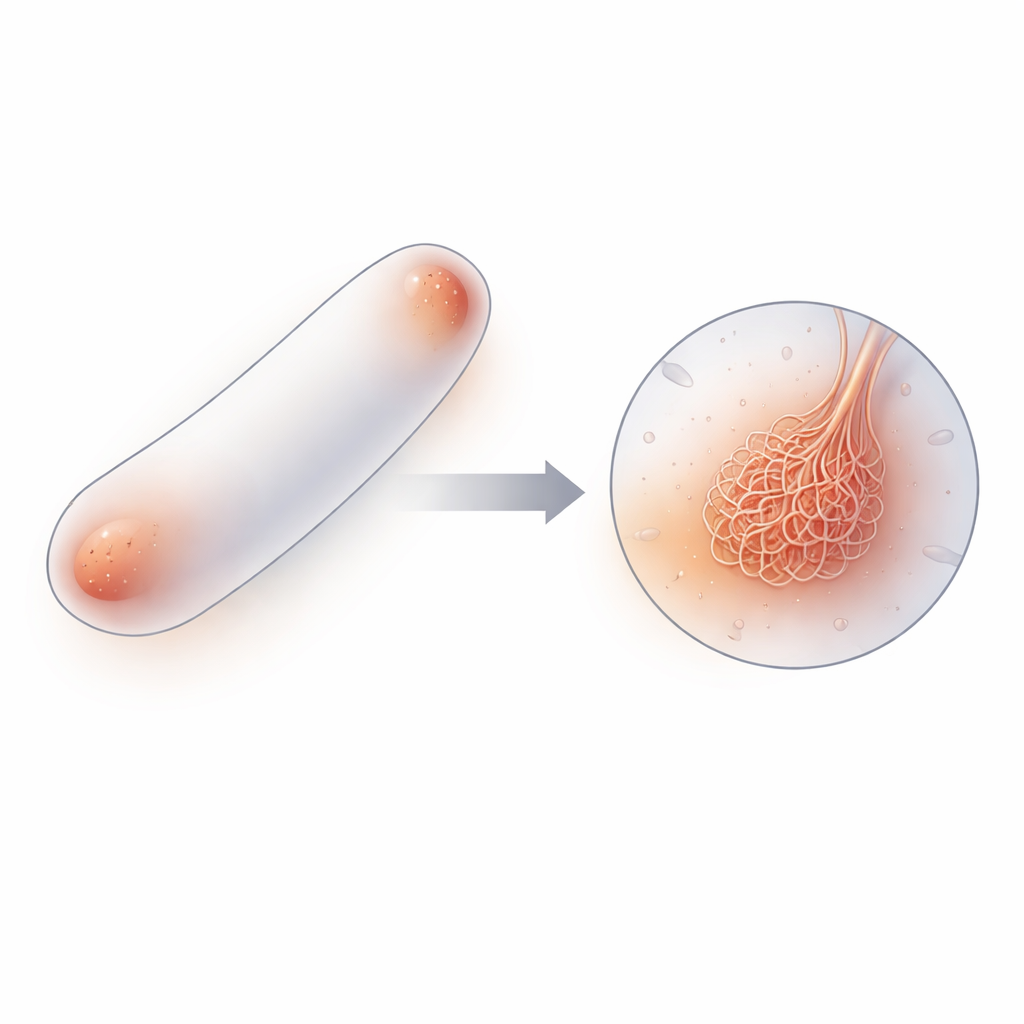
From single molecules to a mesh of filaments
Using a combination of cryo–electron tomography (a 3D imaging method at very low temperatures), biochemical assays, single-molecule fluorescence, and computer simulations, the authors mapped out how PopZ molecules assemble across scales. Individual PopZ proteins first join into three-part units (trimers), which then pair up to form six-part units (hexamers). These hexamers stack end-to-end into short, flexible filaments tens of nanometers long, and tangled networks of these filaments make up the PopZ condensate. Imaging both purified PopZ droplets and PopZ in intact cells revealed the same filamentous meshwork, showing that this architecture is not an artifact of test-tube experiments but a core feature of how PopZ works in living bacteria.
Built-in brakes and switches for droplet formation
PopZ is made of distinct sections that each play a different role in this assembly line. A compact helical segment at the tail end acts as the main oligomerization and filament-forming module, powerful enough on its own to build filaments and droplets. In contrast, a floppy, negatively charged stretch in the middle tends to keep molecules apart, making condensation harder. At the opposite end, a short helix both recruits client proteins and, in the dilute state, folds back to contact the tail, further discouraging premature clustering. When conditions change — for example, when certain salts are present — these repulsive interactions are weakened. PopZ then shifts its shape: the client-binding helix swings away from the tail, the inhibitory cloud of disordered segments opens up, hexamers stack more readily into filaments, and inter-filament contacts become favorable. This phase-dependent conformational change means that the same protein region that blocks binding in the dilute state becomes an active docking site once a condensate has formed.
What happens when the filaments are removed
To see how filaments affect the droplet’s physical behavior, the team engineered PopZ mutants that could still form hexamers but could no longer stack them into filaments. These variants did produce condensates, but with strikingly different properties. Instead of forming neat spheres that sit on the surface, the droplets flattened and spread, indicating lower surface tension and stronger wetting of their surroundings. Within these filament-poor condensates, both PopZ itself and its client proteins moved much more quickly, as measured by fluorescence recovery after photobleaching. In other words, removing filaments made the droplets softer and leakier. When such mutants were introduced into Caulobacter cells, they disrupted normal PopZ behavior at the poles, interfered with chromosome anchoring, and severely impaired growth, even when wild-type PopZ was still present. Other engineered variants showed the opposite mismatch: they could form filaments but failed to condense efficiently, and these too were unable to fully rescue PopZ’s cellular role.
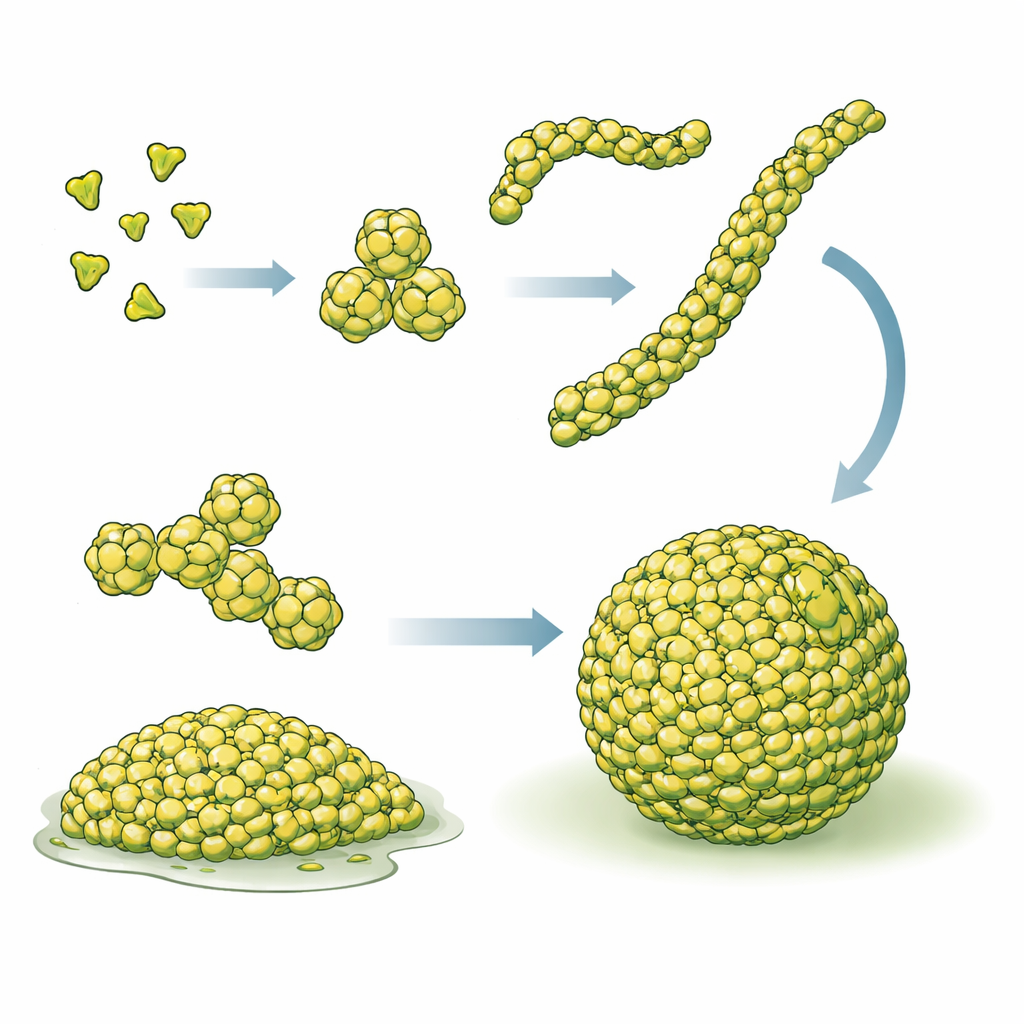
A recipe for functional cellular droplets
The work leads to a clear message for non-specialists: it is not enough for a protein like PopZ to simply clump together, nor is it enough to form filaments in isolation. What matters for cell function is a very specific ultrastructure — condensates that are themselves made of short, interconnected filaments. This filamentous framework boosts the number of contact points between molecules, raises the droplet’s surface tension, slows the motion of key clients, and provides a built-in molecular switch that turns client binding on only in the dense phase. By tracing this chain of cause and effect from amino-acid sequence to molecular assemblies to whole-cell behavior, the study offers a general blueprint for how cells may tune the “feel” of their soft internal droplets to control vital processes.
Citation: Scholl, D., Boyd, T., Latham, A.P. et al. The filamentous ultrastructure of the PopZ condensate is required for its cellular function. Nat Struct Mol Biol 33, 420–432 (2026). https://doi.org/10.1038/s41594-025-01742-y
Keywords: biomolecular condensates, protein filaments, cell polarity, phase separation, bacterial cell cycle