Clear Sky Science · en
TGFβ signaling mediates microglial resilience to spatiotemporally restricted myelin degeneration
Why the Brain’s Wiring Needs Caregivers
As we age, the “insulation” around our nerve fibers—called myelin—naturally wears down. This insulation is vital for fast, reliable communication between the brain and body. The new study explores why one specific highway of nerve fibers in the spinal cord, the dorsal column, is unusually vulnerable to this wear and tear, and how a built‑in immune system brake, a signaling pathway called TGFβ, keeps damage from spiraling out of control. Understanding this hidden safeguard matters because its failure may help explain certain spinal cord disorders and could influence how we treat brain and cancer diseases in older adults.
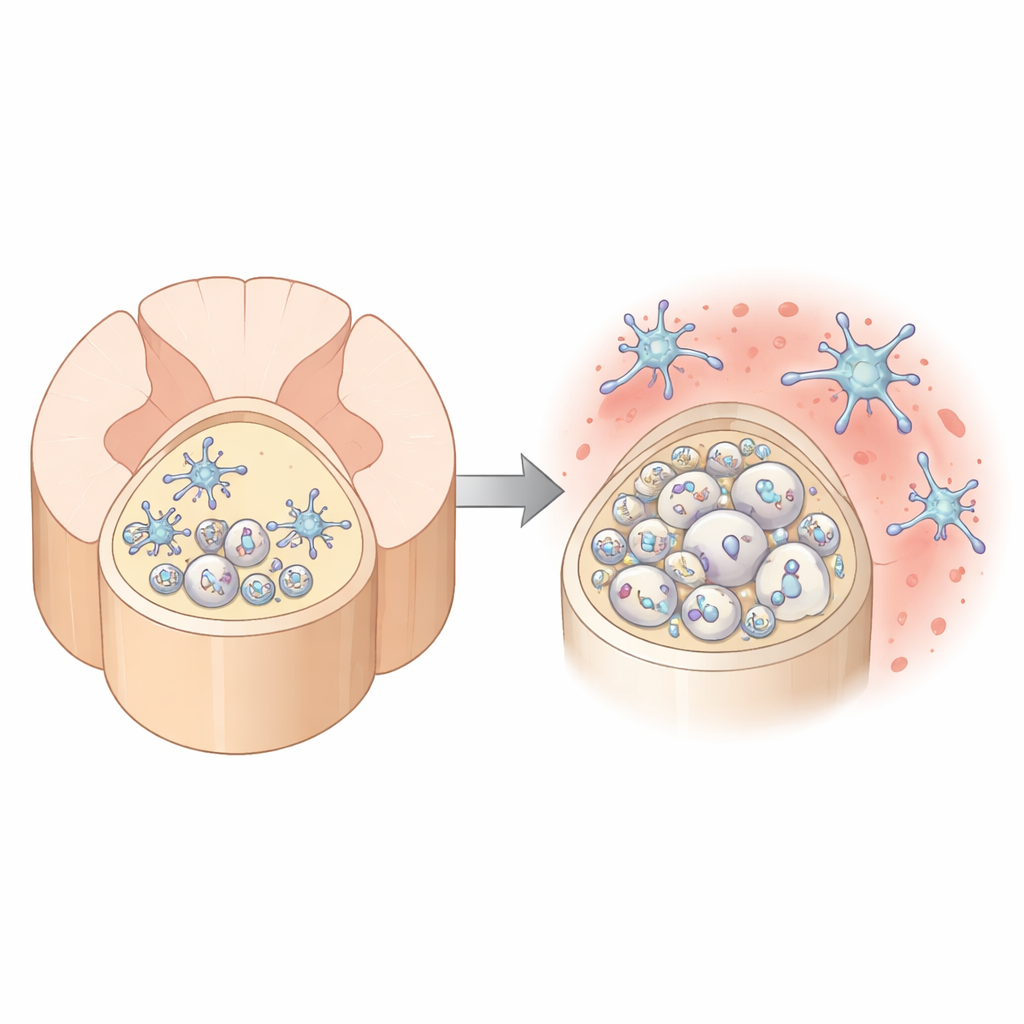
Where Aging Hits the Spinal “Superhighway” Hardest
The authors first compared two major white‑matter tracks in the mouse spinal cord: the dorsal column in the back and the ventral column in the front. Using high‑resolution electron microscopy, they found that with age, myelin in the dorsal column becomes increasingly distorted. The normally tight wraps around nerve fibers loosen, bulge outward, or detach, leaving enlarged spaces between nerve and insulation and a rise in outright myelin breakdown. In contrast, the ventral column stays relatively stable. The dorsal region also packs many more, but thinner, axons, making it a particularly demanding environment for the cells that build and maintain myelin.
Immune Watchdogs Under Growing Pressure
Surrounding these fibers are microglia, the resident immune cells of the brain and spinal cord. They constantly patrol tissue, clearing debris and fine‑tuning connections. As dorsal column myelin degenerates with age, these microglia start to look and act different: they upregulate genes linked to immune activation, accumulate oily droplets inside, and show signs of having engulfed more damaged myelin. At the same time, the tissue environment in the dorsal column becomes strongly enriched in TGFβ1, a signaling molecule known to keep microglia in a restrained, home‑protective state. Microglia themselves are the major source of this TGFβ1, suggesting they are trying to hold themselves in check even as stress signals mount.
What Happens When the Safety Brake Fails
To probe how important this brake is, the researchers genetically disabled TGFβ signaling specifically in microglia of adult mice, either by removing the receptor they use to sense TGFβ or by preventing them from making TGFβ1. In both cases, the outcome was strikingly similar and highly localized: microglia in the dorsal column became overabundant, highly activated, and took on a foamy appearance packed with ingested myelin. Myelin in this region was stripped away, axons showed signs of degeneration, and mice developed worsening movement and coordination problems, particularly as they aged. Other spinal regions were far less affected, revealing a region‑specific dependence on this pathway.
A Special Microglial Subtype and Stressed Myelin Makers
Single‑nucleus RNA sequencing allowed the team to catalog individual cell types as disease unfolded. They discovered a distinct subset of microglia—termed TGFβ‑sensitive microglia—that expanded dramatically when TGFβ signaling was removed. These cells expressed high levels of genes associated with strong inflammatory responses, intense myelin eating, and lipid handling, and they were concentrated in the dorsal column. At the same time, the population of mature myelin‑forming cells, oligodendrocytes, shifted. Healthy subtypes declined, while a “disease‑associated” oligodendrocyte group expanded in the damaged dorsal column but failed to rebuild functional myelin, leaving axons exposed despite an apparent attempt at repair.
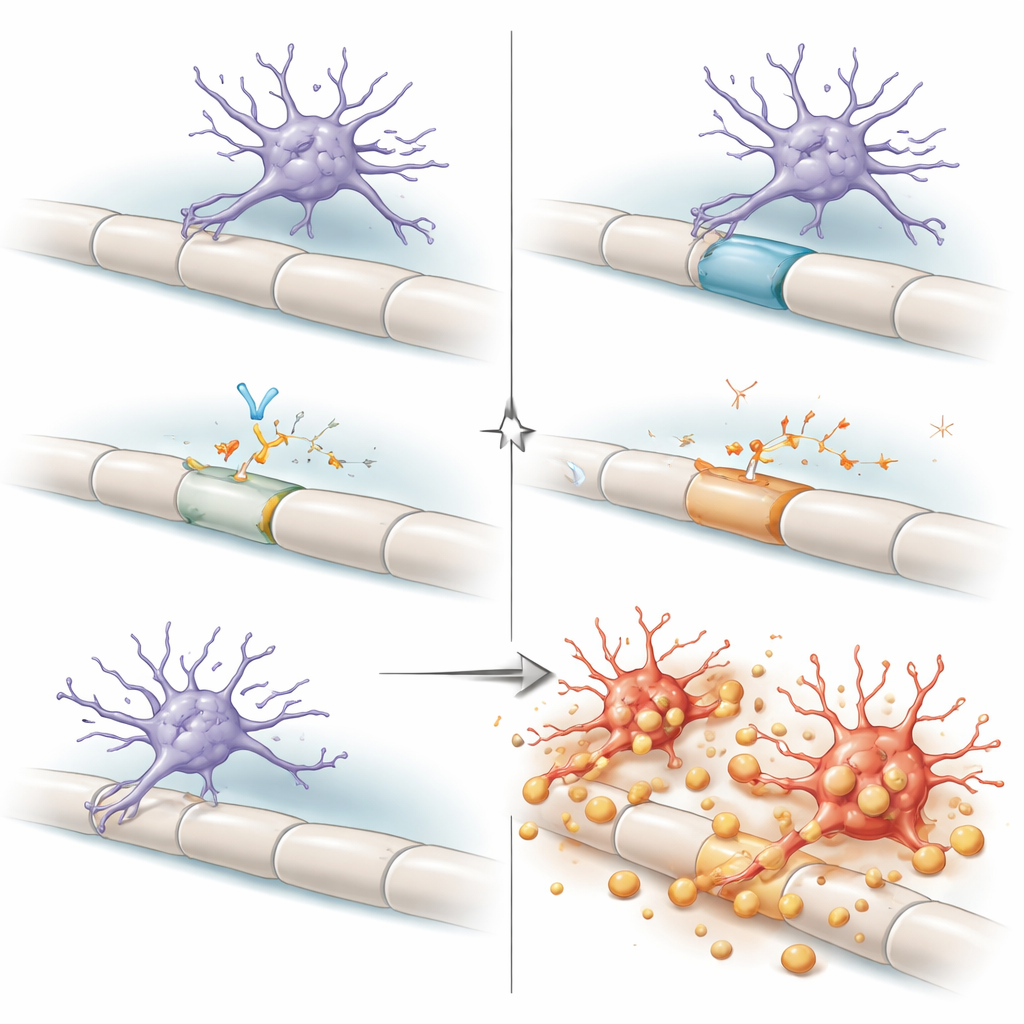
Implications for Aging and Future Therapies
Viewed together, the findings paint the dorsal column as a high‑stress neighborhood where aging myelin and overworked support cells push microglia toward a tipping point. Under normal conditions, an autocrine loop—microglia producing and sensing their own TGFβ1—keeps them in a “primed but resilient” state, able to handle ongoing debris without attacking relatively intact myelin. When this loop is broken, microglia shift into a maladaptive mode, aggressively stripping myelin and driving neurological decline. For a lay audience, the key message is that the brain’s immune cells are not simply good or bad; their behavior depends heavily on local wiring demands and on molecular brakes like TGFβ. Because TGFβ‑blocking drugs are being developed for cancer and some brain diseases, this work raises an important caution: disrupting this pathway, especially in older people, could unintentionally damage vulnerable spinal cord regions by unleashing microglia on the very insulation our nerves need to function.
Citation: Zhu, K., Liu, Y., Min, JH. et al. TGFβ signaling mediates microglial resilience to spatiotemporally restricted myelin degeneration. Nat Neurosci 29, 617–631 (2026). https://doi.org/10.1038/s41593-025-02161-4
Keywords: microglia, myelin aging, spinal cord dorsal column, TGF beta signaling, neuroinflammation