Clear Sky Science · en
Next-generation multicolor indicators for in vivo imaging of norepinephrine
Seeing a Hidden Brain Messenger
Norepinephrine is a chemical in the brain that quietly shapes how we wake up, pay attention, form memories and respond to stress. Until recently, scientists could only glimpse its activity with slow or imprecise tools. This paper introduces a new pair of glowing markers that let researchers watch norepinephrine ebb and flow in the living brain, in real time and in fine detail. These advances could deepen our understanding of sleep, anxiety, learning and neurodegenerative disease.
Why Tracking This Signal Has Been So Hard
Norepinephrine is released by a small group of brainstem cells that send fibers throughout the brain, changing how circuits behave rather than simply turning them on or off. Classic measurement methods, such as tiny chemical probes or implanted cells that respond to norepinephrine, either blur events over many seconds or cannot distinguish it cleanly from related molecules. A newer strategy uses modified cell-surface receptors that brighten when they bind norepinephrine, allowing light-based readout. But first-generation versions were relatively dim, especially in the red part of the spectrum, and were not flexible enough for experiments that needed to combine several colors at once.
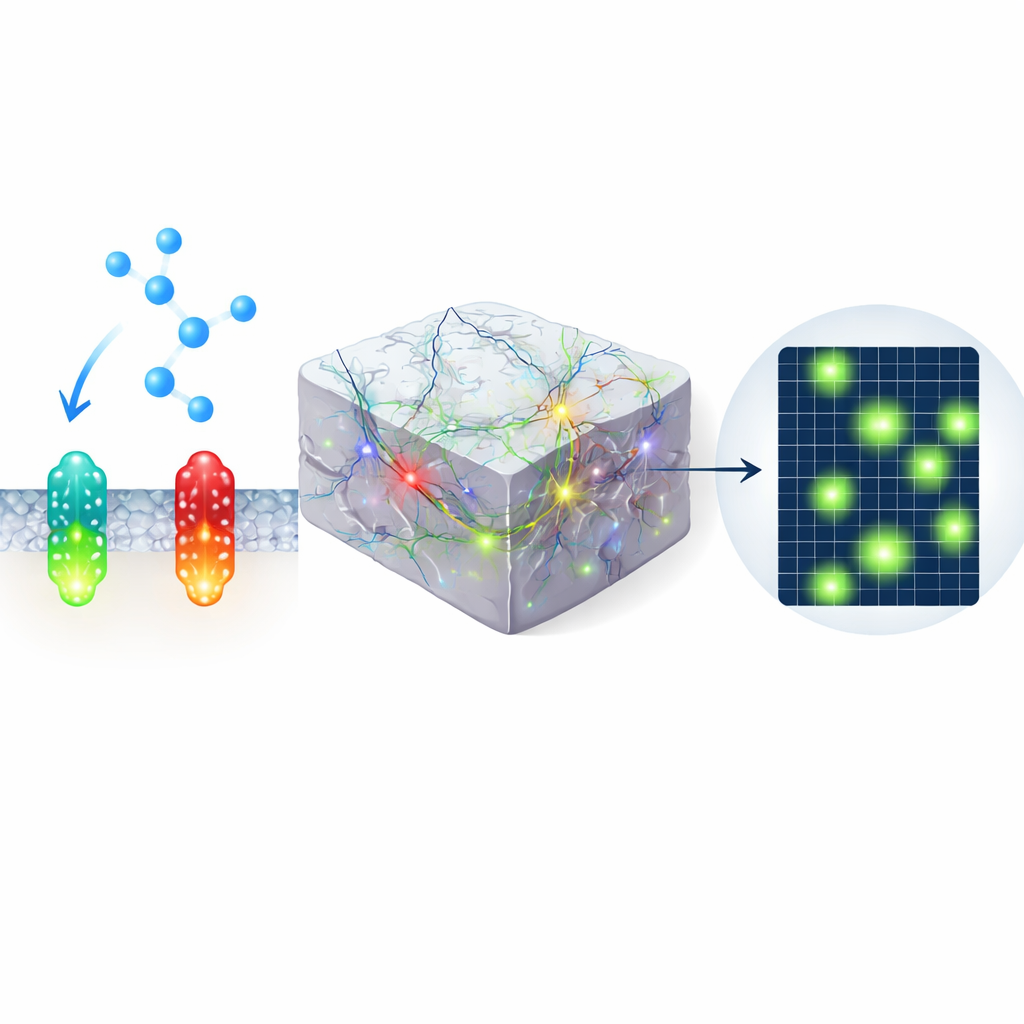
Building Brighter Green and Red Watchdogs
The authors engineered improved green and red indicators, called nLightG2 and nLightR2, by combining pieces from earlier dopamine and norepinephrine sensors and systematically testing dozens of mutations. These changes made the sensors much brighter when norepinephrine was present, without greatly changing their baseline glow. In cell cultures, the new tools showed several-fold larger responses to norepinephrine than previous versions, reacted within tens of milliseconds and reset within less than a second. They also barely responded to other brain chemicals, such as dopamine, and did not activate the cells’ own internal signaling pathways, an important safety check to ensure they are observers rather than participants.
Proving Their Power in Brain Tissue
Next, the team introduced the sensors into slices of mouse brain and used two-photon microscopy, which can see deep into tissue, to compare old and new designs. When norepinephrine was puffed onto the tissue, or when local fibers were electrically stimulated to release it naturally, nLightG2 and nLightR2 produced much larger and more easily detectable flashes than earlier sensors. The green and red tools performed similarly fast, meaning that color choice no longer requires a trade-off in speed. This higher sensitivity allowed the researchers to map where norepinephrine spread in space, rather than simply knowing that it had been released somewhere in the field of view.
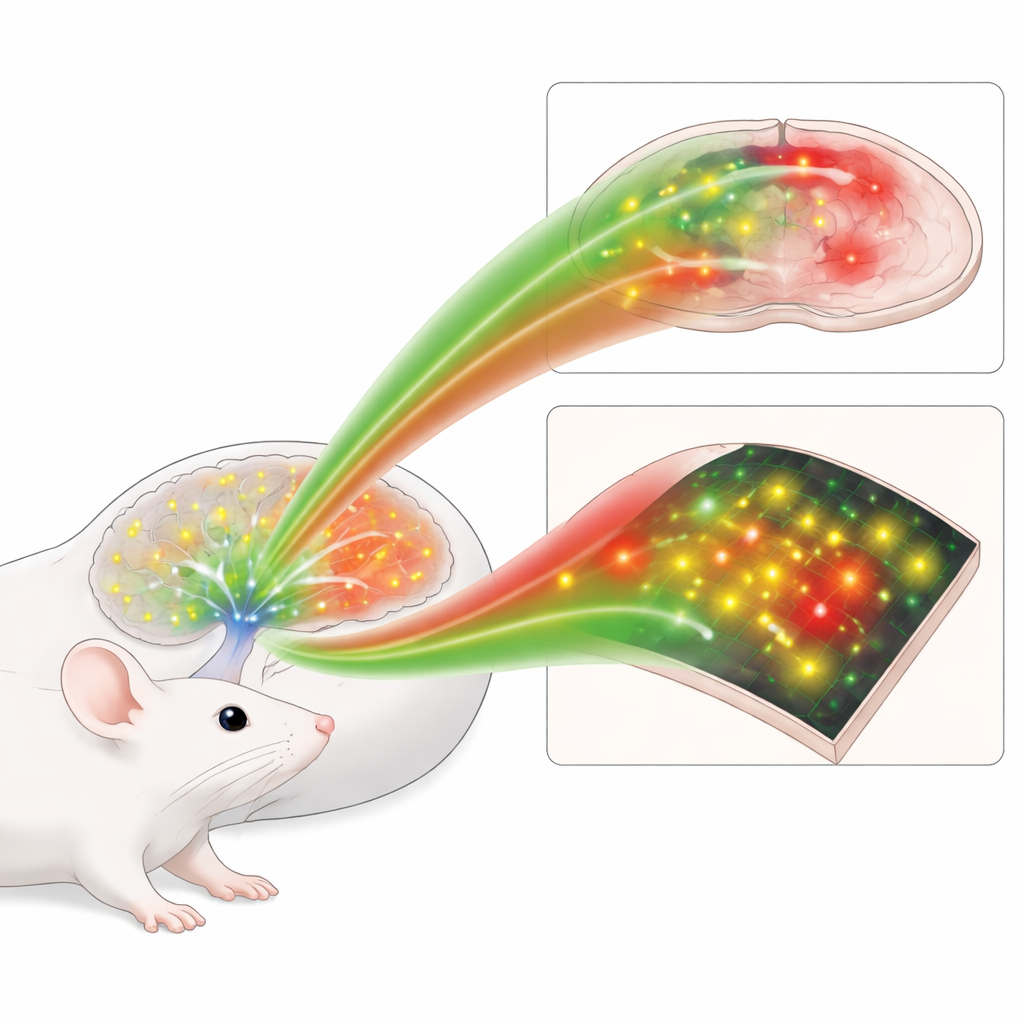
Watching Brain States, Fear and Navigation in Action
The real promise of these tools lies in living animals. Using hair-thin optical fibers, the authors combined the red norepinephrine indicator with a green calcium sensor that reports nerve cell firing. In the sleep center of the brain, they saw that bursts of activity in norepinephrine-producing cells during deep sleep were closely followed by rises in local norepinephrine levels, event by event. In the amygdala, a brain region important for emotion, the green norepinephrine sensor revealed that a harmless tone came to trigger a sustained rise in norepinephrine once it had been paired with a mild shock, mirroring the strengthening of fear memories. In the hippocampus, which helps map space, the red sensor was imaged together with a green astrocyte calcium sensor while mice ran through a virtual corridor for water rewards. Here, astrocyte activity near a reward site was tightly linked to local norepinephrine surges, hinting at a dialogue between this chemical and support cells during rewarding experiences.
Revealing Tiny Pockets of Activity in Visual Cortex
In another set of experiments, the authors expressed the green sensor in the visual cortex of awake mice and imaged it with two-photon microscopy while the animals viewed looming stimuli and switched between rest and running. Instead of a smooth wash of signal, they found brief, highly localized patches of increased fluorescence—microdomains—scattered across the field of view. Some microdomains responded preferentially to the visual threat, others to movement, and many lit up spontaneously. These patterns were largely invisible when using an older green sensor or a mutated nonbinding control, underscoring the improved sensitivity of nLightG2 and suggesting that norepinephrine sculpts brain activity in a much more fine-grained way than previously appreciated.
What This Means for Brain Research
Together, these results show that nLightG2 and nLightR2 form a powerful toolkit for watching norepinephrine in the living brain, across scales ranging from single microdomains to entire behavioral states. Because they come in distinct colors and can be combined with other fluorescent reporters, scientists can now track norepinephrine alongside electrical or calcium signals in specific cell types, during sleep, learning or stress. This ability to see when and where this key neuromodulator acts may eventually clarify how it supports healthy attention and memory, and how its disruption contributes to conditions such as anxiety, depression and neurodegenerative disease.
Citation: Rohner, V.L., Curreli, S., Lamothe-Molina, P.J. et al. Next-generation multicolor indicators for in vivo imaging of norepinephrine. Nat Methods 23, 636–652 (2026). https://doi.org/10.1038/s41592-026-03006-z
Keywords: norepinephrine, neuromodulation, genetically encoded sensors, two-photon imaging, sleep and learning