Clear Sky Science · en
A quantitative DOPA decarboxylase biomarker for diagnosis in Lewy body disorders
Why better brain diagnoses matter
Dementia with Lewy bodies and Parkinson’s disease can look a lot like Alzheimer’s or normal aging, especially in the early stages. Yet the wrong diagnosis can lead to dangerous treatments and missed chances to plan care or join clinical trials. This study introduces a promising lab test based on a molecule called DOPA decarboxylase (DDC) found in the fluid that bathes the brain and spinal cord. The work suggests this simple fluid measurement could help doctors tell Lewy body–related diseases apart from Alzheimer’s and from healthy aging with far greater confidence.
A hidden signature in brain fluid
Lewy body disorders, which include dementia with Lewy bodies and Parkinson’s disease, are driven by clumps of a protein called alpha‑synuclein that damage nerve cells, especially those using the chemical messenger dopamine. DDC is an enzyme that helps make dopamine and serotonin. Earlier, broad protein surveys hinted that DDC levels are often higher in the clear cerebrospinal fluid (CSF) of people with Lewy body disorders, but those findings relied on research-only methods that were not ready for routine hospital laboratories. The authors set out to build precise, scalable tests that could measure the actual concentration of DDC in CSF and blood, and then to see how well those measurements track with diagnosis, symptoms, brain scans and, in some patients, changes seen at autopsy.
Building a reliable test
The team developed two highly sensitive DDC immunoassays—one running on a platform called Ella and another on a system known as Simoa. Both use pairs of antibodies to capture DDC molecules and generate a measurable signal, and both were carefully validated for accuracy, consistency and stability under different storage and handling conditions. The assays could detect DDC in every CSF sample tested, and results from the two platforms, as well as from an earlier proximity extension assay, lined up closely. One version of the Simoa assay was also tuned to work in blood plasma and serum, although the main focus of the study remained CSF, which more directly reflects changes in the brain.
Clear separation between diseases
Using these tools, the researchers measured CSF DDC in more than 700 people across six independent cohorts from several countries. In every cohort, people with dementia with Lewy bodies or Parkinson’s disease had substantially higher DDC levels in CSF—up to about two-and-a-half times those of healthy controls and clearly higher than in Alzheimer’s disease. When DDC was combined with a person’s age and sex in standard diagnostic models, the test distinguished Lewy body disorders from healthy controls with areas under the curve near or above 0.9, meaning excellent accuracy. It also separated them from Alzheimer’s disease with high performance, including in a group of patients whose diagnoses were confirmed either by a highly specific alpha‑synuclein “seed” test or by examination of the brain after death. In contrast, DDC levels in blood did not differ reliably between diagnostic groups and were strongly influenced by dopaminergic medications, making blood less suitable for diagnosis.
Linking the test to symptoms and brain changes
Beyond diagnosis, the team explored what higher CSF DDC levels might reflect inside the brain. Within Lewy body and Parkinson’s groups, raised DDC was linked to the presence—but not the severity—of motor symptoms and to visual hallucinations, two hallmark features of Lewy body dementia. DDC levels did not track consistently with memory scores or with standard Alzheimer’s CSF markers, suggesting they carry information that is partly independent of classic Alzheimer’s changes. In people who had detailed dopamine transporter imaging, higher DDC was associated with a pattern of more widespread dopaminergic damage resembling that seen in Lewy body dementia. In an autopsy cohort, higher CSF DDC went hand in hand with more extensive Lewy body pathology, especially in the substantia nigra, a midbrain region crucial for movement.
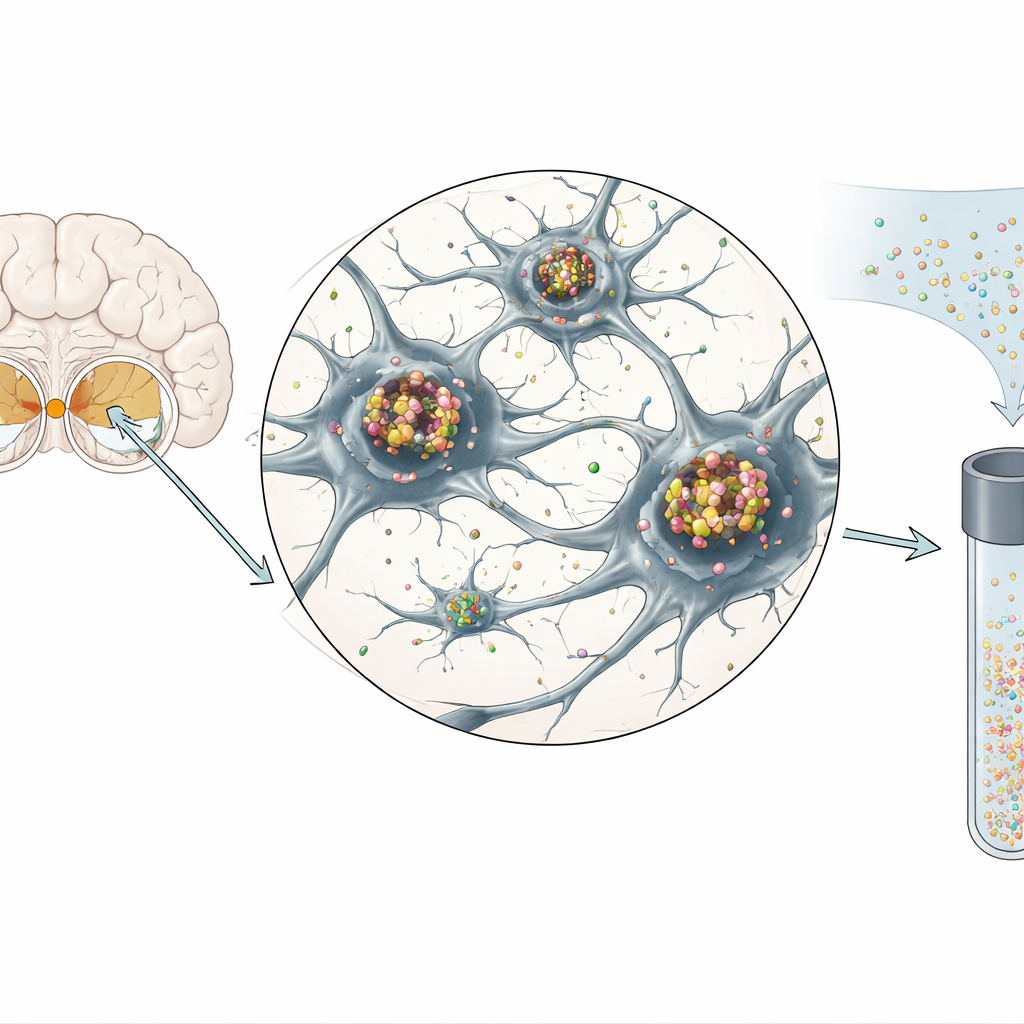
Seeing DDC inside vulnerable neurons
To connect the fluid signal to actual brain tissue, the researchers examined postmortem samples from people with dementia with Lewy bodies, Parkinson’s disease with dementia, and neurologically healthy controls. Using the same antibody employed in the fluid tests, they showed that DDC is abundant in dopamine-producing neurons of the substantia nigra and in serotonin-producing neurons of the brainstem raphe nuclei. In patients with Lewy body disorders, DDC was not only present in surviving neurons but also concentrated within Lewy bodies and related abnormal structures, and it occupied the same cellular spaces as disease‑related, phosphorylated alpha‑synuclein. Fewer neurons remained overall, but those that did often showed strong DDC signal, consistent with either a compensatory buildup of the enzyme or leakage from damaged cells into the surrounding fluid.
What this means for patients
Taken together, the study shows that DDC levels in cerebrospinal fluid are robustly and specifically increased in dementia with Lewy bodies and Parkinson’s disease, correlate with the spread of Lewy body pathology in the brain, and can be quantified reliably using lab platforms already common in clinical settings. While the test does not replace careful clinical evaluation or imaging, it could become a valuable tool to support diagnosis, help separate Lewy body disorders from Alzheimer’s disease, and better select patients for clinical trials. The authors propose CSF DDC as a practical new biomarker that brings us closer to clear, biology-based diagnoses for some of the most challenging forms of dementia.
Citation: Bolsewig, K., Bellomo, G., Hok-A-Hin, Y.S. et al. A quantitative DOPA decarboxylase biomarker for diagnosis in Lewy body disorders. Nat Med 32, 1073–1084 (2026). https://doi.org/10.1038/s41591-026-04212-0
Keywords: Lewy body dementia, Parkinson’s disease, cerebrospinal fluid biomarker, DOPA decarboxylase, alpha-synuclein