Clear Sky Science · en
Single-cell atlas of the developing Down syndrome brain cortex
Why this research matters
Down syndrome is the most common genetic cause of intellectual disability, yet we still know little about how it changes the developing human brain before birth. This study zooms in to the level of individual cells in the fetal cortex—the brain region crucial for thinking and memory—to map what goes wrong, when it starts, and which molecular switches might be targeted to help.
Looking closely at the growing brain
The researchers analyzed nearly a quarter of a million cells taken from the cerebral cortex of 15 fetuses with Down syndrome and 15 without, between 10 and 20 weeks after conception. Using advanced "single-cell" methods, they measured both which genes were switched on and how accessible the DNA was in each cell. This allowed them to identify all major cell types present at this mid-gestation stage—such as stem-like progenitor cells, different kinds of excitatory and inhibitory neurons, and early glial cells—and to compare their abundance and gene activity between Down syndrome and typical brains.
Early changes in key thinking cells
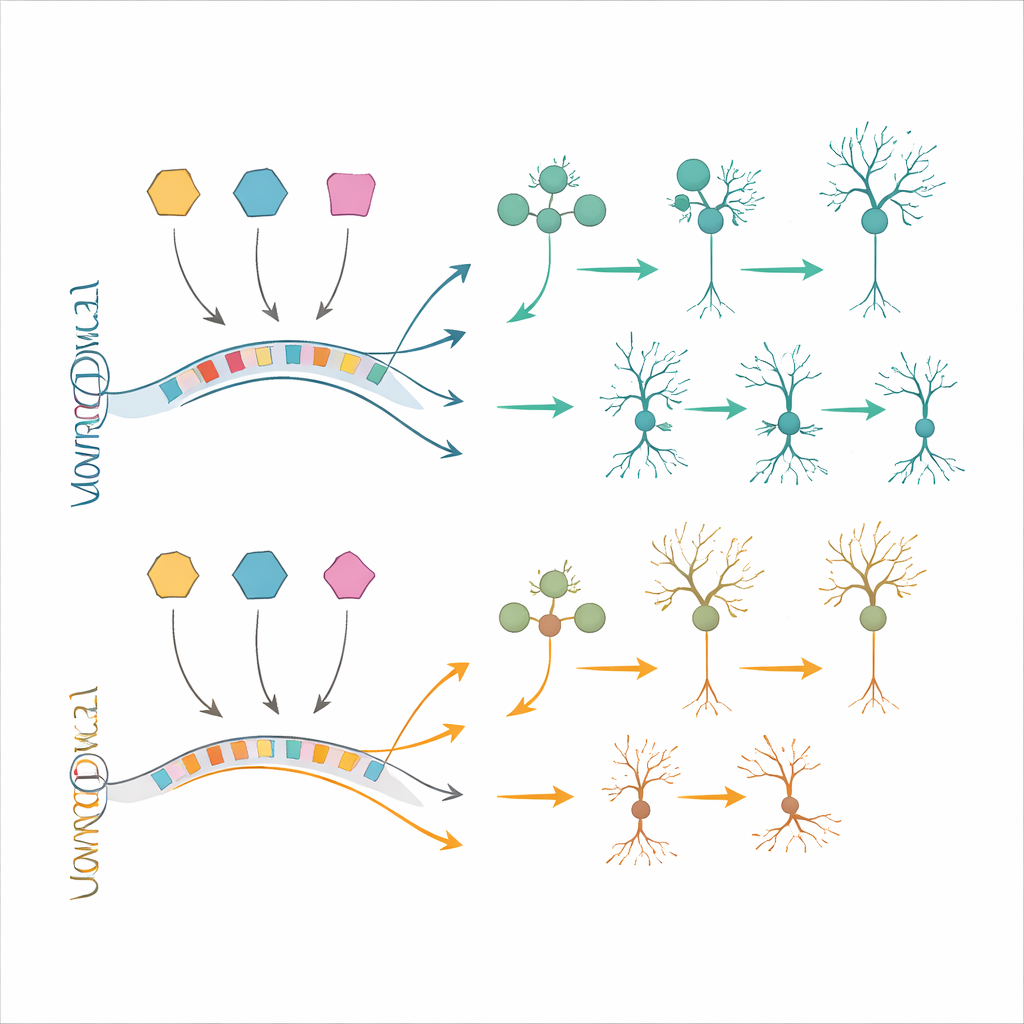
Most broad cell classes were present in similar numbers in both groups during this early window. However, the team found a striking and selective shortfall of one particular kind of excitatory neuron that normally occupies layer 4 of the cortex and is important for processing incoming information. These neurons are defined by proteins called RORB and FOXP1. In fetuses with Down syndrome, RORB–FOXP1 neurons were already reduced by mid-gestation, especially between 16 and 20 weeks, while other neuron types looked relatively spared. This suggests that problems in generating or maturing this subset of cells begin in utero and may contribute directly to later cognitive difficulties.
Disrupted genetic programs and master switches
Beyond cell counts, the study revealed that hundreds of genes were subtly misregulated, particularly in excitatory neurons and their progenitors. Many of these genes are involved in building the forebrain, shaping neuron branches, forming connections, and supporting higher brain functions. Rather than just the extra copies of roughly 200 genes on chromosome 21 acting alone, the data point to a disturbed network of gene regulation. Using an approach that combines gene activity with DNA accessibility, the authors mapped out regulatory circuits and highlighted three transcription factors—BACH1, PKNOX1 and GABPA—encoded on chromosome 21 as dosage-sensitive "hubs." These molecules appear to influence other critical regulators of cortical development, including factors already linked to intellectual disability, and help explain how a modest 1.5-fold increase in gene dosage can ripple through entire developmental programs.
Testing rescue strategies in cells and living brains
To see whether these molecular changes could be corrected, the team turned to stem-cell models. They generated neural progenitor cells and neurons from induced pluripotent stem cells carrying trisomy 21 and from matched control cells. Many of the gene-expression changes seen in fetal tissue reappeared in these lab-grown cells, confirming the relevance of the models. The researchers then used antisense oligonucleotides—short strands of engineered DNA-like material—to dial down BACH1, PKNOX1 or GABPA toward normal levels. This partial normalization of the overexpressed transcription factors led to partial recovery of several downstream genes, including ones known to be involved in intellectual disability and neuronal differentiation. In a complementary approach, they transplanted human trisomy 21 neural cells into mouse brains and let them mature in vivo. These grafts reproduced additional Down syndrome–like features, such as altered neuron–glia balance and gene changes that were not fully captured in dishes, offering a powerful testing ground for future therapies.
What this means for the future
Taken together, the work provides a detailed atlas of how Down syndrome reshapes the genetic landscape of the developing cortex at single-cell resolution. For a general reader, the key message is that the extra chromosome does not simply add a few rogue genes; it nudges many interconnected molecular switches, leading to early and selective shortages of certain thinking-related neurons. By pinpointing a small set of chromosome 21 transcription factors as central players—and showing that their effects can be partially reversed in human cells—the study opens the door to more targeted strategies aimed at improving brain development and function in Down syndrome.
Citation: Lattke, M., Tan, W.L., Sukumaran, S.K. et al. Single-cell atlas of the developing Down syndrome brain cortex. Nat Med 32, 1061–1072 (2026). https://doi.org/10.1038/s41591-026-04211-1
Keywords: Down syndrome, fetal brain development, single-cell genomics, cortical neurons, transcription factors