Clear Sky Science · en
CD19 CAR-T cells for treatment-refractory autoimmune diseases: the phase 1/2 CASTLE basket trial
Why a one-time treatment matters
Many people with serious autoimmune diseases live on a treadmill of constant medication, flare after flare and mounting side effects. This study explores a radically different idea: using a single infusion of reprogrammed immune cells to "reboot" the immune system so that it stops attacking the body. For patients with severe lupus, hardening of the skin and organs, or muscle inflammation who have run out of options, such a reset could mean long-lasting relief without daily drugs.
Three tough diseases, one common problem
Systemic lupus erythematosus, systemic sclerosis and idiopathic inflammatory myopathies may look different on the surface—affecting joints, skin, lungs, kidneys or muscles—but they share a core flaw: certain immune cells called B cells misfire and help drive chronic inflammation. Standard treatments, from steroids to powerful immune-suppressing drugs and antibody infusions, can calm symptoms but rarely remove the root cause. Many patients cycle through multiple therapies that only partly work or trigger intolerable side effects, leaving them with ongoing organ damage and poor quality of life.
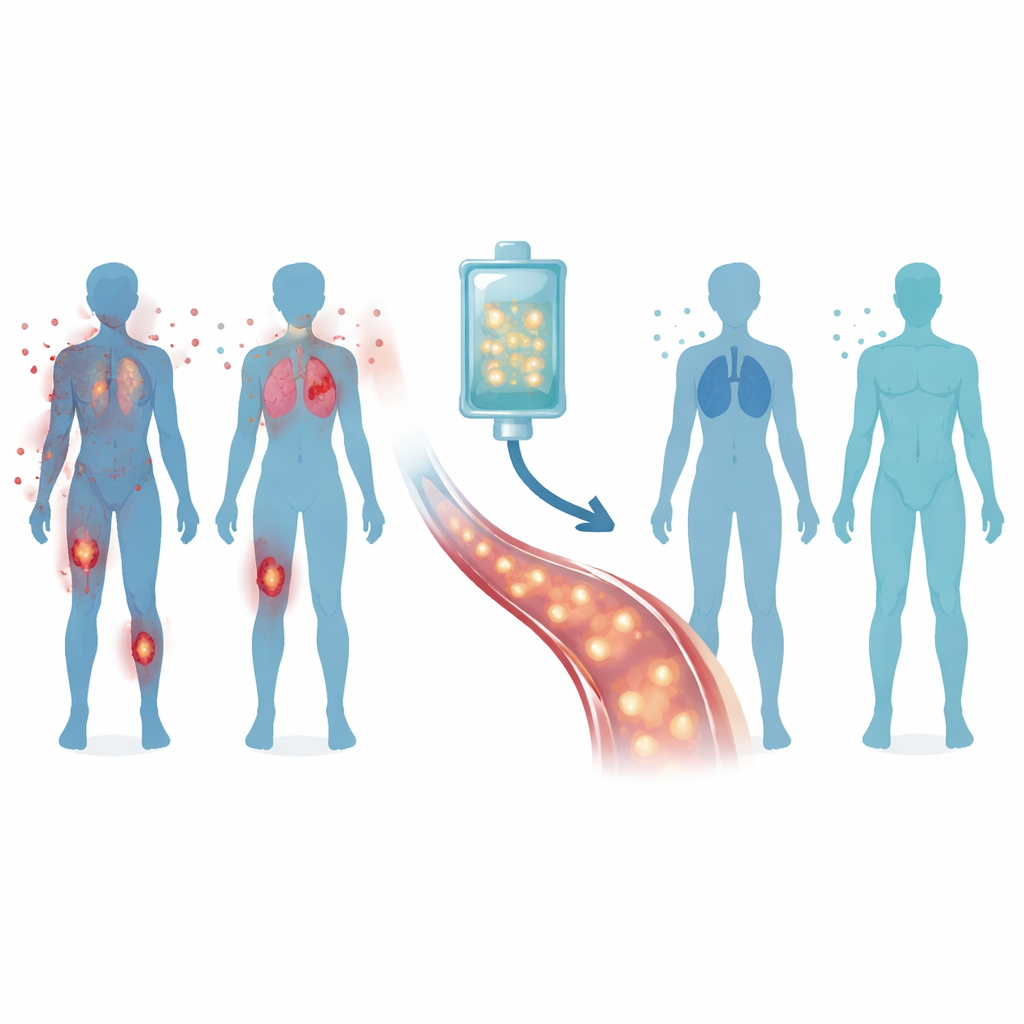
Reprogramming the body’s defenders
The CASTLE trial tested a personalized cell therapy called Zorpo-cel in 24 adults with severe, treatment-resistant forms of these three diseases. Doctors first collected each patient’s T cells, the "soldiers" of the immune system, and in the lab equipped them with a new sensor—a chimeric antigen receptor—that recognizes a marker called CD19 found on most B cells. After a short course of chemotherapy to make space in the immune system, patients received a single infusion of these engineered cells, known as CD19 CAR-T cells. The hope was that they would hunt down and remove the misbehaving B cells, allowing a healthier set of cells to grow back.
Safety first, then signs of relief
Because this treatment can be intense in cancer patients, the main goal of this early-stage trial was safety. The researchers watched closely for a dangerous inflammatory reaction called cytokine release syndrome and for brain-related side effects. In CASTLE, nearly three-quarters of patients had only mild, short-lived fevers and inflammation; no severe cytokine storms, no brain toxicity and no lasting blood cell problems were seen. The most common issues were temporary drops in white blood cells and mild infections, which were managed with standard care. One patient developed kidney injury linked to a virus reactivation and a flare of underlying disease, prompting tighter rules to control disease activity before infusion.
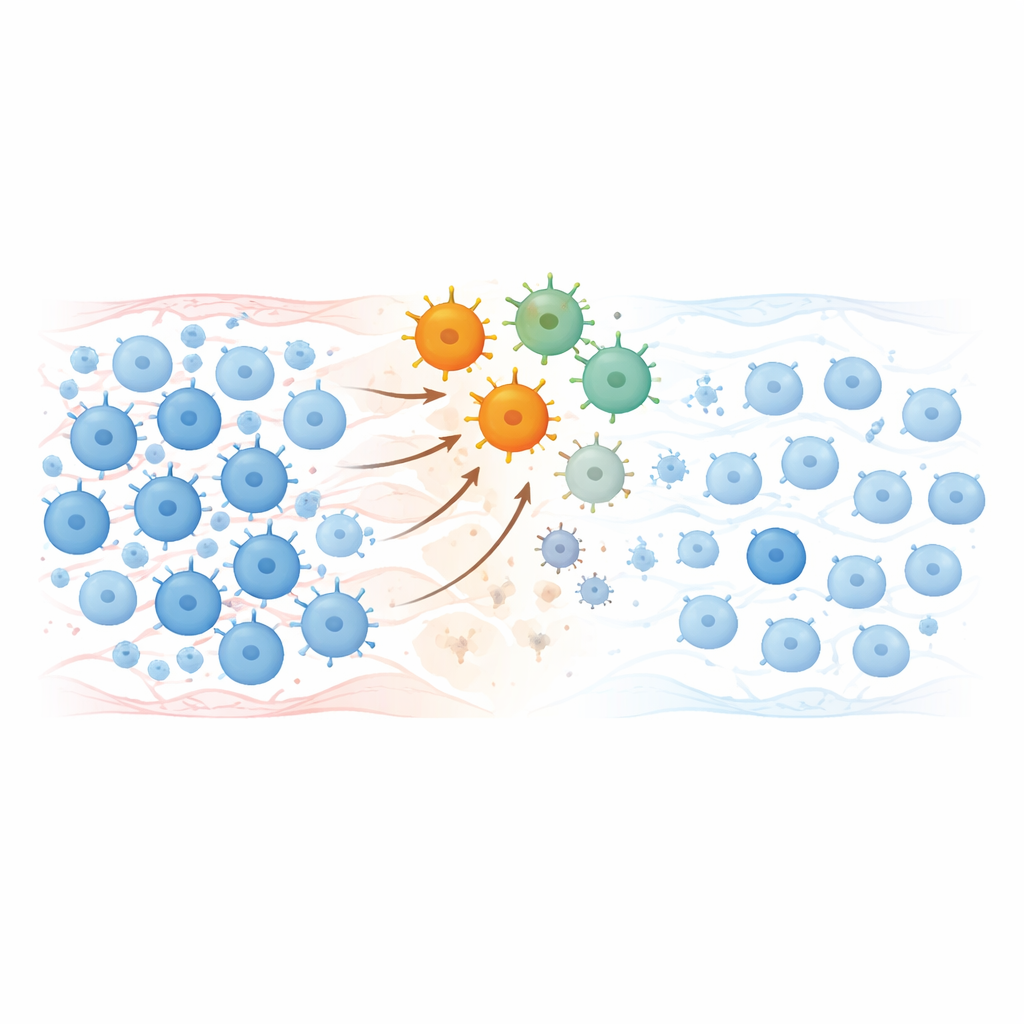
Resetting the immune system
Although safety was the top priority, the clinical improvements were striking. After six months, 22 of 24 patients met predefined success criteria tailored to their disease: most lupus patients reached a strict remission definition, all systemic sclerosis patients had no worsening of lung disease and showed better lung function and softer skin, and most patients with muscle inflammation gained strength and had lower muscle enzyme levels. Importantly, all participants were able to stop steroids and other immune-suppressing drugs and stay off them during the six-month study and beyond, with no relapses seen over a median follow-up of about a year.
What happens inside the blood
Lab tests showed that the infused CAR-T cells expanded for a few days, wiped out circulating B cells and then gradually disappeared over a couple of months. When B cells later returned, their population looked different: naive, "fresh" cells dominated, while memory cells and antibody-producing precursors that often harbor autoimmune memory were greatly reduced. Many harmful autoantibodies fell to very low or undetectable levels, yet protective antibodies from past vaccinations against infections like measles or tetanus largely remained, suggesting a targeted reset rather than a full wipe of immune memory.
A glimpse of a new treatment era
For people living with severe autoimmune disease, the idea of a one-time treatment that brings durable, drug-free remission has long seemed out of reach. This trial suggests that engineered T cells can safely silence rogue B cells across three different illnesses and may allow the immune system to rebuild in a calmer, less self-destructive form. CASTLE is an early, relatively small study, and larger pivotal trials are needed to confirm how long the benefit lasts and which patients stand to gain the most. Still, it points toward a future in which resetting the immune system—rather than endlessly suppressing it—could become a realistic goal.
Citation: Müller, F., Hagen, M., Wirsching, A. et al. CD19 CAR-T cells for treatment-refractory autoimmune diseases: the phase 1/2 CASTLE basket trial. Nat Med 32, 1142–1151 (2026). https://doi.org/10.1038/s41591-025-04185-6
Keywords: CAR-T cell therapy, autoimmune disease, systemic lupus erythematosus, systemic sclerosis, B cells