Clear Sky Science · en
Humoral IgG1 responses to tumor antigens underpin clinical outcomes in immune checkpoint blockade
Why this matters for cancer patients
Immunotherapy has transformed cancer care by helping the body’s own defenses attack tumors, yet only some patients benefit. Most attention has focused on one set of immune warriors—T cells—but this study reveals that another group, antibody-producing B cells, can make the difference between success and failure. By uncovering how a specific kind of antibody response, called IgG1, tracks with better outcomes, the work points toward new ways to predict who will benefit from immunotherapy and how to make these treatments work for more people.
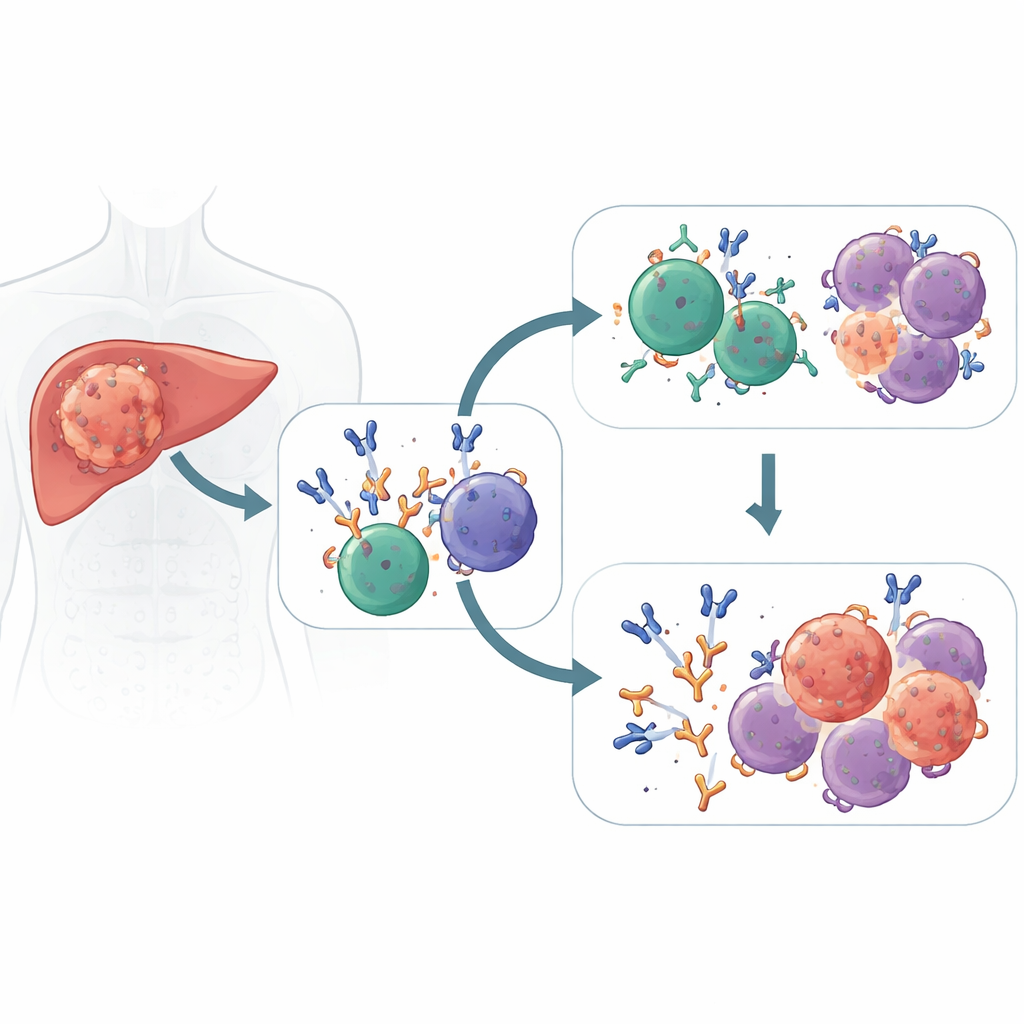
Two lines of defense working together
Modern drugs known as immune checkpoint blockers release the “brakes” on T cells so they can recognize and kill cancer cells. The researchers studied people with liver cancer receiving these drugs before surgery and discovered that good responders did not rely on T cells alone. Their tumors were also packed with plasma cells—mature B cells that act as miniature antibody factories—producing a dominant wave of IgG1 antibodies aimed at cancer targets. In contrast, patients whose tumors did not shrink had more idle or dysfunctional B cells and fewer of these specialized plasma cells inside their cancers.
Following the antibody trail through the body
To understand where these helpful plasma cells come from, the team combined single-cell sequencing with detailed tracking of the unique antibody “barcodes” each B cell carries. They found that successful patients had families of closely related IgG1-producing cells expanding both in the tumor and in nearby lymph nodes—the immune system’s command centers. The same clonal signatures appeared before treatment and then grew after therapy, indicating that checkpoint blockade amplifies pre-existing, tumor-aware B cell clones rather than creating entirely new ones. This suggested a coordinated circulation of trained antibody producers between lymph nodes and tumor sites.
How antibodies reshape the tumor neighborhood
Microscopic imaging and spatial gene-mapping showed that, in responders, plasma cells did not sit at the tumor’s margins—they infiltrated deep into the cancer alongside killer T cells and helpful macrophages. These regions were rich in signals that promote B cell activation and antibody production, forming immune “neighborhoods” optimized for attack. Non-responders, by contrast, accumulated memory-like B cells and regulatory immune cells in fibrous stromal pockets that dampened effective responses. Across several independent patient groups and different immunotherapy combinations, a skew toward IgG1-producing plasma cells consistently marked better tumor control and slower disease progression.
Antibodies that recognize cancer-specific flags
Because plasma cells secrete antibodies into the bloodstream, the investigators asked whether these molecules could be detected in blood samples. In many responders, they found high levels of IgG1 antibodies targeting so-called cancer/testis antigens—proteins like NY-ESO-1 that are usually absent from normal tissues but reappear in tumors. Patients with these antibodies were more likely to also show strong T cell activity against the same targets, implying that antibodies help present tumor fragments to the cellular arm of immunity. Large survival datasets from melanoma and lung cancer confirmed that tumors with strong IgG1 signatures tend to respond better to checkpoint drugs, whereas cancers treated mainly with standard chemotherapy showed no such link.
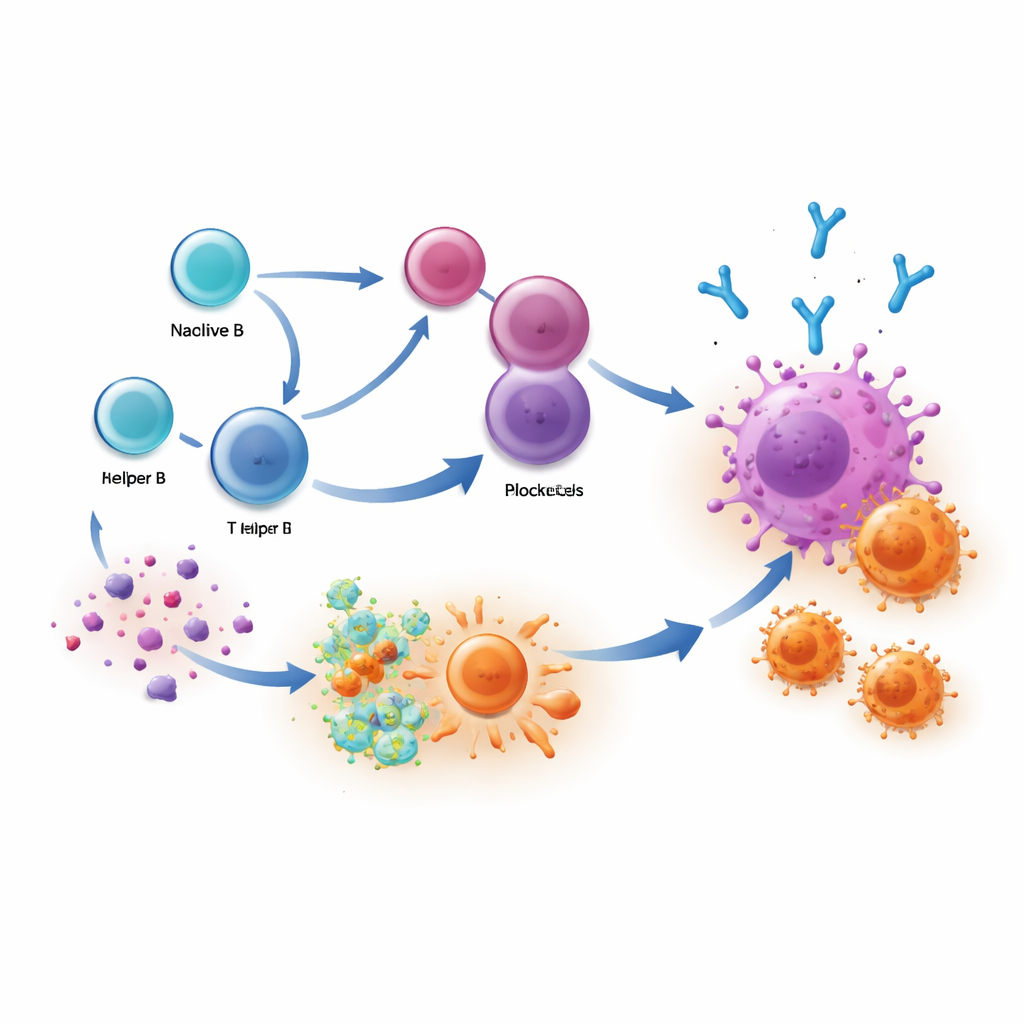
What this means for future treatments
Put simply, patients do best when their immune system mounts a two-pronged attack: IgG1 antibodies from plasma cells tag cancer cells, and T cells move in for the kill. This study shows that checkpoint therapies can boost these antibody responses, especially when helpful B cell clones are present before treatment. Measuring IgG1-rich plasma cells or their antibodies in blood and tumor samples could help doctors identify likely responders and monitor how well treatment is working. In the long run, vaccines or drugs that steer B cells toward making more of these precise, tumor-homing IgG1 antibodies may amplify the benefits of immunotherapy and bring durable responses to more people living with cancer.
Citation: Gonzalez-Kozlova, E., Sweeney, R., Figueiredo, I. et al. Humoral IgG1 responses to tumor antigens underpin clinical outcomes in immune checkpoint blockade. Nat Med 32, 978–991 (2026). https://doi.org/10.1038/s41591-025-04177-6
Keywords: cancer immunotherapy, B cells, IgG1 antibodies, immune checkpoint blockade, hepatocellular carcinoma