Clear Sky Science · en
BCMA-directed mRNA CAR T cell therapy for myasthenia gravis: a randomized, double-blind, placebo-controlled phase 2b trial
Why this study matters to everyday life
Myasthenia gravis is a long-term illness that makes simple actions—like climbing stairs, chewing a meal, or keeping your eyelids open—unexpectedly hard. Many people need years of strong immune-suppressing drugs that can cause serious side effects. This study tested a new kind of one-time cell treatment that aims to quiet the disease for at least a year with just six weekly infusions, potentially reducing the need for constant medication.
A disease of tired muscles
In myasthenia gravis, the body’s own defenses mistakenly attack the “handshake” between nerves and muscles. Antibodies made by certain immune cells block and damage the docking sites that normally let nerve signals trigger muscle movement. Over time, people develop drooping eyelids, slurred speech, trouble swallowing, and weakness in their arms, legs, and breathing muscles. Standard treatments dampen the immune system broadly, which can help, but often leave patients with lingering symptoms and higher risks of infections, weight gain, mood changes, and other drug-related problems.
A new kind of custom cell therapy
The therapy tested here, called Descartes-08, takes a more targeted approach. Doctors first collect a patient’s own T cells—immune cells that can be trained to recognize specific targets—from the blood. In the lab, these cells are temporarily equipped with genetic instructions, carried as mRNA, that teach them to recognize a protein called BCMA on the surface of the antibody-producing cells driving the disease. Unlike traditional cancer cell therapies that use permanent viral changes and require chemotherapy beforehand, this design is short-lived and does not need pre-treatment to wipe out the immune system. Patients receive Descartes-08 as six brief weekly infusions in an outpatient clinic. 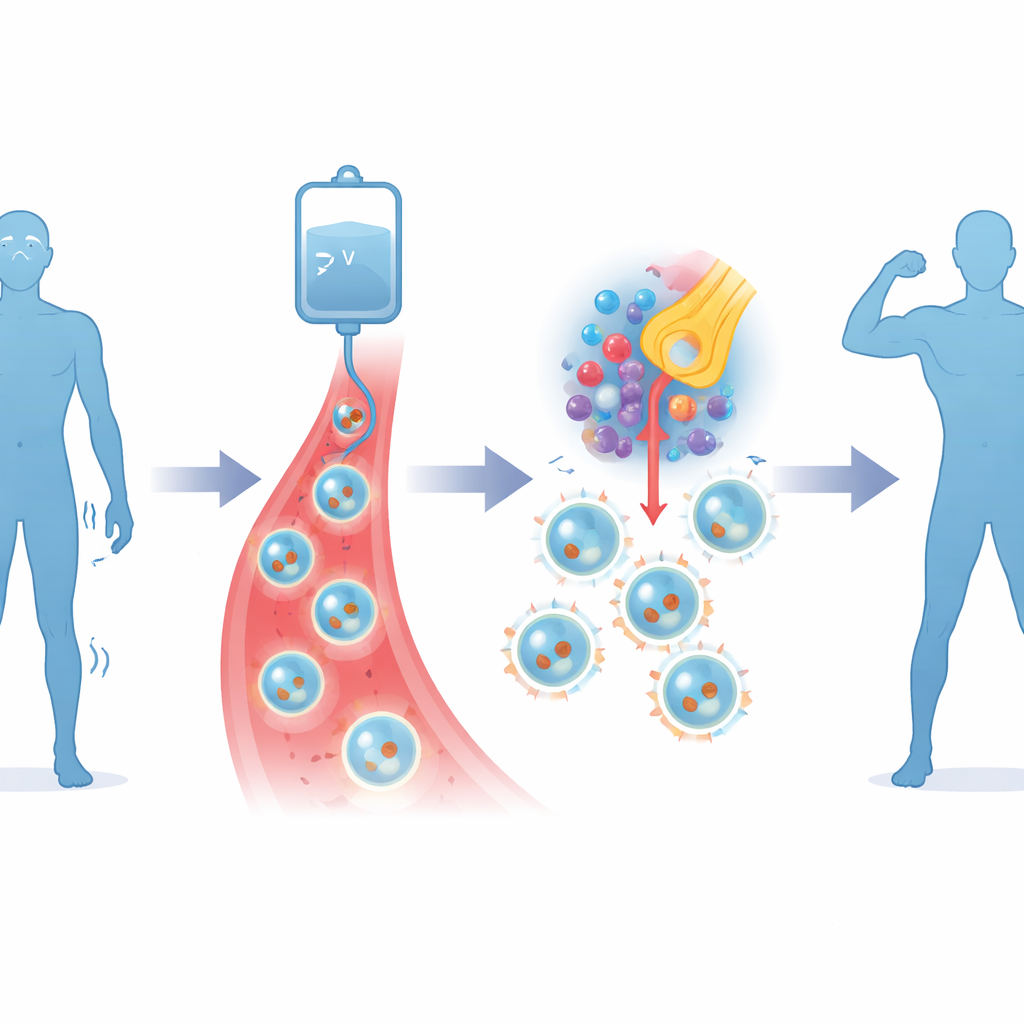
Putting the idea to a fair test
This phase 2b trial enrolled adults with generalized myasthenia gravis whose disease affected more than just their eye muscles and who were already on standard treatments. Participants were randomly assigned to get either Descartes-08 or a look-alike placebo infusion, and neither they nor their doctors knew which they were receiving. The main yardstick was a widely used score that combines patients’ reports of daily function with doctors’ strength testing. The researchers asked: three months after treatment, how many people had improved by at least five points—more than what experts consider a meaningful change? They also tracked how long improvements lasted, whether patients could lower their steroid doses, and what side effects occurred over a full year.
Stronger muscles, fewer symptoms
Among the 26 participants included in the main analysis, those who received Descartes-08 were more than twice as likely to achieve the targeted improvement at three months than those on placebo (about two-thirds versus one-quarter). On average, treated patients showed larger drops in symptom severity scores and better strength testing than the placebo group, and these gains generally deepened over the first four months and were still present at one year. About one in three patients reached “minimum symptom expression,” meaning their day-to-day symptom score was near zero, and this was even more common—over half—in people who had never received newer biologic drugs before. Many patients on Descartes-08 were also able to cut their steroid dose roughly in half after six months, while the placebo group’s doses stayed about the same. 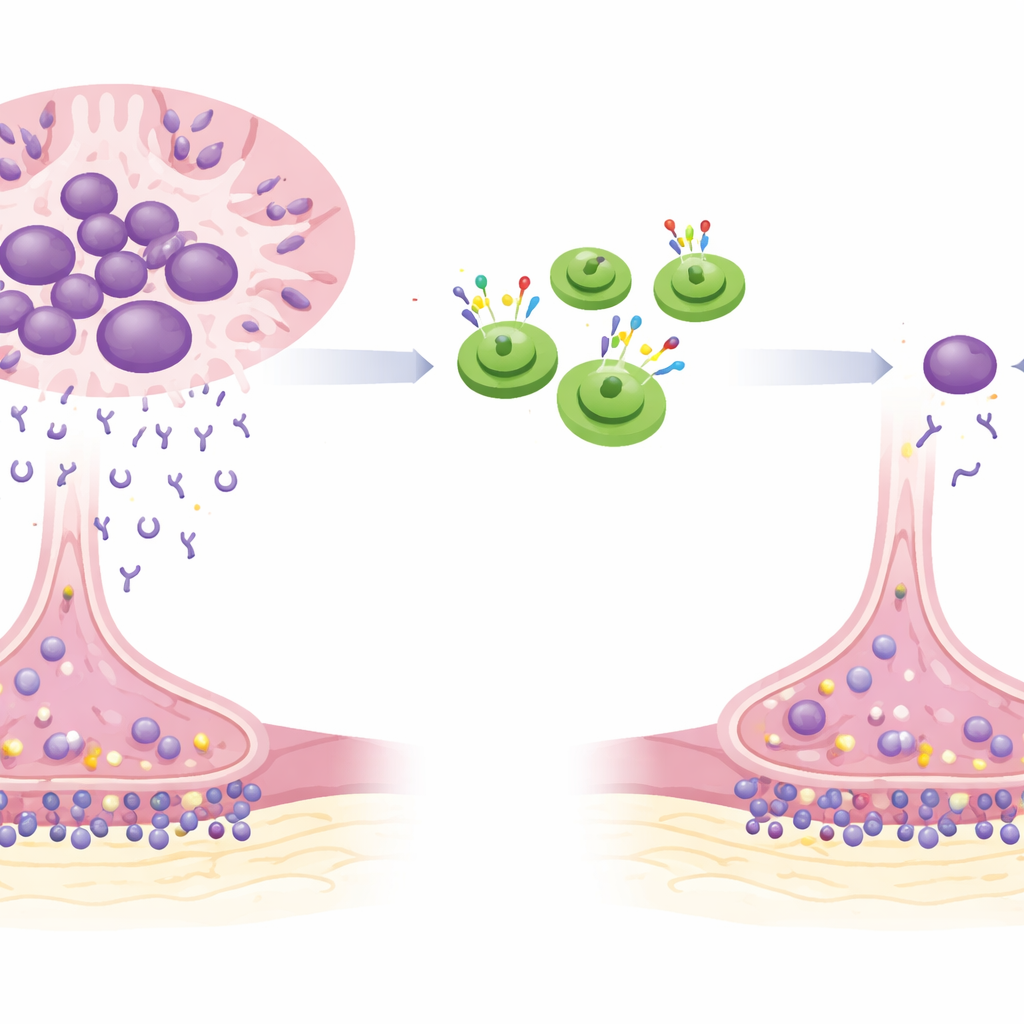
Safety and what happens inside the immune system
Side effects were common but mostly mild to moderate in both groups. The most frequent problems with Descartes-08 were short-lived infusion reactions—fever, chills, headache, and muscle aches—that usually settled within a day without special treatment. Serious complications like life-threatening immune storms or major brain side effects, which can occur with some cancer cell therapies, were not seen. Importantly, blood tests showed that overall antibody levels and protective vaccine responses were preserved, suggesting that the therapy mainly trimmed disease-causing cells instead of broadly wiping out the immune system. Exploratory measurements hinted that certain inflammatory signals tied to autoimmunity fell after treatment, consistent with a more precise “reset” of the misdirected immune response.
What this could mean for patients
For people living with myasthenia gravis, these results suggest that a short course of customized cell infusions might provide meaningful, long-lasting relief while allowing a reduction in everyday immune-suppressing drugs. The study was relatively small, and some differences beyond the main outcome need confirmation in larger trials, but the consistent pattern of improvement and the generally manageable side effects are encouraging. If future studies confirm these findings, mRNA-based cell therapy could become a new option that controls the disease more precisely and conveniently, bringing many patients closer to life with minimal symptoms rather than constant fatigue and medical burdens.
Citation: Vu, T., Durmus, H., Rivner, M. et al. BCMA-directed mRNA CAR T cell therapy for myasthenia gravis: a randomized, double-blind, placebo-controlled phase 2b trial. Nat Med 32, 1131–1141 (2026). https://doi.org/10.1038/s41591-025-04171-y
Keywords: myasthenia gravis, cell therapy, CAR T cells, autoimmune disease, mRNA therapeutics