Clear Sky Science · en
An oral, liver-restricted LXR inverse agonist for dyslipidemia: preclinical development and phase 1 trial
Why this new pill matters for heart and liver health
Atherosclerotic heart disease remains the top killer worldwide, even though many people already take cholesterol-lowering drugs. One big gap is high blood fats called triglycerides and the cholesterol carried in their remnants, which current medicines do not fully control. This paper describes the first human tests of a new kind of oral drug, TLC-2716, designed to act mainly in the liver and gut to lower these risky blood fats while avoiding side effects elsewhere in the body.
The problem of stubborn blood fats
Many adults have dyslipidemia, an unhealthy mix of blood fats that includes high triglycerides, high “bad” cholesterol and low “good” cholesterol. Extra triglycerides and remnant cholesterol build up in the bloodstream, raising the risk of heart attacks, inflamed pancreas and fatty liver disease. Today, lifestyle changes and older drugs such as fibrates help, but often leave a “residual risk” behind. The authors focus on a molecular switch called liver X receptor (LXR), which controls how the body makes, absorbs and clears fats. By sifting through large human genetic databases, they show that higher activity of one LXR gene, NR1H3, is linked to higher triglycerides, altered cholesterol levels and markers of liver disease. This suggests that carefully dialing down LXR activity in the right tissues could improve blood fats and liver health.
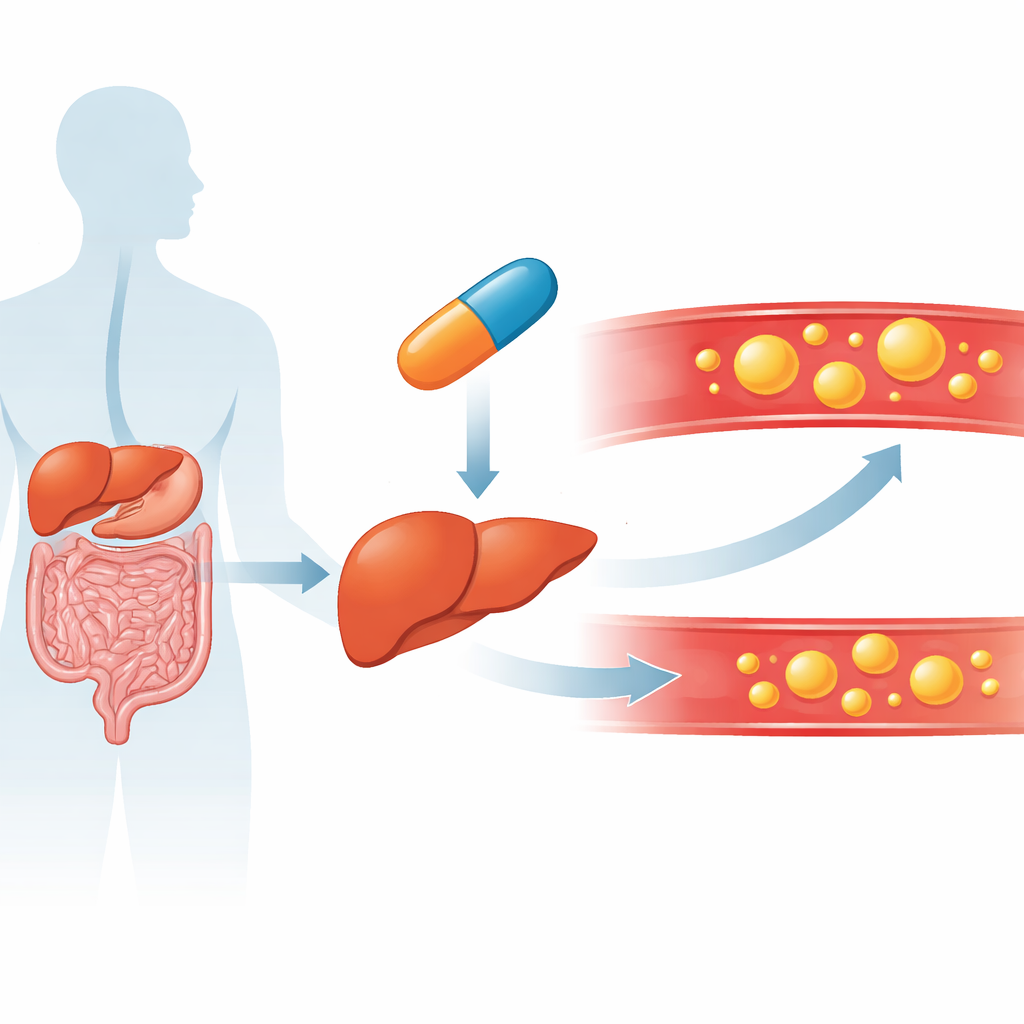
A targeted way to nudge the liver’s fat thermostat
Turning LXR off everywhere in the body would be risky, because LXR also helps move cholesterol out of artery wall cells. To solve this, the team created TLC-2716, an “inverse agonist” that quiets LXR activity but is largely confined to the gut and liver after swallowing. In obese and diabetic rodents fed high-fat diets, TLC-2716 and a closely related compound lowered triglycerides and total cholesterol in the blood and liver without signs of liver injury. The drugs worked through several routes at once: they reduced the liver’s own fat production, decreased fat absorption from the intestine and boosted the breakdown and clearance of triglyceride-rich particles by lowering the levels of natural brakes on fat-clearing enzymes.
From organoids and animal models to human volunteers
Because rodent fat handling differs from humans, the researchers turned to more human-like systems. In mice whose livers were largely replaced with human liver cells, short-term TLC-2716 treatment reduced the activity of key cholesterol- and triglyceride-making genes. Human liver organoids—tiny three-dimensional liver models grown from stem cells—were loaded with fat to mimic steatohepatitis, an advanced form of fatty liver disease. Adding TLC-2716 shrank fat buildup inside these mini-livers and dampened gene programs linked to inflammation and scarring, especially in organoids carrying a genetic variant that normally drives fat production. Together, these experiments suggested that the drug can calm an overactive liver fat program without triggering harmful inflammation.
Testing safety and effect in a first-in-human trial
The team then ran a randomized, placebo-controlled phase 1 trial in 100 healthy adults. After confirming that single doses were safe, they gave daily doses of TLC-2716 or placebo for 14 days. The drug was rapidly taken up from the bloodstream into the liver, and blood tests from both animals and people showed little effect on cells involved in artery-wall cholesterol removal, supporting the idea of liver- and gut-focused action. Across doses, TLC-2716 was well tolerated, with mostly mild side effects such as brief diarrhea or headache and no serious safety signals. Importantly, people taking 6 or 12 milligrams daily showed striking placebo-adjusted drops in fasting triglycerides—around 38% overall, and over 45–60% in those who started with higher levels—as well as large reductions in remnant cholesterol and the number of LDL particles, including the small, dense type thought to be especially harmful.
What this could mean for future treatment
The researchers conclude that selectively turning down LXR activity in the liver and gut with TLC-2716 can substantially improve blood fat profiles in humans over just two weeks, without obvious harm to the liver or to cholesterol-clearing pathways elsewhere in the body. Because the drug is an oral pill that tackles both fat production and clearance, it could one day complement or replace injectable therapies that target single fat-regulating proteins. Larger and longer studies in people with high triglycerides, fatty liver disease and insulin resistance will be needed to confirm whether these early changes in blood markers translate into fewer heart attacks and better liver health, but this work lays the groundwork for a promising new class of cardiometabolic medicines.
Citation: Li, X., Benegiamo, G., Vijayakumar, A. et al. An oral, liver-restricted LXR inverse agonist for dyslipidemia: preclinical development and phase 1 trial. Nat Med 32, 883–893 (2026). https://doi.org/10.1038/s41591-025-04169-6
Keywords: triglycerides, liver-targeted therapy, dyslipidemia, fatty liver disease, cardiometabolic risk