Clear Sky Science · en
Discovery and development of a new oxazolidinone with reduced toxicity for the treatment of tuberculosis
A New Hope Against a Persistent Killer
Tuberculosis (TB) remains one of the world’s deadliest infectious diseases, killing more than a million people every year. Treatments can last many months, and some TB strains have become resistant to multiple drugs, making them much harder to cure. This study describes a new experimental antibiotic, called MK-7762, designed to work powerfully against TB while avoiding some of the serious side effects that limit the use of an older drug, linezolid. If successful in people, MK-7762 could help shorten treatment and extend modern TB therapy to many more patients.
Why Today’s Powerful TB Drug Is Hard to Use
Linezolid, an antibiotic from a class called oxazolidinones, has been a breakthrough for people with highly drug-resistant TB. When combined with other new medicines, it has helped turn once hopeless cases into curable infections and has already changed World Health Organization treatment guidelines. But linezolid can also damage the body’s own cells, especially the bone marrow and nerves, because it interferes with tiny protein factories in human cell “power plants,” the mitochondria. These side effects—such as anemia, nerve pain, and vision problems—mean linezolid is usually reserved for the sickest patients with few other options, rather than being used broadly in people who have strains of TB that are still sensitive to standard drugs.
Designing a Smarter TB Antibiotic
The research team set out to build a drug that keeps the good parts of linezolid—strong activity against TB—while greatly reducing its tendency to harm human cells. They created and tested about 2,800 chemical variations, looking for molecules that killed Mycobacterium tuberculosis efficiently but had much weaker effects on mitochondrial protein production. One promising compound, MK-7762, emerged from this exhaustive search. In laboratory tests, it blocked TB growth more potently than linezolid, yet it was far less likely to interfere with mitochondrial function, suggesting a lower risk of the blood and nerve toxicities that trouble patients on linezolid.
How the Drug Targets TB but Spares Our Cells
To understand why MK-7762 is more selective, the scientists used high-resolution cryogenic electron microscopy to visualize how the drug sits inside the TB bacterium’s protein-making machinery—the ribosome. They found that MK-7762 nestles into a pocket in the mycobacterial ribosome where a specific chemical group on the drug forms a stabilizing bond with one of the bacterial RNA building blocks. In human mitochondria, the matching RNA piece is oriented differently and actually clashes with this part of the drug, making binding far less favorable. This subtle structural difference helps explain how MK-7762 can shut down protein production in TB bacteria while largely sparing the equivalent machinery in human cells.
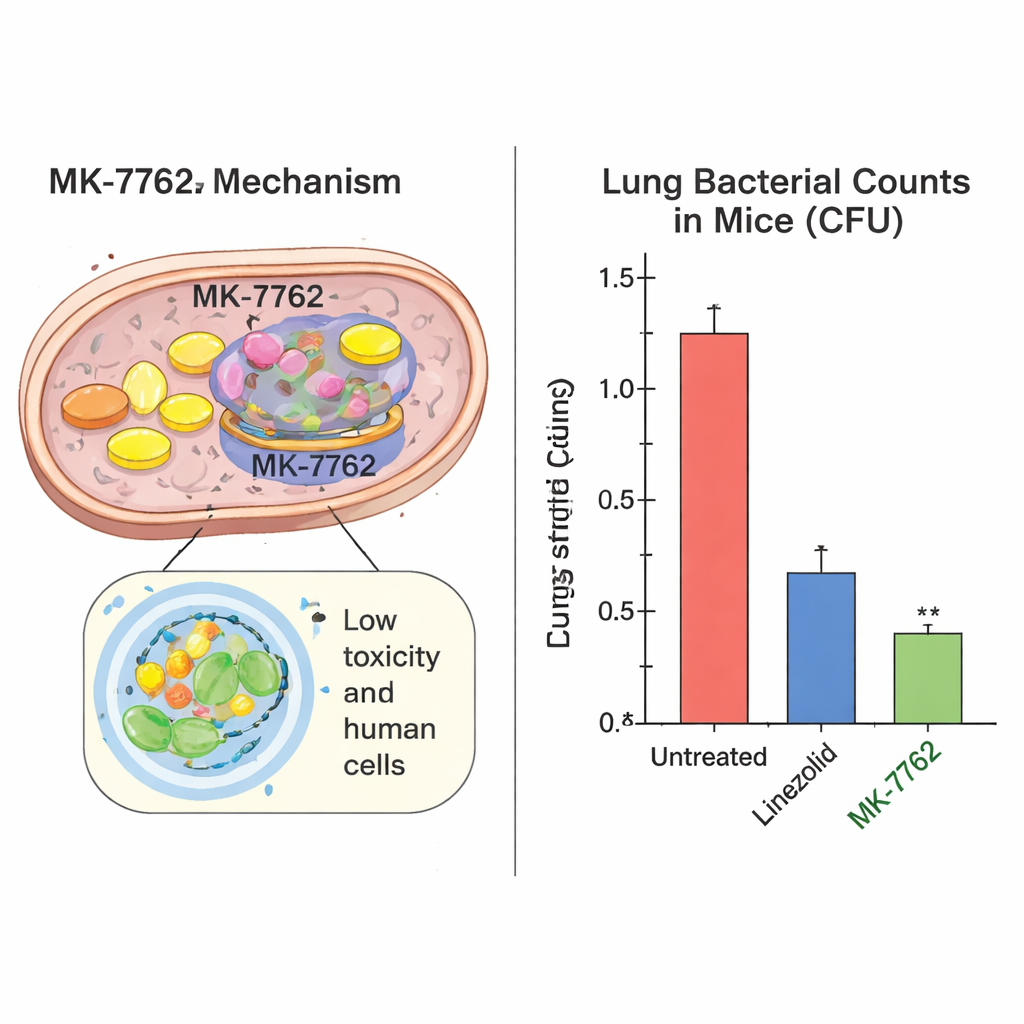
Proof of Power and Safety in Animal Studies
The team then tested MK-7762 in mice infected with TB. In an acute (early) infection model, once-daily dosing of MK-7762 sharply reduced the number of bacteria in the lungs—by up to about 100,000-fold—matching or beating linezolid at similar or lower doses. In a chronic (longer-term) infection model, MK-7762 performed at least as well as linezolid. Crucially, the drug reached the difficult-to-penetrate, cheese-like lesions in the lungs where TB bacteria often hide, achieving concentrations above the level needed to inhibit most clinical TB strains for many hours. In rats and dogs, MK-7762 showed a strong safety margin: animals tolerated exposures many times higher than those expected in humans at a projected 100-milligram once-daily dose, with minimal and reversible changes in blood counts and no clear signs of bone marrow or organ damage over four months of dosing.
What This Could Mean for Future TB Treatment
Taken together, these results suggest that MK-7762 combines the strengths of linezolid with a much gentler safety profile. It remains active against a wide range of drug-sensitive and drug-resistant TB strains, behaves in the body in a way that should allow convenient once-daily dosing, and appears less likely to cause the mitochondrial-related side effects that have held back linezolid. While human trials are still needed to confirm its benefits and uncover any new risks, MK-7762 raises the possibility that a safer oxazolidinone could become part of shorter, all-oral treatment combinations for virtually all forms of TB—not just the most drug-resistant cases.
Citation: Crowley, B.M., Boshoff, H.I., Boving, A. et al. Discovery and development of a new oxazolidinone with reduced toxicity for the treatment of tuberculosis. Nat Med 32, 553–560 (2026). https://doi.org/10.1038/s41591-025-04164-x
Keywords: tuberculosis, antibiotic, drug resistance, linezolid, MK-7762