Clear Sky Science · en
Linavonkibart and pembrolizumab in immune checkpoint blockade-resistant advanced solid tumors: a phase 1 trial
Why stubborn cancers matter to all of us
Immunotherapy drugs that “take the brakes off” the immune system have changed how many cancers are treated, yet most patients either never benefit or eventually see their tumors grow again. This study tests a new antibody medicine, linavonkibart, designed to disable a powerful shield that tumors use to hide from immune attack, and combines it with an existing immunotherapy, pembrolizumab. The results offer an early glimpse that even in patients whose cancers had already stopped responding to checkpoint drugs, it may be possible to restore immune control while keeping side effects in check.
A hidden shield around tumors
Many modern immunotherapy drugs work by blocking so‑called checkpoint molecules, such as PD‑1, that normally restrain T cells. When these brakes are released, immune cells can attack cancer more vigorously. But tumors often adapt by building a hostile neighborhood around themselves that keeps killer T cells out or weakens them once they arrive. A key architect of this protective bubble is a signaling protein called TGFβ1, which is produced by both cancer cells and nearby support cells. Earlier efforts to block the entire TGFβ family showed promise but ran into serious heart and bleeding problems, because other family members are important for healthy tissues. Linavonkibart was engineered to thread this needle by selectively neutralizing only the latent, inactive form of TGFβ1 before it is switched on, while leaving closely related molecules that the body needs untouched.
Designing a careful first‑in‑human test
The DRAGON trial was a multicenter phase 1 study carried out at 22 hospitals in the United States and South Korea. It enrolled 112 adults with advanced solid tumors, all of whom had limited treatment options. In the first part of the study, linavonkibart was given alone at gradually increasing doses to small groups of patients, or combined with their previous PD‑1‑based drug. This stepwise approach was used to map out safety, how the drug moved and persisted in the bloodstream, and to choose a dose for broader testing. In the second part, 78 patients whose cancers had already resisted at least one course of PD‑1 immunotherapy received a fixed dose of linavonkibart together with pembrolizumab. These patients had difficult‑to‑treat cancers, including clear cell kidney cancer, melanoma, head and neck cancer, urothelial (bladder) cancer and lung cancer, and most had undergone several prior treatments.
Safety first: what happened to patients
The main goal of this early‑phase study was safety, and linavonkibart performed encouragingly on that front. Across all dose levels, no maximum tolerated dose was reached, and no dose‑limiting or fatal side effects linked to the drug were seen. When linavonkibart was combined with pembrolizumab at the phase 2 dose, about three‑quarters of patients experienced some treatment‑related side effect, similar to what is often observed with immunotherapy alone. Rashes and itching were the most frequent additional problems, and a small number of patients developed more serious skin or lung inflammation. Importantly, the dangerous immune overreaction called cytokine release syndrome, which can occur with some antibody therapies, was not observed, and the overall safety pattern largely resembled that of pembrolizumab by itself.
Hints that the immune system can be re‑awakened
Although this was not a large, definitive efficacy trial, several signs suggested that shutting down TGFβ1 could help rekindle responses to PD‑1 blockade. In the expansion phase, the linavonkibart–pembrolizumab combination shrank measurable tumors in a notable fraction of patients who had previously stopped benefiting from PD‑1 drugs. Confirmed response rates were 20% in clear cell kidney cancer, 18.2% in melanoma, and just over 9% in head and neck and urothelial cancers, with one kidney‑cancer patient achieving a complete disappearance of visible disease. Many of these responses lasted for many months, and patients who responded generally stayed on the combination far longer than they had on their prior PD‑1 therapy. Biopsies taken before and after treatment offered a biological explanation: tumors showed greater infiltration of CD8 “killer” T cells, higher levels of activated T cells, fewer suppressive regulatory T cells and myeloid cells, and an overall shift toward a more inflamed, attack‑ready microenvironment.
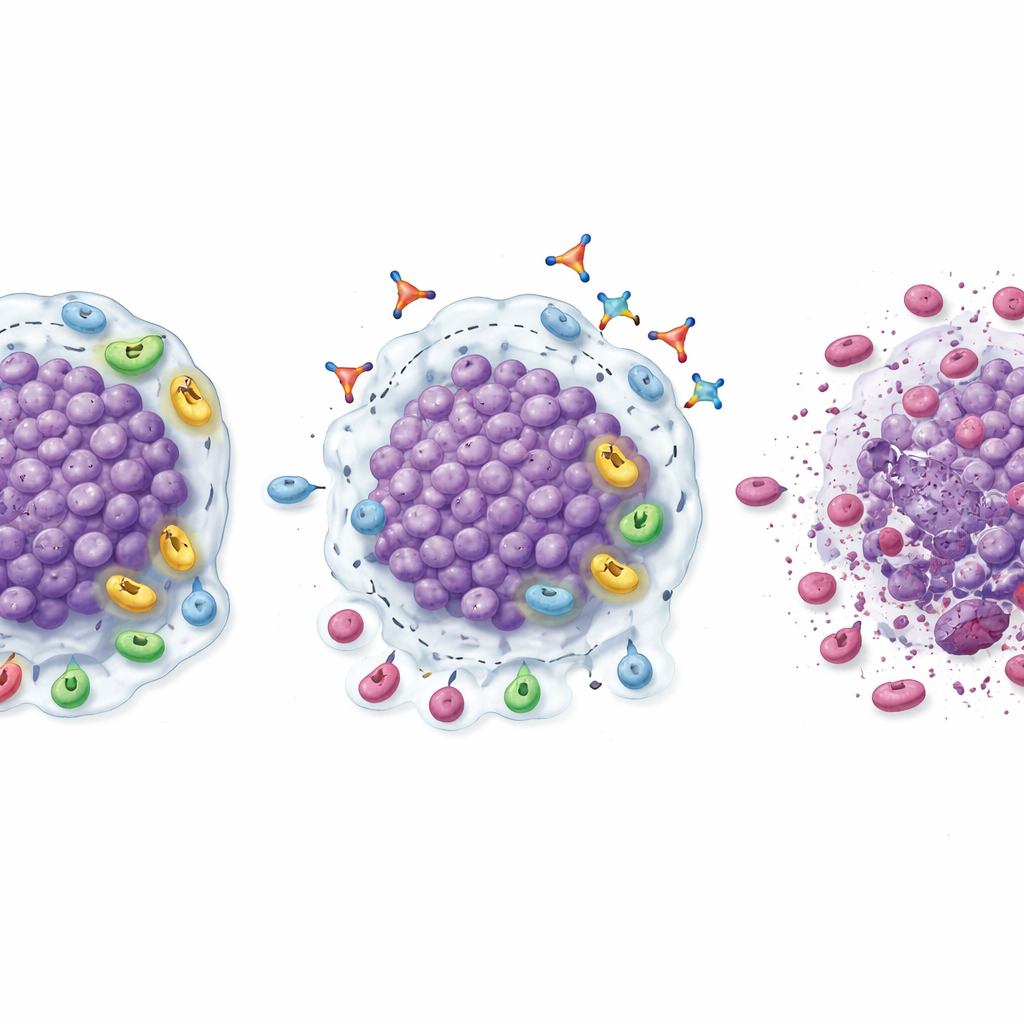
Finding who might benefit the most
The researchers also explored which patients might be especially likely to respond. In clear cell kidney cancer, tumors that already contained many CD8 T cells at baseline—but were also rich in regulatory T cells and expressed high levels of TGFβ1—were more likely to shrink on the combination therapy. In these patients, response rates and progression‑free survival were substantially higher than in the broader kidney‑cancer group. This pattern suggests a “sweet spot”: tumors that have plenty of immune soldiers present but are held in check by TGFβ1‑driven suppression may be ideal candidates for linavonkibart plus PD‑1 blockade. If validated in larger studies, simple tissue tests for these markers could help doctors select patients most likely to benefit.
What this work means going forward
For people facing advanced cancers that have already resisted immunotherapy, the prospect of re‑energizing their own immune system without adding severe toxicity is compelling. This first‑in‑human trial provides early evidence that selectively disabling the TGFβ1 shield with linavonkibart can be done safely and may restore meaningful tumor control when paired with a PD‑1 inhibitor, particularly in certain kidney cancers. While the study was small and not randomized, its safety record, biological readouts and durable responses together build a strong case for larger phase 2 trials. If future studies confirm these findings, this approach could expand the circle of patients who can benefit from immunotherapy, turning some currently stubborn cancers into ones that are once again vulnerable to the body’s own defenses.
Citation: Yap, T.A., Sweis, R.F., Vaishampayan, U. et al. Linavonkibart and pembrolizumab in immune checkpoint blockade-resistant advanced solid tumors: a phase 1 trial. Nat Med 32, 992–1001 (2026). https://doi.org/10.1038/s41591-025-04157-w
Keywords: cancer immunotherapy, TGFβ1 inhibition, immune checkpoint resistance, clear cell renal cell carcinoma, tumor microenvironment