Clear Sky Science · en
Innate antiviral and immune functions associated with the HIV reservoir decay after anti-PD-1 therapy
Why this research matters
Modern HIV drugs can keep the virus under control for decades, but they cannot fully eliminate it. A small "reservoir" of infected cells lies dormant in the body, ready to restart infection if treatment stops. This study explores whether a type of cancer immunotherapy that "releases the brakes" on the immune system can also help shrink this hidden HIV reservoir and points to biological clues that may tell us who will benefit most.
Hidden virus and a new kind of medicine
People living with HIV today often take antiretroviral therapy (ART), which reduces virus in the blood to levels too low to detect. Yet the virus persists in a tiny population of immune cells, mostly memory CD4 T cells, where it lies silent and out of reach of drugs. Many of these cells display a surface protein called PD-1, part of a braking system that normally prevents the immune system from overreacting. Cancer drugs that block PD-1 have transformed treatment for several tumors by reawakening immune cells. Because PD-1 is also found on HIV-harboring cells and on exhausted virus-fighting cells, researchers have wondered whether the same drugs might help loosen HIV’s grip.
A trial in people with both HIV and cancer
In this study, 30 adults living with HIV and various cancers received the PD-1–blocking antibody pembrolizumab every three weeks, on top of their usual ART. The treatment was generally safe, and some participants experienced partial or complete tumor responses. The researchers collected blood before treatment, 24 hours later, one week later, and at the end of therapy, which could be many months away. They measured HIV DNA inside immune cells as a marker of the latent reservoir, HIV RNA as a sign of viral activity, and a rich set of immune signals using RNA sequencing, protein measurements, and single-cell analyses. 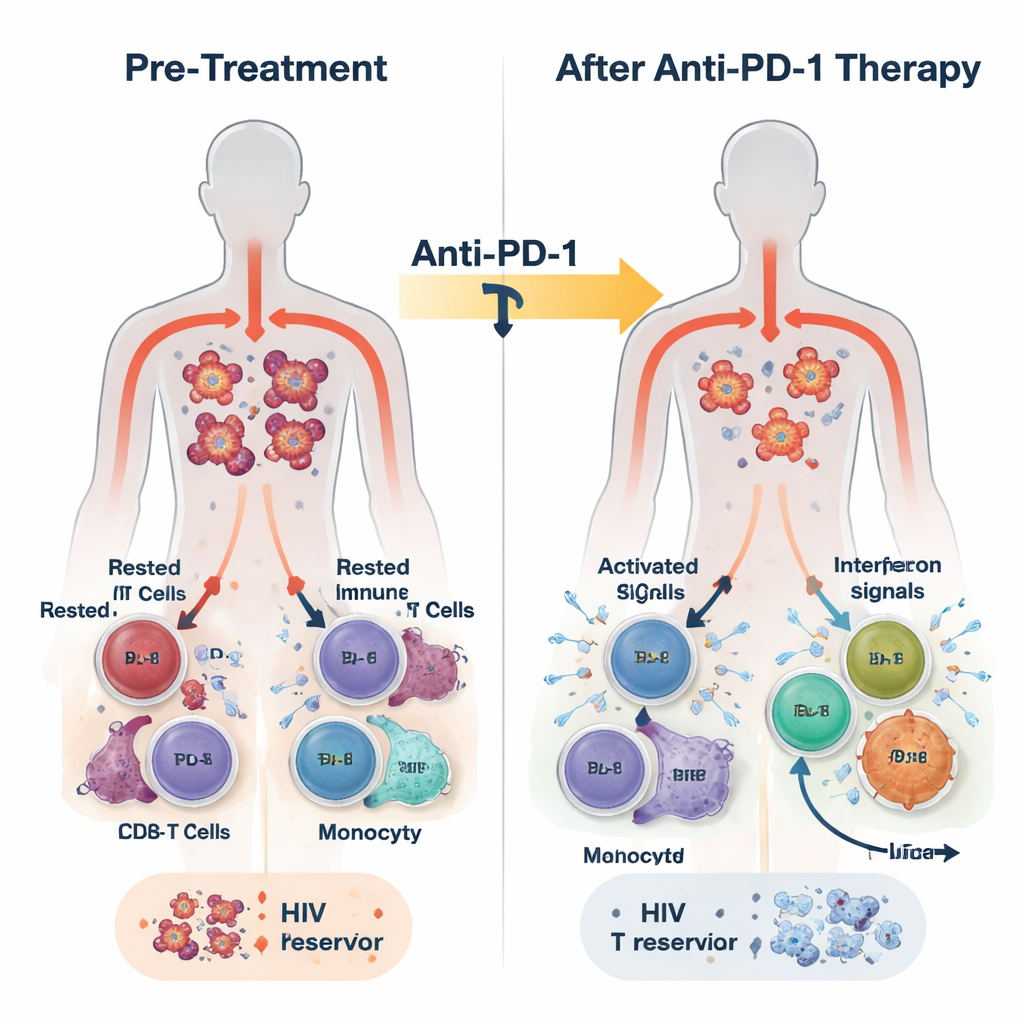
An early antiviral "alarm" in some participants
Within just 24 hours of the first dose, blood from most participants showed a burst of innate immune activity. Levels of inflammatory messengers such as interferons (the body’s classic antiviral alarm molecules) and IL-6 rose, while a suppressive molecule called TGF-beta fell. At the same time, gene programs linked to antiviral defense, monocyte activity, and CD8 T cell effector function were switched on across the blood’s white cells. Closer analysis revealed that this response was not uniform. By the end of treatment, participants could be divided into two groups based on the strength and persistence of interferon-stimulated gene (ISG) activity: an ISG-high group that kept these antiviral programs running, and an ISG-low group in which they faded.
Linking immune patterns to shrinking HIV reservoirs
The ISG-high participants started off with lower amounts of HIV DNA in their CD4 T cells than the ISG-low group and went on to experience a further, roughly twofold drop in this latent reservoir over time. They also tended to show fewer CD4 T cells capable of reawakening HIV in laboratory tests by the end of therapy. In contrast, those in the ISG-low group showed little or no change in HIV DNA and often had more cells that could be induced to produce HIV RNA. High-resolution single-cell studies offered a mechanistic picture: a subset of monocytes rapidly increased antiviral gene expression; CD8 T cells specific for HIV expanded and became more proliferative; and certain CD4 T cell subsets upregulated antiviral defenses while others associated with ongoing viral transcription declined. Experiments in healthy donor cells confirmed that stimulating innate sensors or interferon pathways could both boost antiviral proteins in CD4 T cells and reduce their susceptibility to HIV infection. 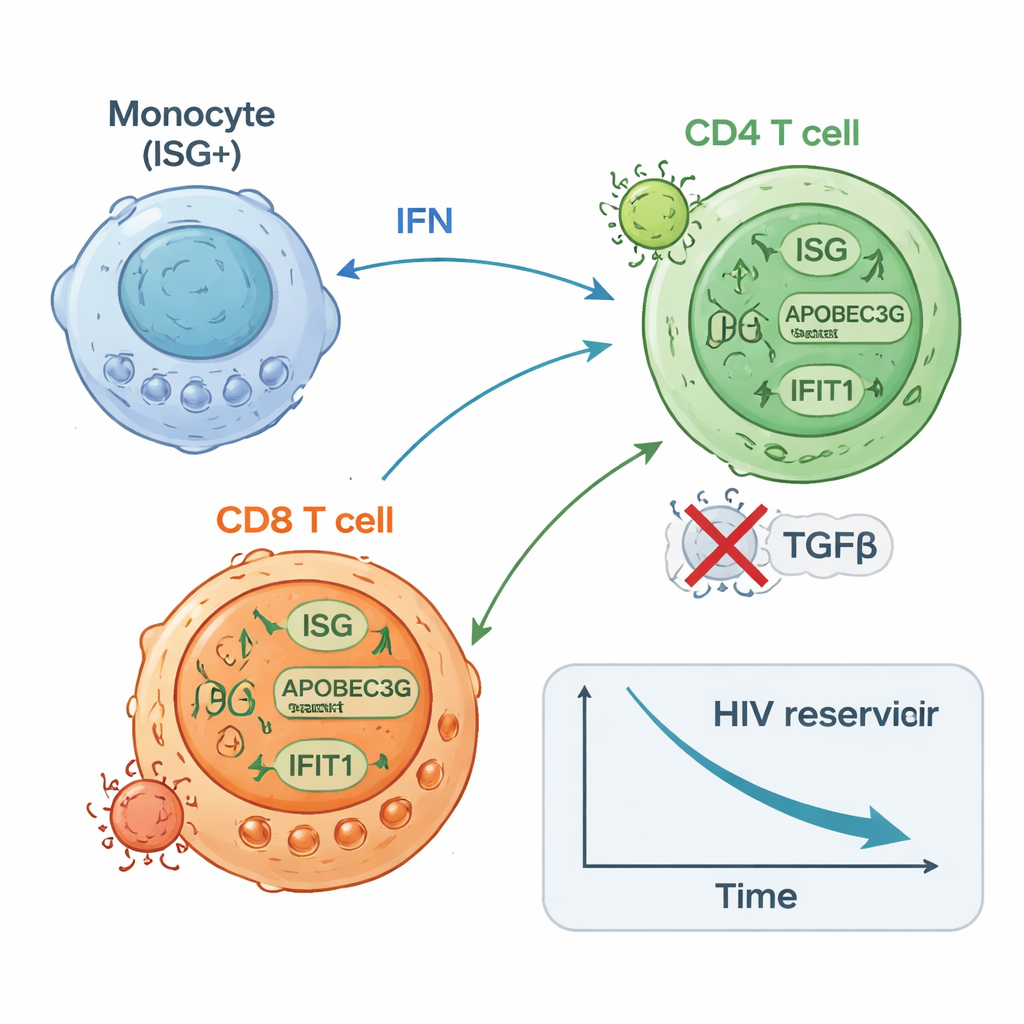
Clues for choosing and improving future therapies
Intriguingly, the same antiviral gene patterns seen in the ISG-high group also appear in blood from some people with other cancers or infections, suggesting that a naturally “primed” immune state may exist in a subset of individuals. In this study, that primed state—defined by strong interferon programs in myeloid cells and T cells and low activity of regulatory pathways like WNT signaling and TGF-beta—was linked to both safer control of the HIV reservoir and to features associated with better cancer outcomes in other trials. The authors propose that combining PD-1 blockade with carefully timed boosts to interferon pathways, or with drugs that block TGF-beta, could further enhance HIV reservoir reduction while still controlling cancer.
What this means for people living with HIV
This work does not yet offer a cure, and PD-1–blocking drugs remain powerful therapies best reserved for serious conditions like cancer. But it provides important proof that briefly lifting immune brakes can trigger a rapid, coordinated antiviral response that measurably shrinks the pool of HIV-infected cells in some individuals on ART. Just as important, it outlines a set of blood-based immune signatures that may allow doctors to identify people most likely to benefit from such approaches and to design combination strategies that push the immune system to expose and clear more of HIV’s last hiding places.
Citation: Talla, A., Azevedo, J.L.L.C., Latif, M.B. et al. Innate antiviral and immune functions associated with the HIV reservoir decay after anti-PD-1 therapy. Nat Med 32, 505–517 (2026). https://doi.org/10.1038/s41591-025-04139-y
Keywords: HIV reservoir, PD-1 blockade, interferon response, cancer immunotherapy, antiretroviral therapy