Clear Sky Science · en
Genotype-stratified adjunctive dexamethasone for tuberculous meningitis in HIV-negative adults: a randomized controlled phase 3 trial
Why this brain infection study matters
Tuberculous meningitis is the most deadly form of tuberculosis, attacking the brain and its protective membranes. Doctors worldwide routinely add a powerful anti-inflammatory drug, dexamethasone, to tuberculosis treatment in the hope of saving more lives. Yet its benefit has been modest and uneven, raising a pressing question: can we use a person’s genes to decide who really needs this drug and who might safely do without it? This large Vietnamese trial set out to test that idea directly.
A dangerous infection of the brain
Tuberculous meningitis begins when tuberculosis bacteria spread from the lungs to the brain. As the immune system reacts, swelling and inflammation build up around the brain and spinal cord, causing headaches, fever, confusion and, without treatment, often death. Standard care combines months of antibiotics to kill the bacteria with steroids to calm damaging inflammation. Earlier studies showed that dexamethasone slightly reduced deaths overall, but not in every group of patients, leaving uncertainty about when it truly helps.
A genetic clue to steroid response
Previous work pointed to a gene called LTA4H, which helps control how strongly the immune system responds during tuberculosis. People inherit different versions, or genotypes, of this gene, labeled CC, CT and TT. Earlier, small studies suggested that patients with the TT type had a very strong inflammatory reaction but gained clear benefit from dexamethasone, while those with CC or CT types might not benefit and, in some cases, might even be harmed. If true, doctors could tailor steroid use based on a rapid genetic test at the bedside.
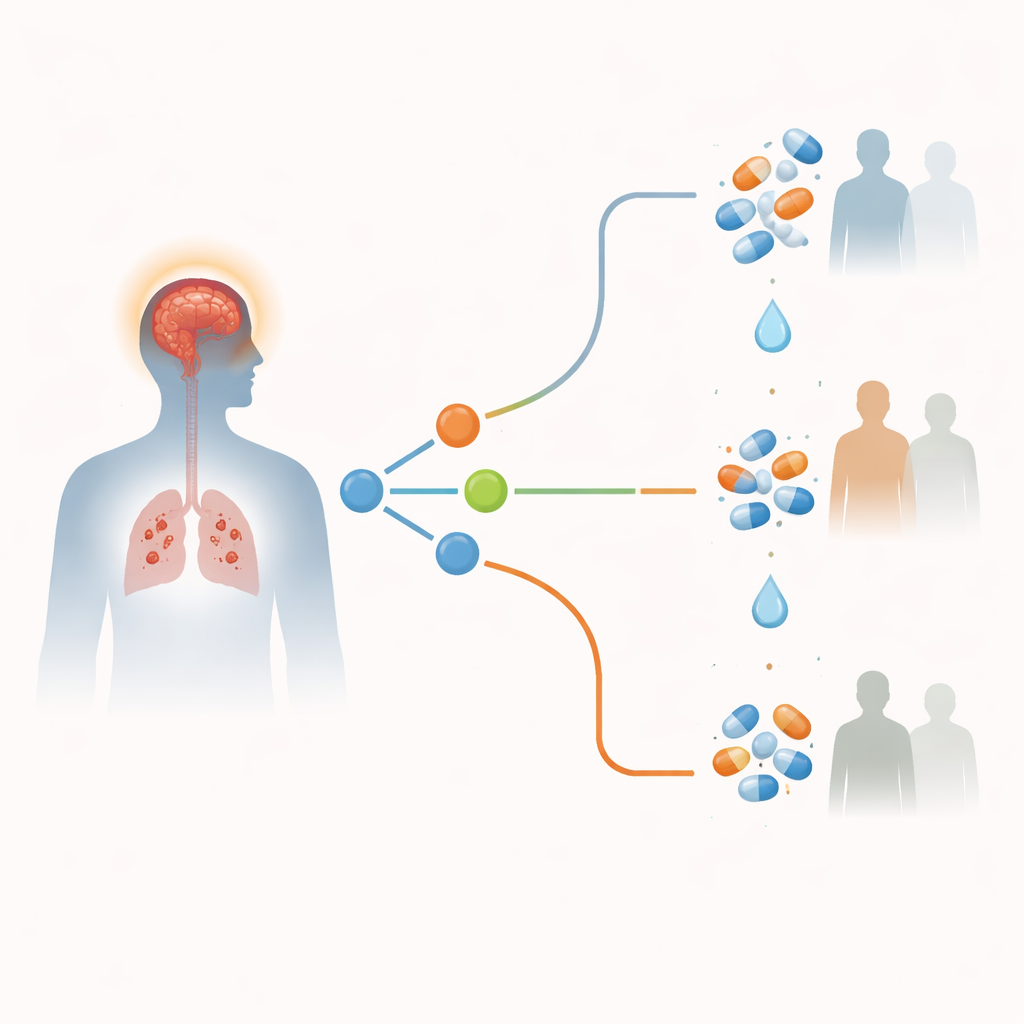
A genotype-guided clinical trial
To put this idea to a rigorous test, researchers conducted a large phase 3 trial in two hospitals in Ho Chi Minh City. They enrolled 720 adults with suspected tuberculous meningitis who did not have HIV. All received standard tuberculosis antibiotics. After a rapid blood test determined their LTA4H genotype, 613 patients with the common CC or CT types were randomly assigned to receive either dexamethasone or a matching placebo for 6–8 weeks. Because earlier data suggested strong benefit, the 89 patients with the rare TT type all received open-label dexamethasone and were not randomized. The main outcome was whether patients died or developed a new serious brain problem, such as a major drop in consciousness or new paralysis, within 12 months.
What the trial found
Among CC and CT patients, outcomes were strikingly similar whether or not they received dexamethasone. About one in three patients in each group died or had a serious new brain event within a year. Statistical checks could not show that skipping dexamethasone was “no worse than” using it, but they also did not show that dexamethasone clearly improved survival or disability in these genotypes. Serious side effects—such as severe infections or bleeding—were about equally common with dexamethasone and placebo, suggesting the drug was generally safe in this setting. TT patients, who all received dexamethasone, had outcomes comparable to the other groups overall, though there were hints they might fare better than others when their disease was very severe.
Looking inside the brain’s immune response
The team also examined spinal fluid and blood samples to see how dexamethasone and LTA4H genotype shaped inflammation. Dexamethasone clearly reduced several inflammatory proteins and immune pathways in the spinal fluid of CC patients, and to a lesser extent in CT patients, over the first month of treatment. In the blood, however, stronger steroid-driven changes were seen mainly in CT patients. Despite these measurable shifts, no simple pattern linked the size of the inflammatory response to who lived or died, underlining how complex the disease is.
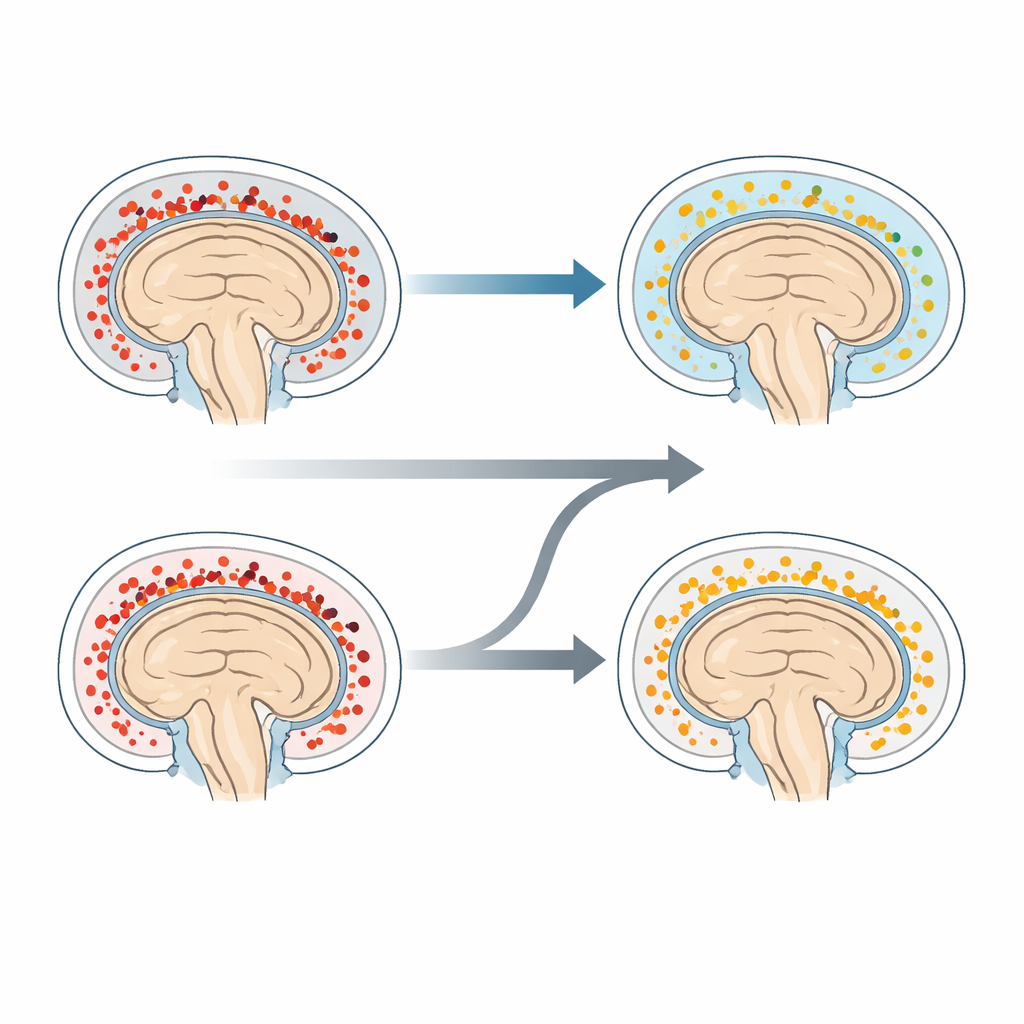
What this means for patients today
When the researchers combined data from this trial with an earlier Vietnamese study of the same steroid regimen, they found that dexamethasone modestly reduced overall deaths in HIV-negative adults with tuberculous meningitis. Taken together, the results support continuing to use dexamethasone for these patients, regardless of their LTA4H genotype. At the same time, the hoped-for simple genetic rule—give steroids only to TT patients and avoid them in others—did not hold up. The work shows that our immune response to brain tuberculosis is governed by many factors, not just a single gene, and that new, more precisely targeted anti-inflammatory treatments are needed. For now, early diagnosis, rapid antibiotics and standard dexamethasone remain the best proven strategy to improve survival in this devastating disease.
Citation: Donovan, J., Duc Bang, N., Dong, H.K.T. et al. Genotype-stratified adjunctive dexamethasone for tuberculous meningitis in HIV-negative adults: a randomized controlled phase 3 trial. Nat Med 32, 849–858 (2026). https://doi.org/10.1038/s41591-025-04138-z
Keywords: tuberculous meningitis, dexamethasone, host genetics, neuroinflammation, clinical trial