Clear Sky Science · en
Anti-inflammatory therapy with low-dose IL-2 in acute coronary syndromes: a randomized phase 2 trial
Why calming artery inflammation matters
Many people who survive a heart attack or serious chest pain remain at high risk of another event, even when they take standard medicines like statins and blood thinners. One major reason is lingering inflammation inside their arteries, which keeps blood vessel walls irritated and unstable. This study asked a simple but important question: can we safely nudge the immune system to calm this inflammation, using very low doses of a natural signaling protein called interleukin‑2 (IL‑2)? If so, it could open a new way to protect the heart beyond traditional cholesterol‑lowering drugs.
A new way to guide the immune system
The researchers focused on a special group of white blood cells called regulatory T cells, or Tregs. These cells act as immune "peacemakers," helping to switch off excessive inflammation and support healing. Earlier studies showed that people with acute coronary syndromes—heart attacks and related conditions—have fewer and less effective Tregs. At high doses, IL‑2 can stimulate aggressive immune cells and is used in cancer therapy, but at very low doses it mainly boosts Tregs. The team designed the IVORY trial to test whether low‑dose IL‑2, given after a heart event in addition to usual care, could safely increase Tregs and reduce inflammation in the arteries.
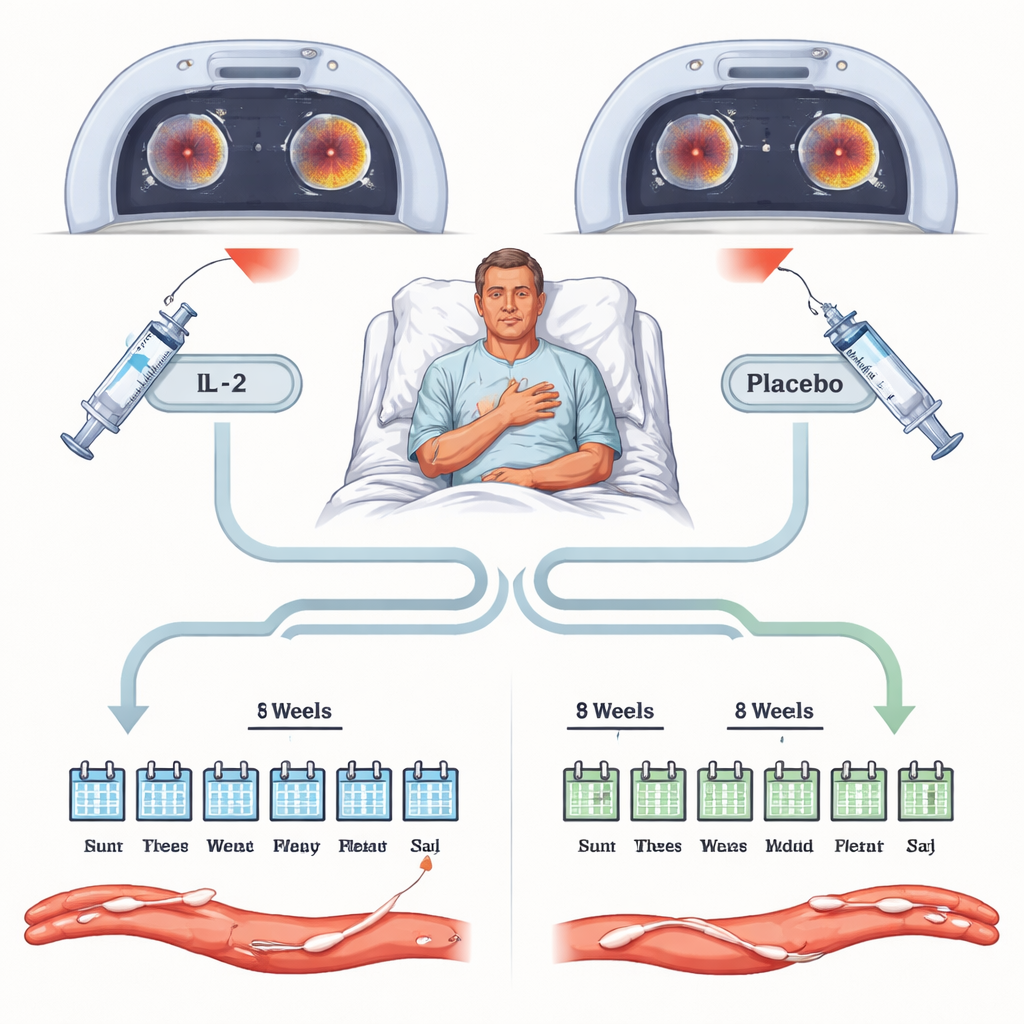
How the trial was run
The study enrolled adults who had recently been hospitalized with an acute coronary syndrome and still had signs of body‑wide inflammation, measured by a blood test called high‑sensitivity C‑reactive protein. Sixty‑three patients were randomly assigned, in double‑blind fashion, to receive either low‑dose IL‑2 or a placebo injection for eight weeks, on top of standard treatments such as statins and antiplatelet drugs. Everyone underwent specialized PET–CT scans of the main chest artery and neck arteries before treatment and again at the end of the eight weeks. These scans measure how much a radioactive sugar is taken up by the artery wall, providing a sensitive readout of inflammation. The main outcome was how much this arterial inflammation differed between the IL‑2 and placebo groups at the end of treatment.
What changed inside the arteries
By the end of the trial, both groups showed some reduction in arterial inflammation, which is expected as patients recover from a heart event and receive intensive standard therapy. However, the drop was significantly greater in those who received low‑dose IL‑2. In the key artery chosen for each patient, inflammation was about 7–8% lower in the IL‑2 group than in the placebo group. When the researchers zoomed in on the most inflamed parts of the vessels, the difference grew to around 9%. These reductions are similar in size to those seen in studies where stronger cholesterol‑lowering drugs have translated into fewer future heart problems, suggesting the change is clinically meaningful.
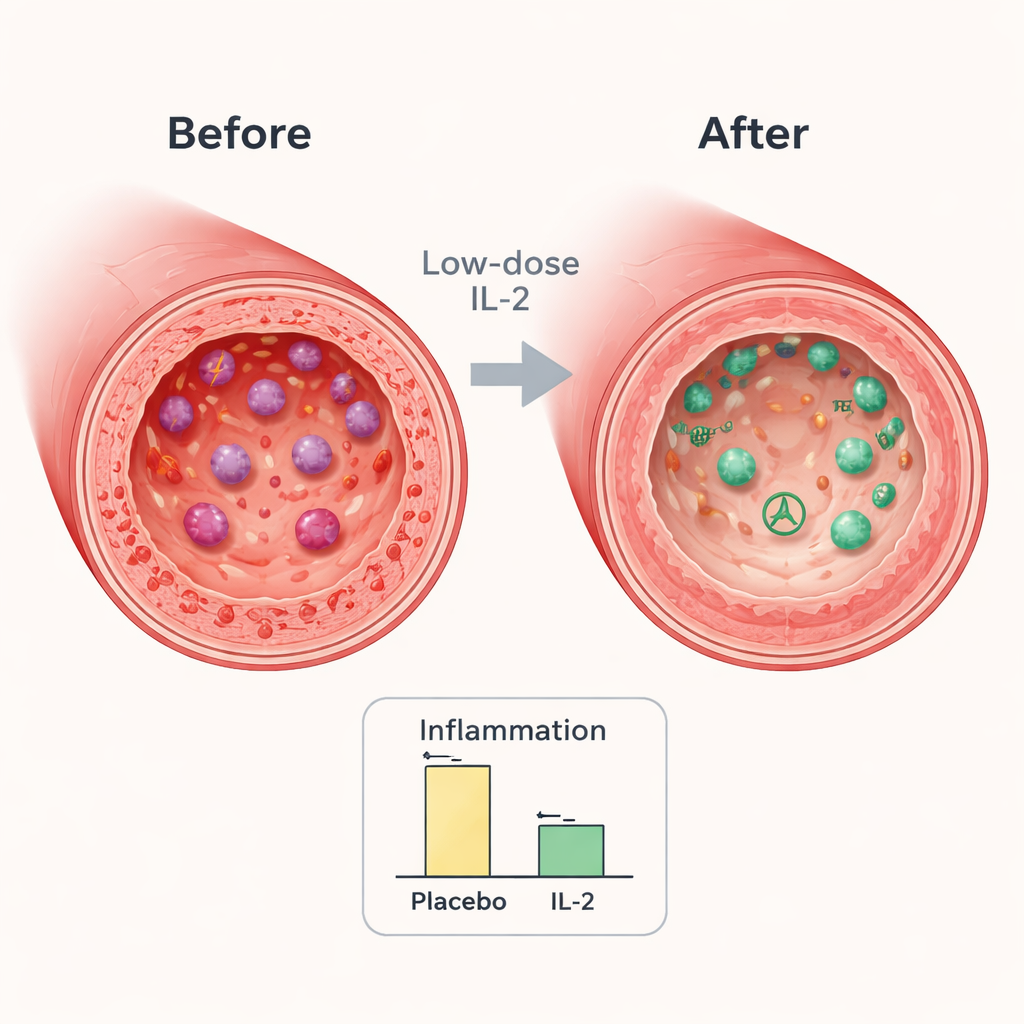
How the immune system responded
Blood tests during the trial revealed that IL‑2 was doing exactly what the scientists had hoped. Regulatory T cell levels rose quickly and stayed 30–50% higher than in the placebo group throughout treatment. At the same time, some cell types linked to more aggressive immune responses, including certain helper T cells and killer T cells, tended to decrease or rise less. Overall, the immune profile shifted toward a calmer, more regulating state. Interestingly, a general inflammation marker in the blood, C‑reactive protein, did not fall with IL‑2, suggesting the drug acts through a more targeted pathway that may not show up in broad blood tests but is visible inside the artery wall.
Safety and early hints about outcomes
Safety was a central concern, because dampening parts of the immune system can sometimes increase infections or other complications. In this trial, low‑dose IL‑2 was well tolerated. The most common side effect was mild redness and itching at the injection site, which cleared within about two days. Rates of infection and other unwanted effects were similar between the IL‑2 and placebo groups, and no treatment‑related serious problems emerged. In a follow‑up study tracking patients for two years, three people in the placebo group experienced major heart‑related events such as heart attack, stroke or cardiovascular death, while none in the IL‑2 group did, although these numbers are too small to draw firm conclusions.
What this could mean for patients
For non‑specialists, the main message is that this study offers an early but encouraging proof of concept: by gently boosting the body’s own "brake" cells in the immune system with tiny doses of IL‑2, doctors may be able to lessen harmful inflammation in the arteries after a heart attack, without adding major side effects. The trial is too small to prove that this approach prevents future heart attacks or strokes, but the size of the anti‑inflammatory effect and the favorable safety profile make larger outcome trials worthwhile. If confirmed, low‑dose IL‑2 or similar immune‑tuning strategies could become a new tool alongside statins and blood thinners to keep hearts safer after a serious cardiac event.
Citation: Sriranjan-Rothwell, R.S., Zhao, T.X., Hoole, S.P. et al. Anti-inflammatory therapy with low-dose IL-2 in acute coronary syndromes: a randomized phase 2 trial. Nat Med 32, 624–632 (2026). https://doi.org/10.1038/s41591-025-04090-y
Keywords: acute coronary syndrome, arterial inflammation, regulatory T cells, interleukin-2 therapy, cardiovascular immunology