Clear Sky Science · en
Repotrectinib in NTRK fusion–positive advanced solid tumors: a phase 1/2 trial
New hope from a targeted cancer pill
Cancer treatments are increasingly designed like precision tools rather than blunt instruments. This study looks at repotrectinib, a targeted pill for people whose tumors carry rare genetic changes called NTRK fusions. Although these fusions occur in fewer than 1 in 100 solid tumors, they can appear in many organs—from lung to thyroid to soft tissue—and often respond dramatically to specially designed drugs. The catch has been that earlier drugs eventually stop working. This trial asks a simple question with big implications: can a smarter, next‑generation drug keep cancers under control for longer, even after other targeted therapies have failed?
A rare but powerful cancer switch
Some cancers are driven by fusions in genes called NTRK1, NTRK2 and NTRK3. These fusions act like jammed‑on growth switches in tumor cells. First‑generation medicines that block the resulting TRK proteins have already helped many patients, but over time the cancer often learns to evade them by acquiring new mutations in the very pocket where the drugs bind. Repotrectinib was built to be smaller and more compact, so it can still fit into this pocket even after such changes. The international TRIDENT‑1 trial tested this drug in adults with advanced solid tumors carrying NTRK fusions, including people who had never received a TRK‑blocking drug and those whose cancer had already outgrown one or two earlier TRK inhibitors.
What the trial did
The study enrolled 144 patients with NTRK fusion–positive solid tumors at centers around the world; 120 of them formed the main group for measuring effectiveness. Their cancers spanned at least 18 different types, most commonly non‑small cell lung cancer, thyroid cancer, salivary gland tumors and soft tissue sarcomas. All had disease that was locally advanced or metastatic, and many had already tried multiple other treatments, including chemotherapy, immunotherapy or earlier TRK inhibitors. Patients took repotrectinib by mouth every day using a dosing schedule worked out in an earlier phase of the trial. Researchers tracked how many patients saw their tumors shrink, how long those responses lasted, how long patients lived without their disease worsening and how well they tolerated the medicine.
Strong and lasting responses
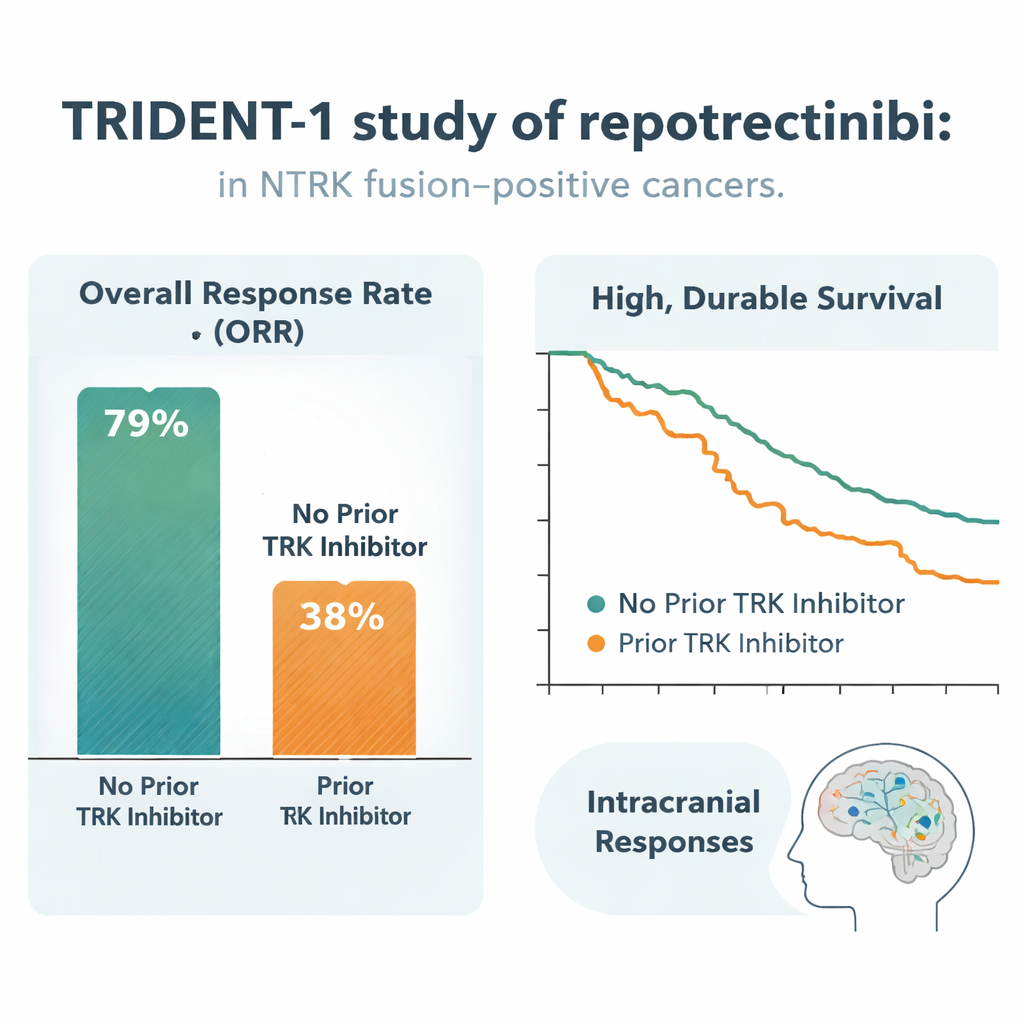
Among 51 patients who had never received a TRK inhibitor, 59% had their tumors shrink enough to count as an objective response, and 16% saw their tumors disappear on scans. Responses tended to appear quickly—within about two months—and were remarkably long‑lasting: after two years, an estimated 85% of responders were still benefiting, and about 60% of the whole group had not yet seen their disease get worse. Even in the more heavily treated group of 69 patients whose cancers had already pushed past another TRK drug, 48% responded to repotrectinib, with a typical response lasting about 10 months and roughly a quarter of patients still free of progression at one year. Importantly, benefit was seen across many tumor types and regardless of which NTRK gene or fusion partner was involved.
Reaching resistant and brain disease
A key challenge for TRK drugs is so‑called solvent front mutations, specific changes in the drug‑binding pocket that can block earlier medicines. In this trial, nearly half of previously treated patients carried such mutations at the start. Even in this hard‑to‑treat subgroup, 53% had meaningful tumor shrinkage with repotrectinib, confirming that the drug can overcome at least some forms of resistance. The brain, a frequent hiding place for cancer, was another focus. Among patients with measurable brain metastases at baseline, two of three in the untreated group and four of six in the previously treated group had tumors shrink in the brain, often for many months. For patients who started without brain involvement, the vast majority remained free of new brain lesions during follow‑up.
Side effects and what it means for patients
Across more than 500 people treated with repotrectinib in this and related groups, the most common side effects were dizziness, altered taste and tingling sensations, most of them mild to moderate. More serious treatment‑related problems, such as anemia or high muscle‑enzyme levels, were less frequent, and only about 4% of patients stopped the drug because of side effects. Measures of overall quality of life stayed stable or improved for most participants over many treatment cycles. Taken together, the results show that repotrectinib can provide durable control of NTRK fusion–positive cancers—including those that have already resisted earlier TRK drugs and those that have spread to the brain—while generally remaining tolerable. For patients with these rare but aggressive tumors, the study supports repotrectinib as an important new option in the expanding toolkit of precision cancer medicine.
Citation: Besse, B., Lin, J.J., Bazhenova, L. et al. Repotrectinib in NTRK fusion–positive advanced solid tumors: a phase 1/2 trial. Nat Med 32, 682–689 (2026). https://doi.org/10.1038/s41591-025-04079-7
Keywords: targeted cancer therapy, NTRK fusion, repotrectinib, TRK inhibitor resistance, brain metastases