Clear Sky Science · en
External trigeminal nerve stimulation in youth with ADHD: a randomized, sham-controlled, phase 2b trial
Why this matters for families
Many children and teens with attention-deficit/hyperactivity disorder (ADHD) struggle with focus, restlessness and impulsive behavior that can disrupt school, friendships and family life. Medicines often help, but they can cause side effects and may not work for everyone. This study tested a promising gadget-based treatment that gently stimulates a facial nerve during sleep, hoping to offer a drug‑free way to ease ADHD symptoms. The findings, however, show a cautionary tale about new brain gadgets that sound too good to be true.
A nighttime device that targets the forehead
External trigeminal nerve stimulation, or TNS, uses sticky electrodes placed on the forehead at bedtime. A battery-powered device sends tiny electrical pulses to branches of the trigeminal nerve, which carries signals into deep brain areas involved in alertness, attention and emotion. Earlier, a small U.S. study in 62 children suggested that four weeks of nightly TNS could noticeably reduce ADHD symptoms, leading to U.S. Food and Drug Administration clearance of a commercial device. Because that earlier trial was small and short, the authors of the new study set out to test TNS far more rigorously in a larger and more diverse group of young people.
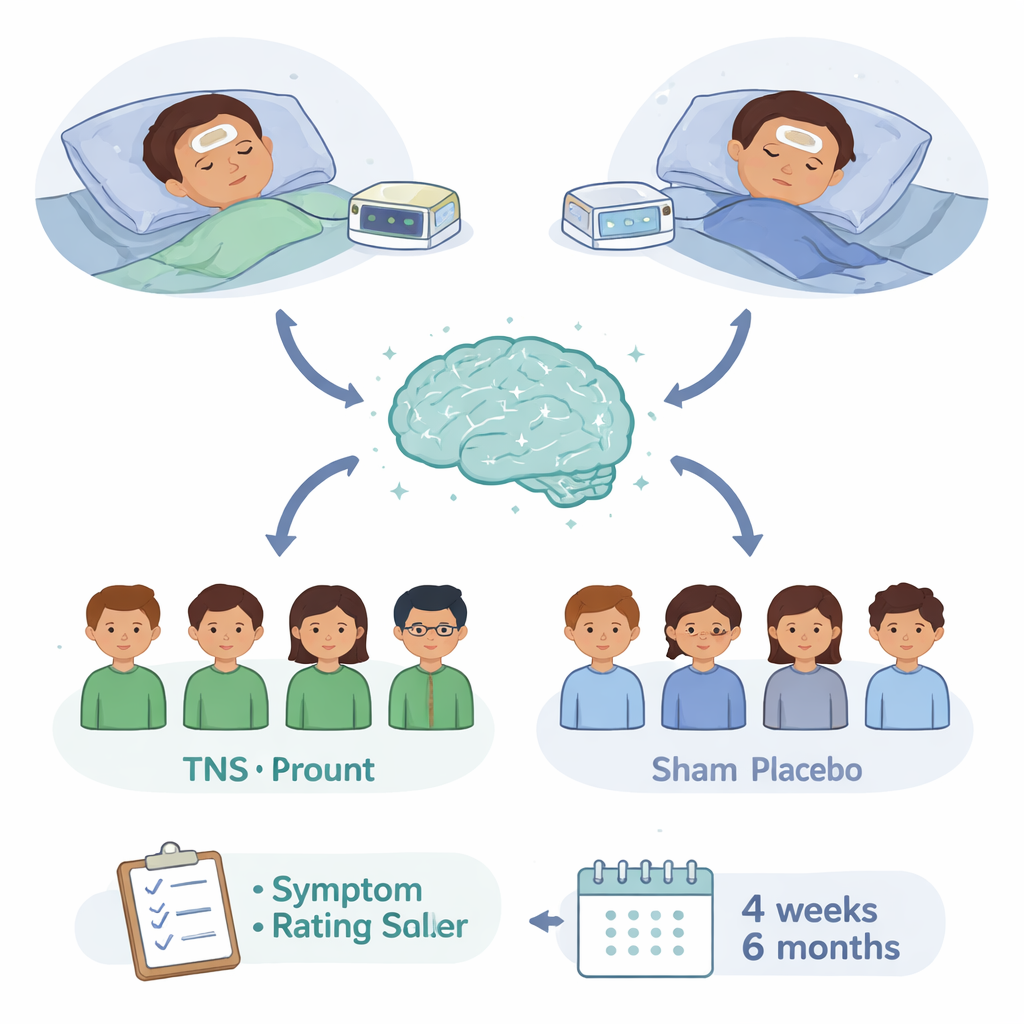
How the trial was run
The researchers enrolled 150 children and adolescents aged 8 to 18 with severe ADHD, recruited from clinics and community referrals in the United Kingdom. Half were randomly assigned to receive real TNS every night for about nine hours over four weeks; the other half used a sham (placebo) device that looked and behaved the same but delivered only brief, very low-intensity pulses each hour. Neither families nor most of the research team knew who was in which group. Parents rated ADHD symptoms weekly, and the team also measured sleep, mood, anxiety, mind-wandering, attention on a computer task, movement with a wrist‑worn sensor and side effects. A subset of measures was repeated six months later to see whether any benefits lasted.
What the researchers found
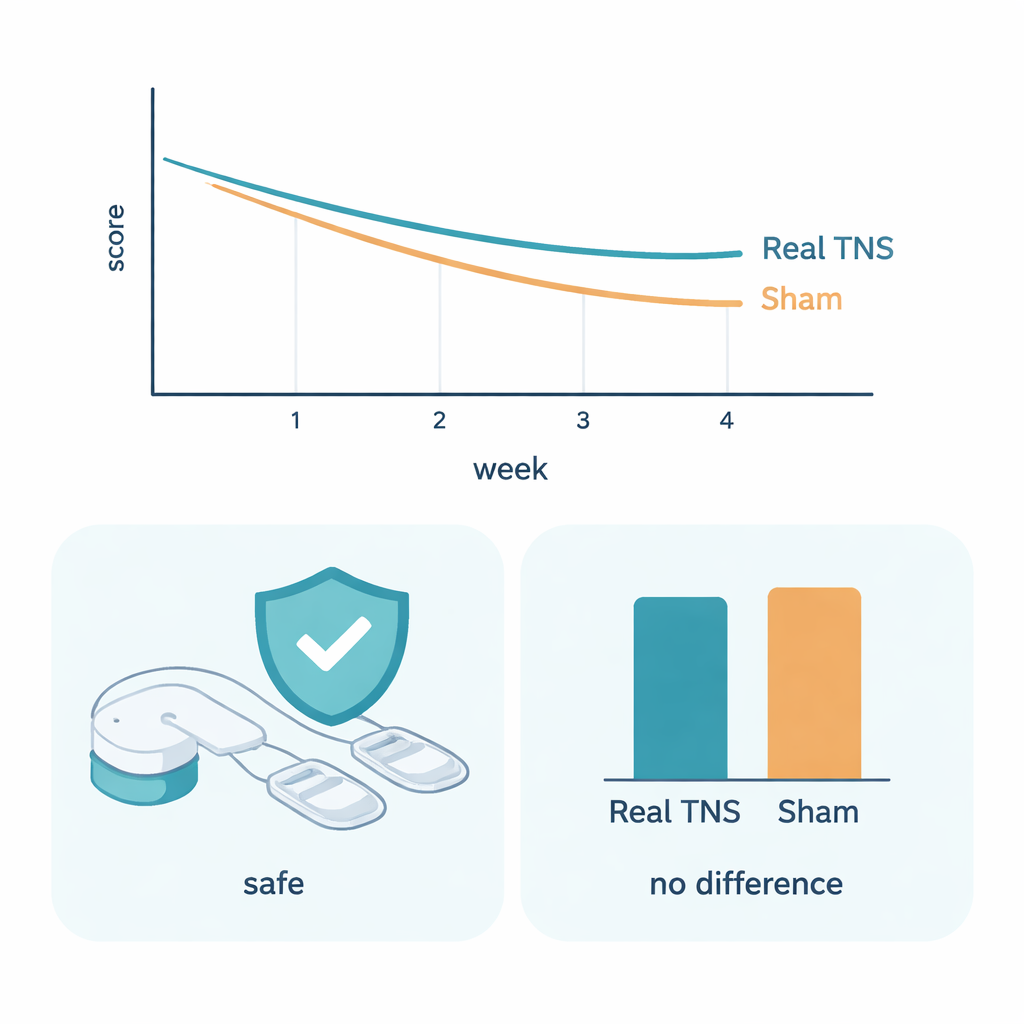
By the end of four weeks, ADHD symptoms had improved substantially in both groups—about a quarter reduction from baseline—but there was no meaningful difference between real and sham TNS on the main parent rating scale. The same pattern held at the six‑month follow‑up: symptoms remained somewhat better than at the start, but real TNS did not outperform the placebo. Across a wide range of other measures, including attention performance, hyperactivity captured by the wrist device, sleep problems, anxiety and depression symptoms, the two groups again looked very similar. The one small exception was a questionnaire asking children about how much their minds wandered, where scores briefly favored the real TNS group at four weeks—but this effect was modest, did not hold up in older teens alone and may have occurred by chance among many tests.
Safety, comfort and the power of expectations
On the positive side, the device proved to be very safe and generally easy to live with. No serious side effects occurred, and common complaints—such as trouble falling asleep, headaches or feeling drowsy—were usually mild and occurred at similar rates in both groups. Families reported high satisfaction with the treatment routine, and almost all participants used the device regularly. Yet the strong improvements in both real and sham groups hint at another powerful force: expectation. Because modern brain devices can seem especially high‑tech and hopeful, simply taking part in such a trial, being cared for by a research team and believing one is receiving active stimulation can, by itself, lead to noticeable symptom relief.
What this means for future ADHD care
This large, carefully controlled trial shows that, despite being safe and acceptable, external trigeminal nerve stimulation does not provide real clinical benefit for children and adolescents with ADHD beyond what a well‑designed placebo can achieve. For families, this means that current TNS devices should not be viewed as effective stand‑alone treatments for ADHD, at least based on the evidence so far. More broadly, the study highlights how crucial rigorous sham comparisons are when evaluating new brain‑stimulation gadgets. Before embracing device‑based therapies that promise drug‑free help, it is essential to test whether they truly change symptoms—or mainly harness the hopes of patients and parents.
Citation: Conti, A.A., Bozhilova, N., Eraydin, I.E. et al. External trigeminal nerve stimulation in youth with ADHD: a randomized, sham-controlled, phase 2b trial. Nat Med 32, 582–590 (2026). https://doi.org/10.1038/s41591-025-04075-x
Keywords: ADHD treatment, brain stimulation, trigeminal nerve stimulation, placebo effect, child mental health