Clear Sky Science · en
Soluble MAdCAM-1 as a biomarker in metastatic renal cell carcinoma
Why Your Gut Could Matter in Kidney Cancer
Immunotherapy has transformed the treatment of advanced kidney cancer, yet many patients eventually stop responding. This study explores a surprising link between the bacteria living in our intestines, a molecule that helps immune cells travel from the gut, and how long patients with metastatic kidney cancer live. The researchers suggest that a simple blood test for a soluble form of this molecule, called MAdCAM-1, could help flag patients whose gut ecosystem is working against their cancer treatment—and who might benefit most from microbiome-focused therapies.
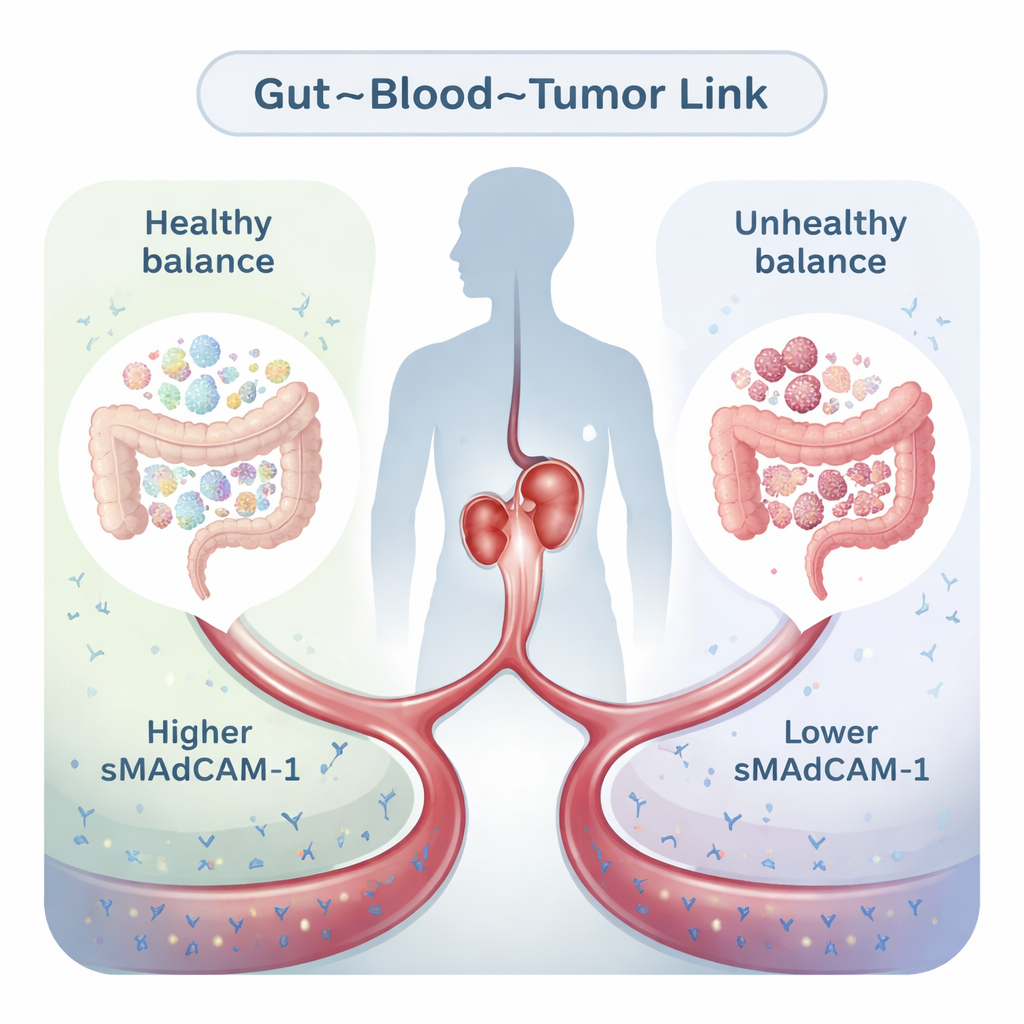
A Traffic Sign Between the Gut and Tumors
Our intestines constantly send immune cells back and forth between the gut and the rest of the body. MAdCAM-1 acts a bit like a road sign on blood vessels in the gut, guiding certain immune cells that recognize signals from intestinal bacteria. A small portion of this protein circulates in the bloodstream as soluble MAdCAM-1 (sMAdCAM-1). Previous work showed that when the gut microbiome is disturbed—by antibiotics, chronic illness or cancer itself—some bacterial groups overgrow, MAdCAM-1 levels drop and immunosuppressive T cells are more likely to leave the gut and settle inside tumors, blunting the effect of immunotherapy. This raised the question: could the amount of sMAdCAM-1 in a blood sample act as a window into both gut health and cancer prognosis?
Following More Than a Thousand Patients
The team analyzed blood samples from 1,051 people with metastatic renal cell carcinoma enrolled in three clinical trials. Patients were treated with either immune checkpoint inhibitors (drugs that release the brakes on immune cells), targeted drugs that block blood vessel growth in tumors (tyrosine kinase inhibitors), or combinations of the two. The researchers compared their sMAdCAM-1 levels to those in healthy volunteers and tracked how long the patients lived without their cancer getting worse, and how long they survived overall. They determined that a blood level of 180 nanograms per milliliter was a key threshold, and used it to group patients into “low” and “high” sMAdCAM-1 categories.
What Low Levels Reveal About Outcomes
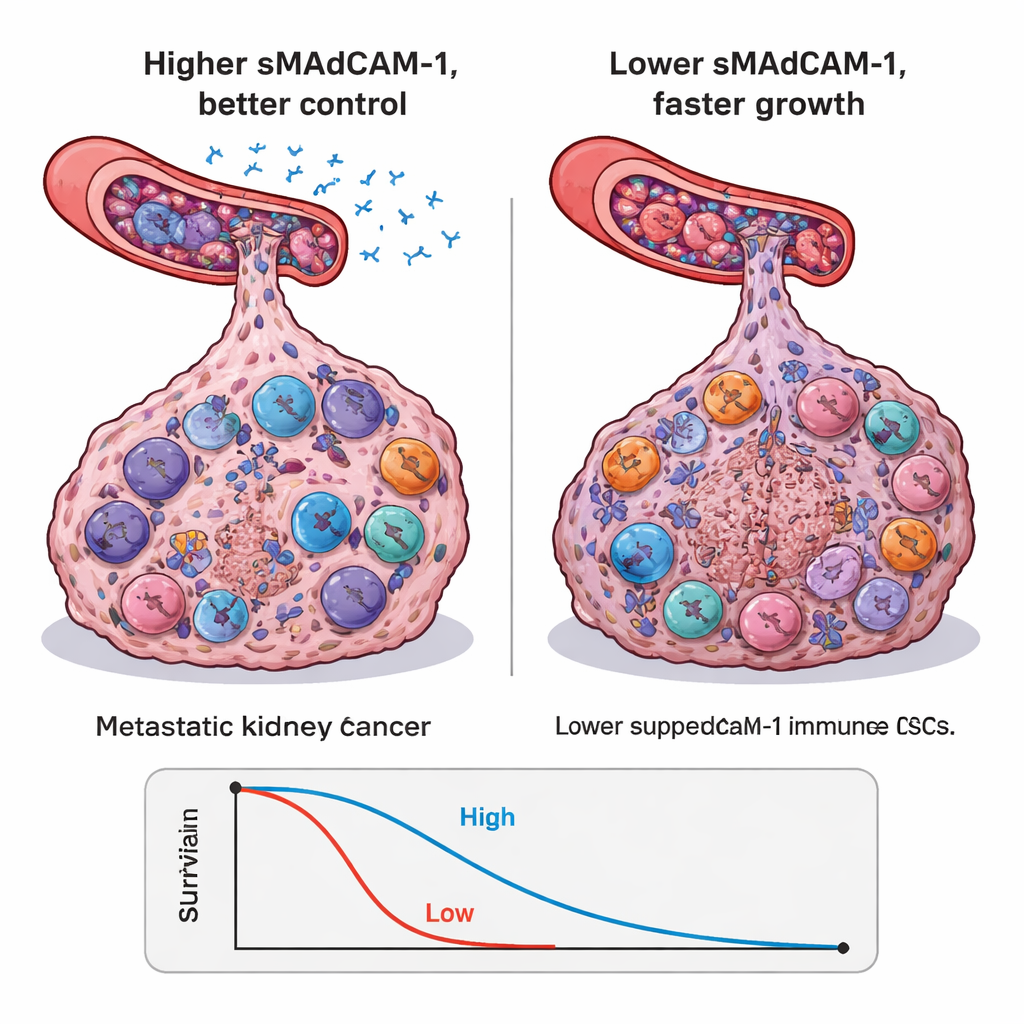
Across the largest trial, patients who started treatment with higher sMAdCAM-1 lived longer and went longer before their disease progressed, regardless of which drug regimen they received. Those with low levels had about half the chance of being alive at 18 months compared with those above the threshold, even after accounting for standard clinical risk scores. These findings held up in two independent validation trials: people with higher sMAdCAM-1 tended to survive longer, including those receiving immunotherapy after other treatments had failed. When the researchers looked at repeated blood samples over time, they saw that immunotherapy tended to nudge sMAdCAM-1 upward, while targeted drugs alone often lowered it. Patients whose levels stayed low over the first two treatment cycles had the worst outcomes.
The Microbiome Fingerprint Behind the Blood Test
To understand what the blood marker was reflecting, the investigators sequenced gut bacteria from patients in a separate microbiome study. Targeted therapies were linked to a less diverse community of gut microbes and an overgrowth of a genus called Enterocloster, previously tied to antibiotic use, chronic inflammation and poor responses to immunotherapy. Beneficial, immune-stimulating bacteria became scarcer. In a large pooled set of cancer patients, low sMAdCAM-1 levels were consistently associated with this kind of “unhealthy” microbiome pattern—dominated by Enterocloster and other species linked to shorter survival—while higher levels corresponded to richer, more balanced microbial communities.
What This Means for Patients and Future Care
For patients with advanced kidney cancer, the study suggests that a relatively simple blood test could flag those whose gut environment may be undermining their treatment. Low sMAdCAM-1 appears to signal both a more aggressive disease state and a microbiome that encourages suppressive immune cells to infiltrate tumors. While the marker by itself will not replace existing risk tools, it may help guide who should be offered microbiome-targeted strategies—such as carefully designed probiotics or fecal transplants—to restore a healthier gut–immune balance and potentially make immunotherapy work better.
Citation: Alves Costa Silva, C., Machaalani, M., Saliby, R.M. et al. Soluble MAdCAM-1 as a biomarker in metastatic renal cell carcinoma. Nat Med 32, 671–681 (2026). https://doi.org/10.1038/s41591-025-04067-x
Keywords: metastatic renal cell carcinoma, gut microbiome, biomarkers, immunotherapy resistance, MAdCAM-1