Clear Sky Science · en
CD38 endows local antigen-specific Treg cells with stress resilience for control of compartmentalized CNS inflammation
Why brain-guarding immune cells matter
Many autoimmune brain diseases, such as multiple sclerosis, behave like smoldering fires: an initial blaze of inflammation dies down, but damage can flare up again years later. This study asks a deceptively simple question with big implications: after an attack in the central nervous system (CNS), what keeps the peace—and what tips the balance back toward disease? The authors focus on a small population of immune cells called regulatory T cells that settle in the brain and spinal cord after inflammation, and uncover how a single surface molecule equips them to survive in a harsh environment and prevent relapses.
Immune peacekeepers that stay behind
Using a mouse model that mimics aspects of multiple sclerosis, the researchers tracked different types of T cells as disease rose to a peak and then entered a recovery phase. While ordinary helper T cells that drive inflammation shrank in number as symptoms improved, regulatory T cells—specialized cells that normally dampen immune responses—remained in the CNS. Their relative share within the local T cell pool actually increased, and they clustered in distinct niches around brain fluid spaces and the protective membranes covering the brain. By labeling where incoming T cells originated, the team showed that, after recovery, very few new regulatory T cells arrived from lymph nodes. Instead, the CNS population largely maintained itself locally, hinting at a long-term resident “peacekeeping” force. 
Stress-tolerant guardians of a hostile niche
The postinflammatory CNS is anything but comfortable for these guardians. After the main wave of disease, levels of the growth signal interleukin-2 (IL-2)—which regulatory T cells strongly rely on—drop because most conventional T cells have receded. At the same time, the tissue is rich in the metabolic molecule NAD+, which can trigger cell death or blunt key surface receptors when modified by a specific enzyme. By genetically tagging and following individual regulatory T cells, the authors distinguished “stable” cells that kept their identity from “unstable” ones that lost their hallmark Foxp3 program. Stable cells showed a much stronger signature of IL-2 signaling and higher levels of IL-2 receptors, suggesting that the ability to sense scarce IL-2 is central to surviving in this stressed environment.
Local control of relapses from within the brain
To test whether these resident regulatory T cells truly matter for long-term disease control, the researchers selectively removed them from the CNS using a toxin delivered directly into the brain fluid, while leaving most systemic regulatory T cells elsewhere in the body intact. Within days, mice that had been recovering developed severe relapses with renewed paralysis. In contrast, depleting regulatory T cells only in the body, sparing those in the CNS, did not trigger flare-ups. The relapse involved rapid reactivation and expansion of effector T cells that were already lodged in the CNS, even when their recruitment from the blood was blocked. This demonstrates that local regulatory T cells, rather than circulating ones, are essential to restrain residual aggressive cells that linger behind after the acute attack has passed.
How one enzyme shields regulatory cells
Digging deeper, the authors searched for molecules that distinguish stress-tolerant CNS regulatory T cells. A key player emerged: CD38, an enzyme on the cell surface that breaks down extracellular NAD+. In the inflamed CNS, regulatory T cells strongly increased CD38, while conventional T cells did not show such a marked shift. When the team transferred myelin-reactive regulatory T cells lacking CD38 into mice, these cells reached the CNS but failed to control disease, unlike their normal counterparts. In mixed chimeric animals, where only the resident regulatory T cells were CD38-deficient, removing the CD38-sufficient fraction was enough to provoke relapses, showing that CD38 acts in a cell-intrinsic way to enable effective control.
Protecting the lifeline signal
Mechanistic experiments revealed how CD38 confers this resilience. High NAD+ levels in the CNS can fuel chemical modification of surface proteins by the enzyme ARTC2.2. One sensitive target is the IL-2 receptor’s high-affinity subunit, which regulatory T cells express at very high levels, especially when recognizing brain antigens. In CD38-deficient regulatory T cells exposed to NAD+, most IL-2 receptors became modified, leading to weaker activation of the downstream messenger STAT5 and reduced responsiveness to IL-2. In contrast, CD38-expressing cells locally lowered NAD+ around themselves, limiting this harmful modification and preserving strong IL-2 signaling. In living mice, injecting NAD+ into the brain selectively killed most regulatory T cells via this pathway, while sparing conventional T cells, underlining how precarious the niche is without this protective enzyme. 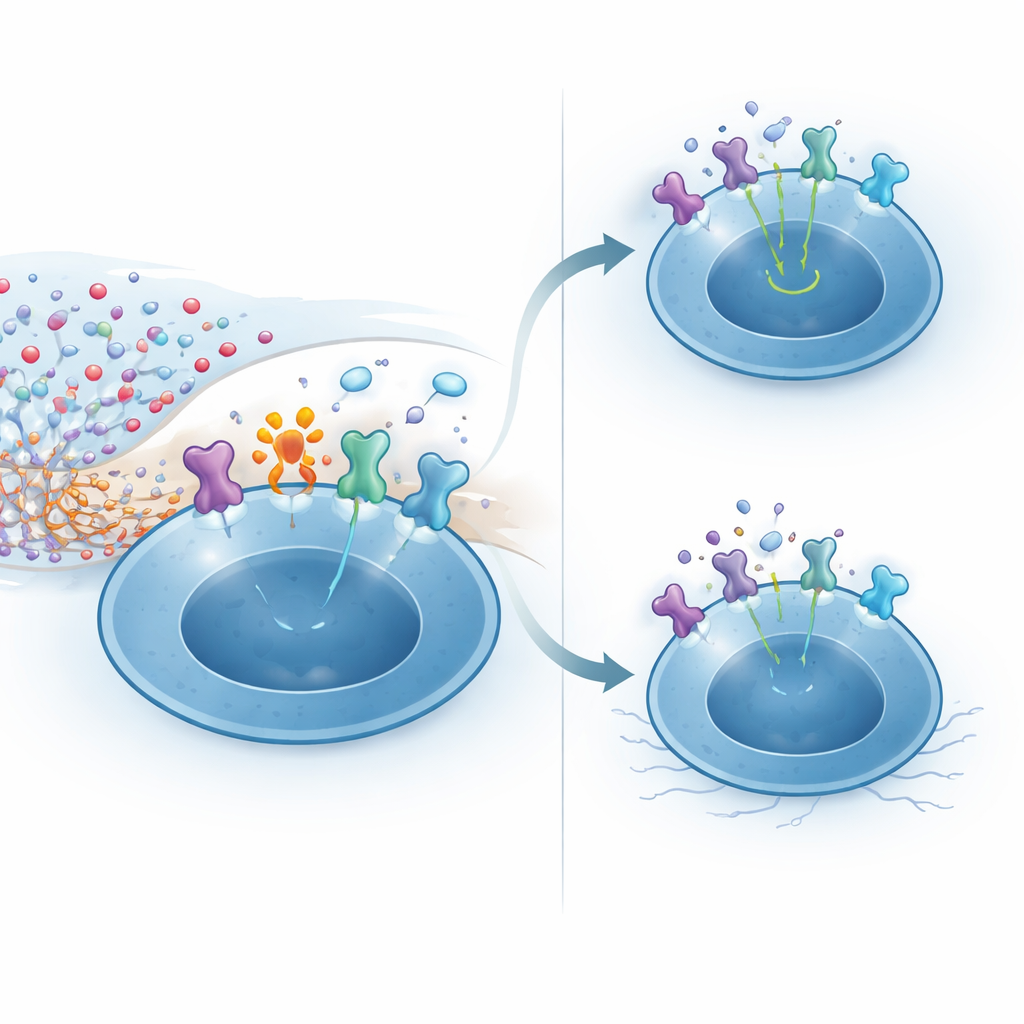
What this means for chronic brain autoimmunity
This work paints a picture of chronic CNS autoimmunity as a compartmentalized battle: small clusters of residual aggressive T cells reside in the brain and spinal cord, held in check by equally specialized local regulatory T cells. These guardians are “stress-tolerant” because CD38 allows them to survive high NAD+ levels and continue sensing minute amounts of IL-2, preserving their identity and suppressive function. For lay readers, the key takeaway is that long-term control of diseases like multiple sclerosis may depend less on the circulating immune system and more on nurturing and protecting these resident peacekeepers within the brain itself—potentially by targeting pathways that boost CD38 function or IL-2 sensitivity in regulatory T cells.
Citation: Chen, HH., Tyystjärvi, S., Ruiz Navarro, D. et al. CD38 endows local antigen-specific Treg cells with stress resilience for control of compartmentalized CNS inflammation. Nat Immunol 27, 516–529 (2026). https://doi.org/10.1038/s41590-025-02416-z
Keywords: regulatory T cells, multiple sclerosis, central nervous system inflammation, CD38 enzyme, immune tolerance