Clear Sky Science · en
Unraveling CD8 lineage decisions reveals that functionally distinct CD8+ T cells are selected by different MHC-I thymic peptides
How Killer T Cells Choose Their Jobs
Our immune system depends on CD8 T cells, often called killer T cells, to destroy virus‑infected or cancerous cells. Yet not all CD8 T cells behave the same way: some are fierce assassins, others act more like helpers, and some are born already poised as rapid‑response "memory" cells. This study uncovers how, during their brief childhood inside the thymus, CD8 T cells are steered into these very different careers by the precise signals they receive from the surrounding tissue.
A Training School Inside the Chest
Deep in the chest, the thymus acts as a finishing school for immature T cells. There, developing cells constantly test their receptors against small protein fragments, or peptides, displayed on other cells. Only those that recognize the body’s own molecules just strongly enough are allowed to mature. The authors focused on CD8 T cells, which are selected by peptides presented on molecules called MHC class I. Earlier models suggested that whether a T cell became a helper‑type or a killer‑type depended mainly on which surface "co‑receptor" protein it expressed, and on how long its receptor kept signaling during selection. But exactly how those pieces fit together, and what role the peptides themselves played, was unclear.
Rewiring T Cells to Expose Hidden Choices
To probe these questions, the researchers engineered mice, called CD8Dual mice, in which both of the usual co‑receptor gene locations were reprogrammed to produce CD8‑type co‑receptors. This clever wiring trick meant that all developing T cells in these animals were tuned to recognize MHC class I, letting the team isolate how signaling patterns alone shaped fate. Surprisingly, even though the cells used the same kind of co‑receptor and recognized the same class of MHC, two distinct CD8 T‑cell populations emerged. One group, marked by activity of a factor named ThPOK, behaved like classical helper cells: they expressed molecules that talk to other immune cells and could become regulatory or natural killer T‑like cells. The other group, marked by the factor Runx3, had the molecular and functional hallmarks of cytotoxic killers.
Signal Timing and the Journey Through the Thymus
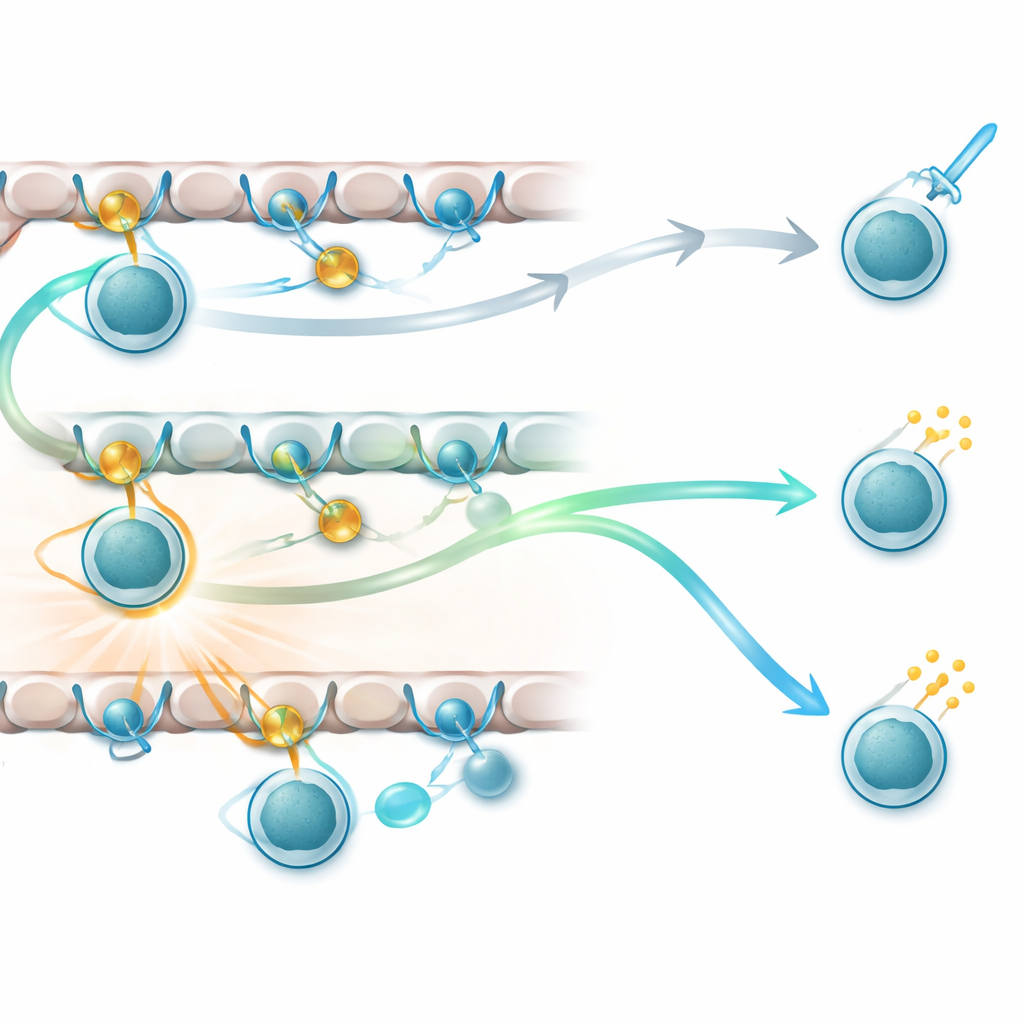
Why did genetically similar cells diverge so sharply? By tracking surface markers that report recent receptor activity, the authors showed that cells using one gene locus experienced continuous signaling as they migrated from the thymic cortex into the deeper regions; these cells adopted the helper‑like fate. In contrast, cells relying on the other locus experienced a break in signaling, after which local cytokines pushed them toward cytotoxic identity. This supported the idea that the duration of signaling, more than its mere presence, is what decides a CD8 T cell’s role. Yet an additional puzzle emerged: even when all cells carried exactly the same receptor for the same peptide—using special "monoclonal" T‑cell receptor mice—some still became helpers and others killers. That pointed to another layer of control: the nature and location of the selecting peptides themselves.
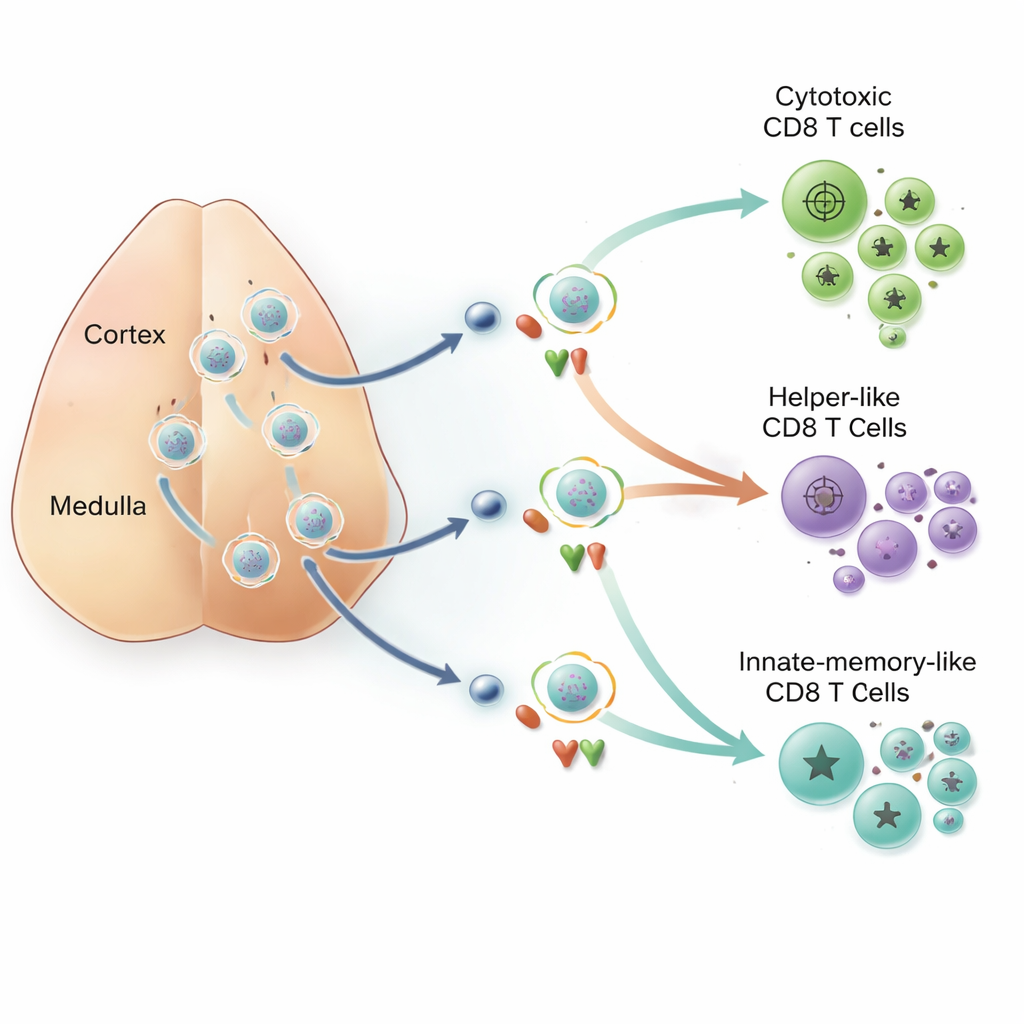
Different Peptides, Different Futures
Within the thymus, peptide fragments are generated by molecular shredders called proteasomes. A specialized version, the thymoproteasome, makes a set of peptides known as β5t‑peptides that appear only on cells in the outer cortex. Other, more widely made peptides, termed non‑β5t peptides, are present both in the cortex and deeper in the organ. By comparing normal mice with those lacking β5t, the team found that β5t‑peptides almost exclusively produced conventional cytotoxic CD8 T cells. In contrast, the helper‑like CD8 cells and a distinct group of "innate memory" CD8 cells—cells that leave the thymus already equipped for rapid responses—were selected only by the widely distributed non‑β5t peptides. These innate‑memory cells arose when developing cytotoxic‑type cells re‑encountered non‑β5t peptides later in their journey, receiving a second wave of signals while bathed in the cytokine IL‑4.
What This Means for Immunity
Put simply, this work shows that not all self‑peptides in the thymus are equivalent. Peptides found only in the cortex give T cells a brief burst of instruction and then go silent, producing classic killer CD8 cells. Peptides that are encountered again and again, as cells migrate deeper into the thymus, can prolong or rekindle signaling. When that signaling is strong and steady, CD8 cells adopt helper‑like traits; when it arrives later and combines with specific local cytokines, they become innate memory cells. By tying together peptide type, where it is displayed in the thymus, and how long T‑cell receptors stay engaged, the study explains how a single training organ can generate a diversified CD8 T‑cell army tailored for immediate killing, immune coordination, or rapid recall responses.
Citation: Shinzawa, M., Ramos, N., Bui, K. et al. Unraveling CD8 lineage decisions reveals that functionally distinct CD8+ T cells are selected by different MHC-I thymic peptides. Nat Immunol 27, 786–798 (2026). https://doi.org/10.1038/s41590-025-02411-4
Keywords: CD8 T cell development, thymic selection, MHC class I peptides, innate memory T cells, T cell lineage fate