Clear Sky Science · en
Cell cycle arrest enhances CD8+ T cell effector function by potentiating glucose metabolism and IL-2 signaling
Putting Cancer-Fighting Cells on Pause
Most cancer drugs that halt cell division are designed with tumors in mind, but they also affect the immune system’s frontline soldiers—CD8 T cells. This study asks an unexpected question: what if briefly pausing these killer T cells could actually make them better at hunting tumors? The answer, drawn from mouse models, human cells, and patient samples, is that a well-timed “stop” can supercharge these cells for a stronger, longer-lasting anti-cancer attack.
A Short Stop Before the Charge
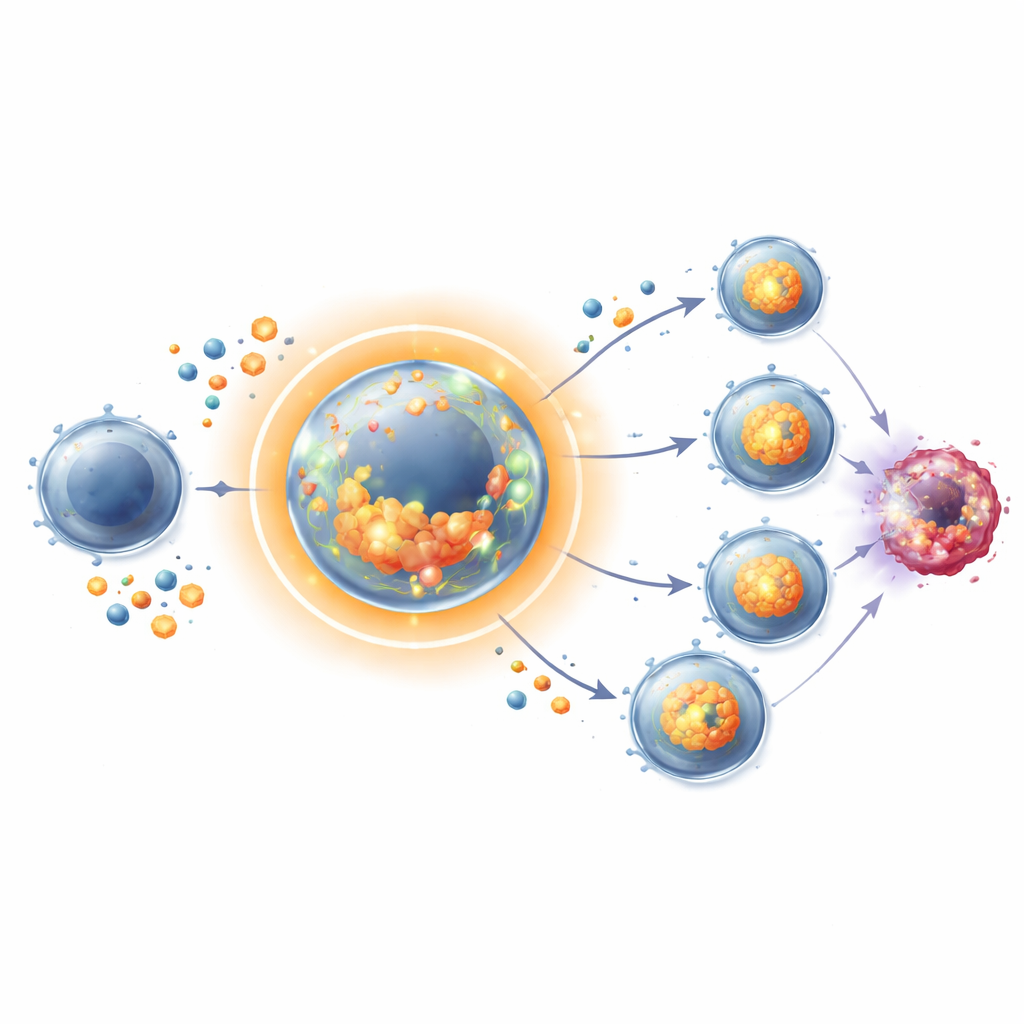
When CD8 T cells first recognize cancer or virus signals, they normally rush through rounds of division while simultaneously maturing into efficient killers. The researchers used common cell cycle–blocking drugs, such as hydroxyurea and CDK4/6 inhibitors, to temporarily uncouple these two processes: cells were allowed to activate and begin differentiating, but their division was briefly halted. Surprisingly, these arrested T cells did not become exhausted or dysfunctional. Instead, after the drug was removed, they divided faster and reached higher numbers than T cells that were never paused, in both dish experiments and vaccinated mice. Effector features—such as production of toxic molecules that kill cancer cells—were preserved or even enhanced upon release.
Stockpiling Fuel Inside Resting Warriors
To understand how a non-dividing T cell could later outgrow its continuously cycling counterpart, the team examined its internal chemistry. During the arrest period, these T cells behaved like athletes carb-loading before a race. They increased the number of transporters that pull in glucose and amino acids, accumulated sugars, and built up glycogen stores—bundles of reserve carbohydrate. They also boosted enzymes that drive glycolysis, the rapid breakdown of sugar for energy, and stepped up mitochondrial and cholesterol metabolism important for making new membranes. In essence, while the cells were not spending energy on division, they were quietly filling their tanks and upgrading their power plants. When the block was lifted, they rapidly burned through these reserves to fuel intense proliferation.
A Self-Made Growth Signal
Fuel alone did not explain the surge. The paused T cells also ramped up production of interleukin-2 (IL-2), a powerful immune growth factor that T cells can both secrete and sense themselves. During arrest, many cells produced high amounts of IL-2 and displayed more of its receptor, making them highly responsive to their own signal. Downstream pathways involving the STAT5 protein lit up strongly when the cells were released, supporting vigorous division even when a major growth regulator, the mTORC1 pathway, was partially blocked. Genetic and drug-based experiments showed that taking IL-2 away sharply curtailed this boosted proliferation, while adding IL-2 back restored it. The pause therefore imprints a memory-like state: metabolically primed cells that are wired to respond explosively to their own growth cues.
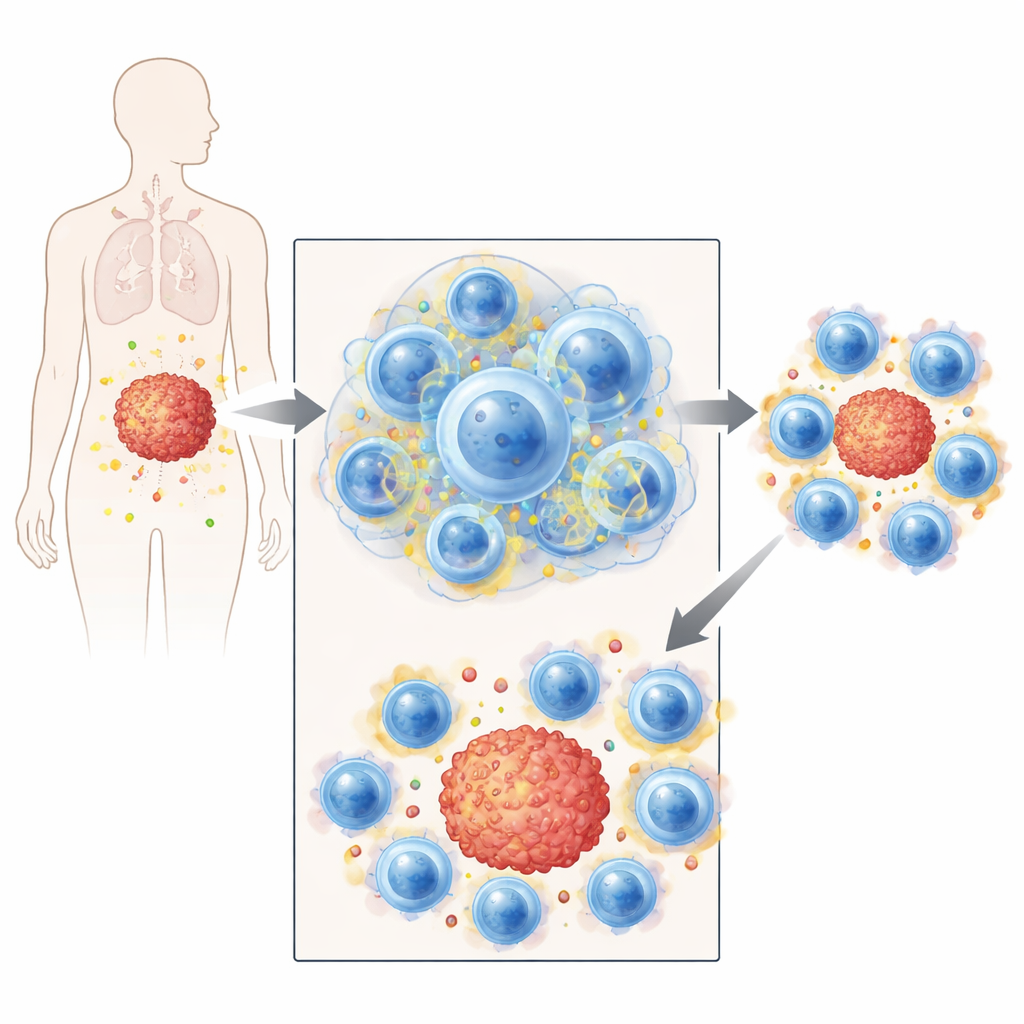
Better Tumor Control and Therapy Combinations
The real test was whether this strategy improves cancer control. In several mouse tumor models, transient treatment with cell cycle–blocking drugs increased the number and activity of metabolically primed CD8 T cells in blood, lymph nodes, and within tumors. These cells showed higher uptake of glucose and elevated markers of cholesterol and energy metabolism. Tumor-specific T cells expanded more strongly after the drug was withdrawn and expressed markers associated with effective tumor killing. Brief arrest synergized with multiple immunotherapy approaches: adoptively transferred T cells pretreated with hydroxyurea eradicated tumors more efficiently; combining transient arrest with PD-L1 checkpoint blockade delayed tumor growth and prolonged survival; and pairing a therapeutic cancer vaccine with arrest significantly improved outcomes. Early clinical biopsy data from women receiving ribociclib plus hormone therapy for breast cancer hinted at a similar metabolic boost in tumor-infiltrating CD8 T cells.
Turning a Chemotherapy Side Effect into an Advantage
Overall, the study reveals that a temporary halt in division can be turned from a liability into a feature for cancer-fighting T cells. By pausing at the right moment, these cells stockpile nutrients, increase their metabolic horsepower, and bathe themselves in their own growth signals. Once released, they expand rapidly and attack tumors more effectively, especially when combined with existing immunotherapies. For patients, this work suggests that carefully timed use of cell cycle–inhibiting drugs could be redesigned not just to poison tumors, but to train the immune system to fight them harder and longer.
Citation: van Haften, F.J., van der Sluis, T.C., Hepp, H.S. et al. Cell cycle arrest enhances CD8+ T cell effector function by potentiating glucose metabolism and IL-2 signaling. Nat Immunol 27, 463–475 (2026). https://doi.org/10.1038/s41590-025-02407-0
Keywords: CD8 T cells, cell cycle arrest, cancer immunotherapy, T cell metabolism, interleukin-2