Clear Sky Science · en
A GDF-15–GFRAL axis controls autoimmune T cell responses during neuroinflammation
Why pregnancy can calm a misfiring immune system
Doctors have long noticed a medical mystery: many women with multiple sclerosis, a disease in which the immune system attacks the brain and spinal cord, often feel better during pregnancy. Their flare-ups drop dramatically, only to return after birth. This study uncovers a key piece of that puzzle, revealing a signal that flows from the body to a tiny region of the brain and back out to the immune system, dialing down harmful immune attacks without shutting defenses off completely. 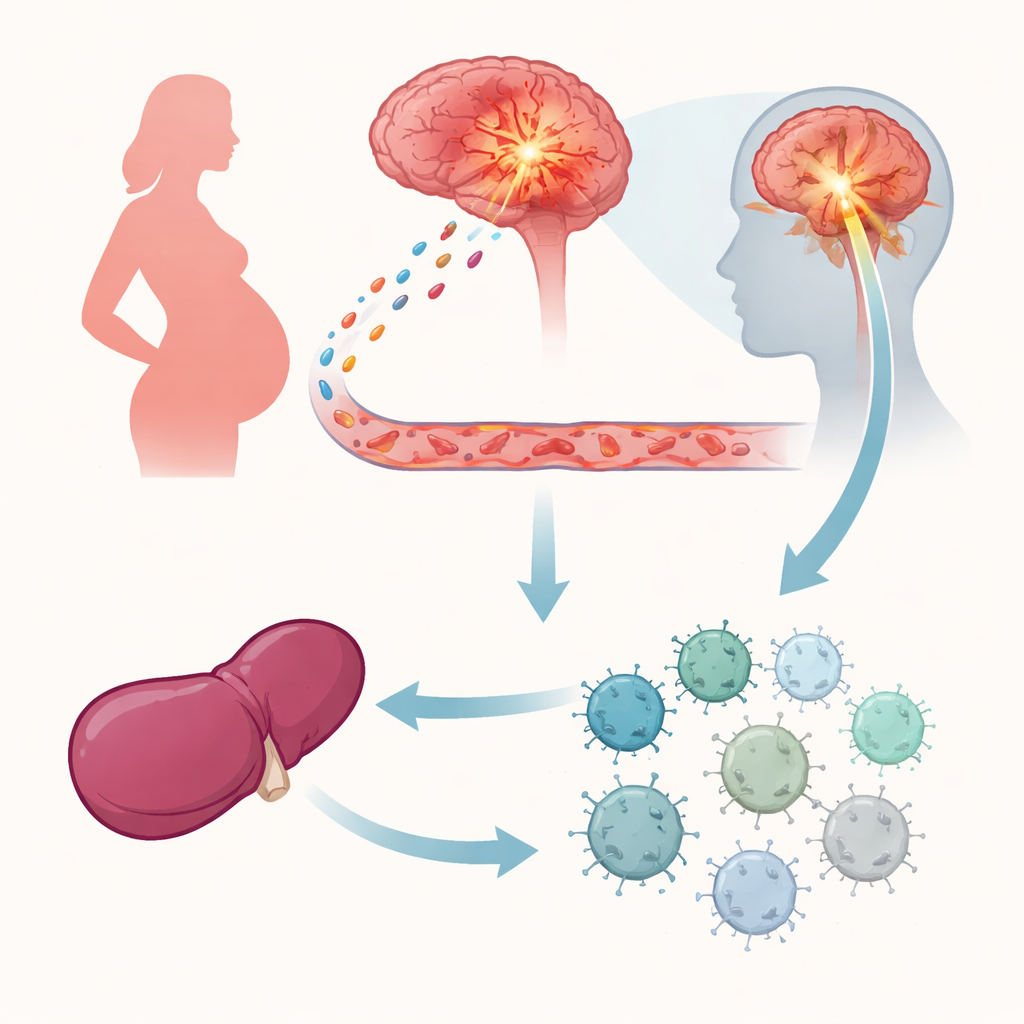
A messenger that rises in pregnancy and brain disease
The researchers focused on a protein called GDF-15, which circulates in the blood. They measured GDF-15 levels in pregnant women and in mice and found that this molecule steadily increases over the course of pregnancy. Women with multiple sclerosis who stayed relapse-free during pregnancy had higher GDF-15 levels than those who experienced disease flare-ups. In mice carrying genetically different offspring, GDF-15 levels rose particularly strongly, and lower levels were linked to smaller litters, hinting that this protein helps the mother’s body tolerate the semi-foreign fetus.
When the brain is inflamed, the signal turns up
The team then explored what happens during experimental neuroinflammation, a mouse model of multiple sclerosis. In these animals, cells in the brain and spinal cord began producing large amounts of GDF-15 right at the sites of inflammation. Immune cells that had infiltrated the central nervous system also contributed to this surge. Mice that lacked GDF-15 became sicker, recovered poorly and showed more aggressive brain immune cells, suggesting that the molecule normally acts as a brake to help resolve inflammation rather than prevent it from starting.
Brainstem “switchboard” sends calming signals to the spleen
GDF-15 can only trigger signals by binding to a specific sensor called GFRAL, which is found almost exclusively on neurons in a small brainstem area that lies outside the usual blood–brain barrier. The scientists used gene therapy and protein injections to raise GDF-15 in mice and found that this powerfully protected against neuroinflammation, even after disease was underway, without relying on stress hormones or simple weight loss. When GFRAL was missing, or when a mutant form of GDF-15 that could no longer bind this receptor was used, the protection disappeared. Activating GFRAL-positive neurons directly, using a chemogenetic tool that lets researchers turn cells on with a designer drug, was enough by itself to prevent immune cells from flooding into the spinal cord. 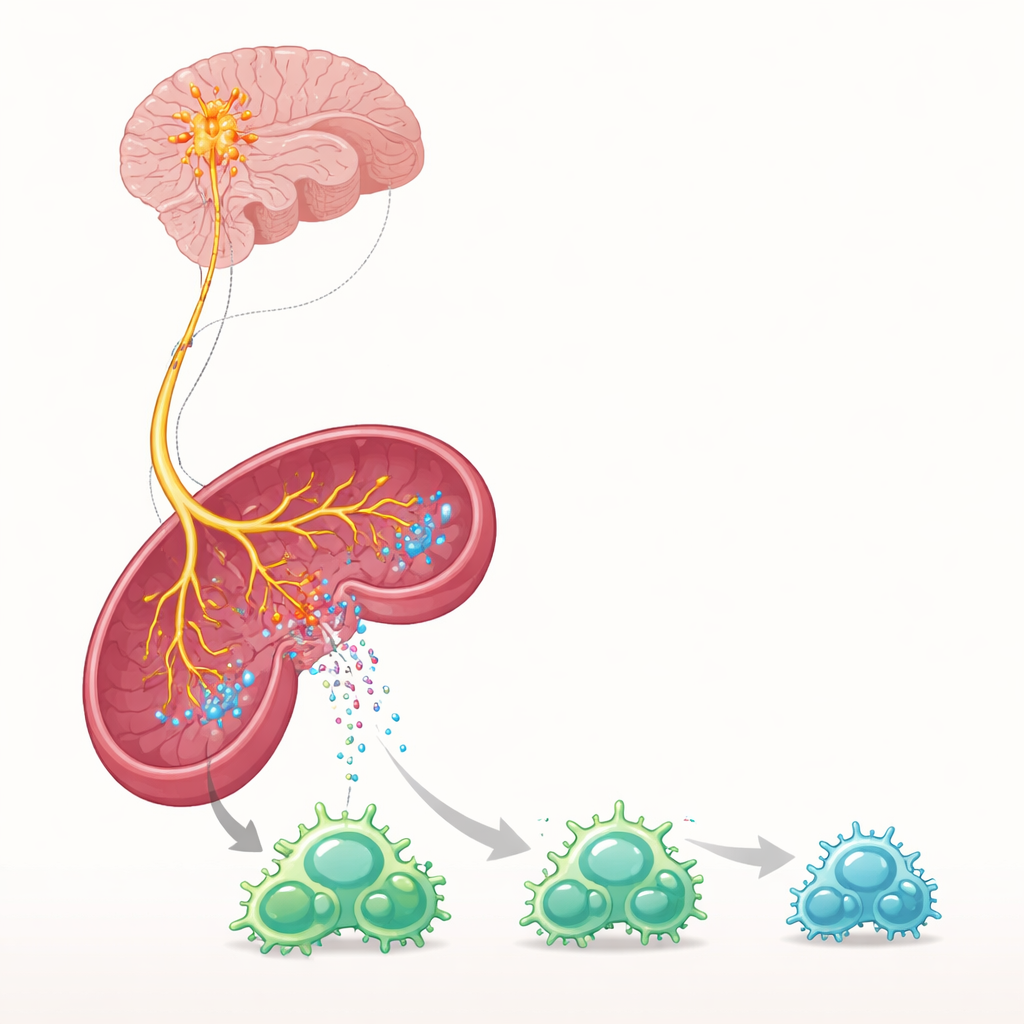
How nerve signals reshape T cells
To understand how a tiny group of neurons could control a large immune response, the team traced their effects to the spleen, a major hub for immune cells. Boosting GDF-15 or stimulating GFRAL-positive neurons increased the activity of sympathetic nerve fibers in the spleen and raised levels of norepinephrine, a chemical messenger better known for its role in the “fight-or-flight” response. When spleen T cells were exposed to norepinephrine or similar drugs, they divided less, displayed weaker activation markers and reduced levels of key surface “grip” proteins that normally help them stick to blood vessel walls and squeeze into the brain. In living animals, this translated into fewer activated T cells in lymphoid organs and dramatically fewer immune cells reaching the brain and spinal cord.
A new brain–immune circuit with treatment potential
Taken together, the findings reveal a finely tuned circuit: pregnancy or tissue stress raises GDF-15, this signal reaches GFRAL-expressing neurons in the brainstem, those neurons activate sympathetic nerves to the spleen, and norepinephrine then reins in potentially harmful T cells by limiting their activation, growth and ability to enter the brain. Rather than broadly suppressing immunity, this pathway selectively cools autoimmune attacks. Because druglike versions of GDF-15 and tools that target its receptor have already been tested for other conditions, this newly charted brain–immune axis offers a promising route to future therapies for multiple sclerosis and possibly other autoimmune diseases.
Citation: Sonner, J.K., Kahn, A., Binkle-Ladisch, L. et al. A GDF-15–GFRAL axis controls autoimmune T cell responses during neuroinflammation. Nat Immunol 27, 503–515 (2026). https://doi.org/10.1038/s41590-025-02406-1
Keywords: multiple sclerosis, neuroinflammation, immune tolerance, neuroimmune circuit, GDF-15