Clear Sky Science · en
BACH2 dosage establishes the hierarchy of stemness and fine-tunes antitumor immunity in CAR T cells
Why this research matters for future cancer treatment
Engineered immune cells called CAR T cells have transformed treatment for some blood cancers, yet many patients still relapse and responses against solid tumors remain modest. This study asks a deceptively simple question with big implications: can we “tune” a single molecular dial inside CAR T cells to make them act more like long-lived, self-renewing stem cells and less like short-lived, exhausted fighters? The answer, the authors find, is yes—and that dial is a protein called BACH2.
A hidden pecking order inside cancer-fighting T cells
Not all CAR T cells are equal. Within an infusion bag there is a hierarchy of cells with different levels of “stemness”—their capacity to renew themselves, persist in the body and repeatedly generate fresh waves of tumor-killing cells. At the top of this hierarchy are long-term stem-like T cells that can endure and remember. Using single-cell genomic tools in mice, the researchers mapped this hierarchy and discovered a subset of CAR T cells whose DNA packaging and gene activity closely matched this long-lived state. These elite cells showed open regions in genes linked to memory and survival, and closed regions in genes tied to terminal differentiation and exhaustion. Among their most distinctive features was strong activity of BACH2, a protein that helps keep T cells in a flexible, stem-like condition.
The better CAR T cells already carry a BACH2 signature
The team then asked whether these long-term stem-like cells actually perform better against tumors. In mouse models of leukemia and neuroblastoma, CAR T cells enriched for a surface marker of the long-lived subset expanded more, resisted cell death, displayed fewer exhaustion signals and controlled tumors more effectively than their more mature counterparts. Looking at human data from 40 people treated with CD19 CAR T cells for B cell lymphoma, the authors reanalyzed single-cell RNA profiles from the infusion products. Patients who went into complete remission had CD8 T cells that more strongly expressed BACH2 and other long-term stem-like genes compared with those who did not. This suggests that having more BACH2-marked stem-like cells in the bag may stack the odds toward durable responses.
A key switch that keeps T cells youthful
To move from correlation to cause, the researchers selectively deleted BACH2 in mouse CAR T cells. Without this protein, CAR T cells expanded poorly, rapidly lost stem-like markers, and upregulated exhaustion molecules such as PD-1, TIM3 and the regulator TOX. Their ability to control tumors and to mount a strong “recall” response upon seeing the cancer again was impaired. Genomic binding experiments showed that BACH2 sits on stretches of DNA that help restrain other factors, like JunB, which are known to drive exhaustion. In effect, BACH2 acts as a guardian that keeps the cells in a poised, long-lived state and prevents an early slide into burnout.
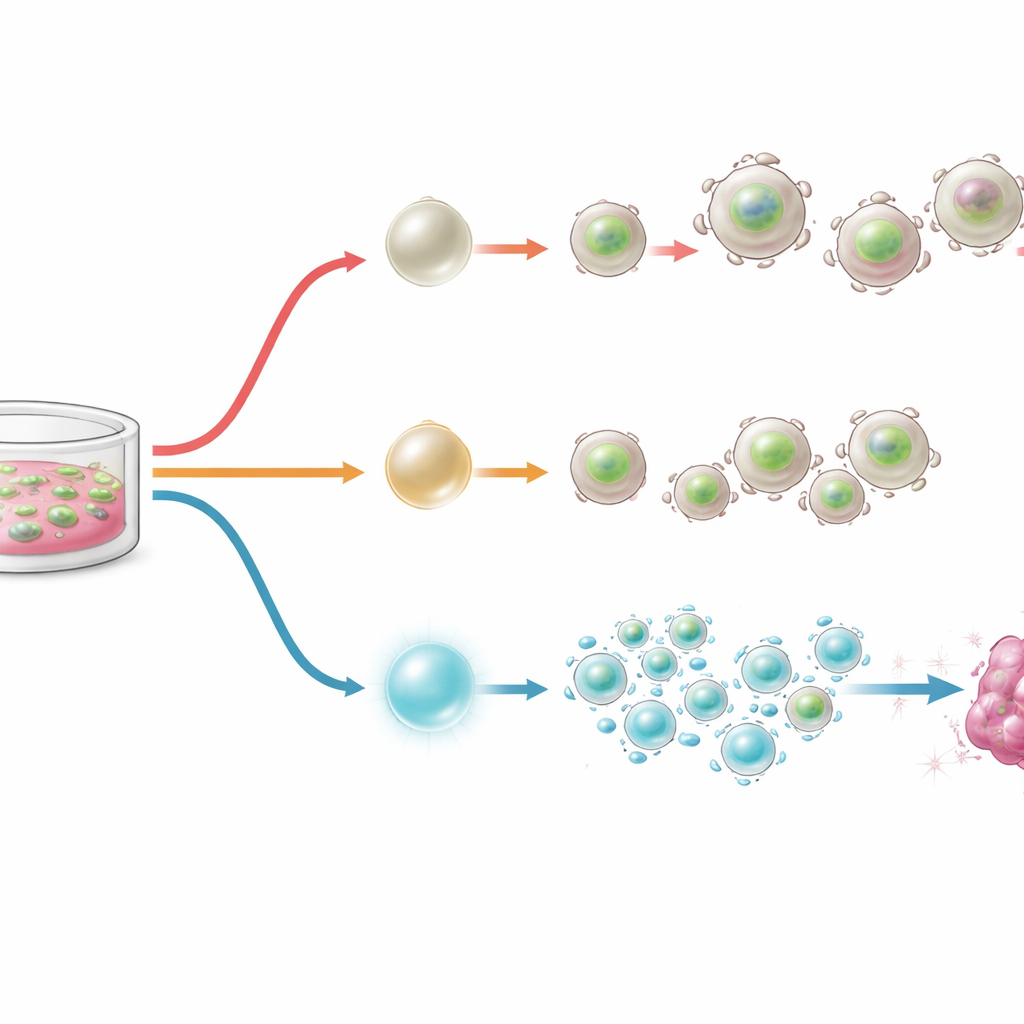
Dialing BACH2 up and down with designer switches
Armed with this knowledge, the authors engineered molecular switches that let them finely adjust BACH2 levels during CAR T cell manufacturing. In one design, BACH2 was fused to a tag that makes it unstable unless a small molecule, Shield-1, is added; more Shield-1 means more BACH2 protein. In another, BACH2 was linked to a hormone-sensitive domain that moves the protein into the nucleus only when exposed to a drug related to tamoxifen. In lab cultures of CAR T cells prone to chronic activation, gradually increasing BACH2 boosted the fraction of stem-like cells and diminished exhaustion markers in a dose-dependent way. Strikingly, even brief activation of BACH2 during manufacturing left a lasting imprint: after the drug was removed, cells retained more stem-like features and fewer exhaustion traits.
Putting tuned CAR T cells to the test against solid tumors
The ultimate test was whether this molecular fine-tuning could make a practical difference in living animals. When mice bearing a difficult-to-treat solid neuroblastoma were given CAR T cells that had BACH2 switched on during manufacturing, the cells controlled tumors better than standard CAR T cells. Similar strategies worked with human CAR T cells in the lab, where chemically induced BACH2 increased markers of memory and reduced exhaustion signals. These results show that BACH2 is not merely a marker of desirable cells but a lever that scientists can pull to sculpt CAR T behavior.
What this could mean for patients
For people facing cancer, this work points to a future in which CAR T therapies are not just turned on or off but carefully tuned. By adjusting the dosage and timing of a single regulator, BACH2, during the manufacturing process, clinicians may be able to enrich for long-lived, stem-like CAR T cells that endure, adapt and keep tumors in check—especially solid tumors that are currently hard to treat. While much remains to be tested in clinical settings, the study offers a blueprint for making smarter, more durable cell therapies by dynamically controlling the internal programs that decide whether a T cell stays youthful or burns out too soon.
Citation: Hu, T., Zhu, Z., Luo, Y. et al. BACH2 dosage establishes the hierarchy of stemness and fine-tunes antitumor immunity in CAR T cells. Nat Immunol 27, 425–435 (2026). https://doi.org/10.1038/s41590-025-02388-0
Keywords: CAR T cells, BACH2, T cell exhaustion, cancer immunotherapy, stem-like T cells