Clear Sky Science · en
Spatial patterning of fibroblast TGFβ signaling underlies treatment resistance in rheumatoid arthritis
Why stubborn joint pain matters
Many people with rheumatoid arthritis take modern medicines that calm the immune system, yet their joints remain painful and stiff. This study asks why some patients do not get better even when inflammation in the joint appears to fade. By looking closely at where and how certain support cells in the joint switch on scarring programs, the researchers uncover a hidden form of damage that could explain ongoing pain and point to new treatment strategies.
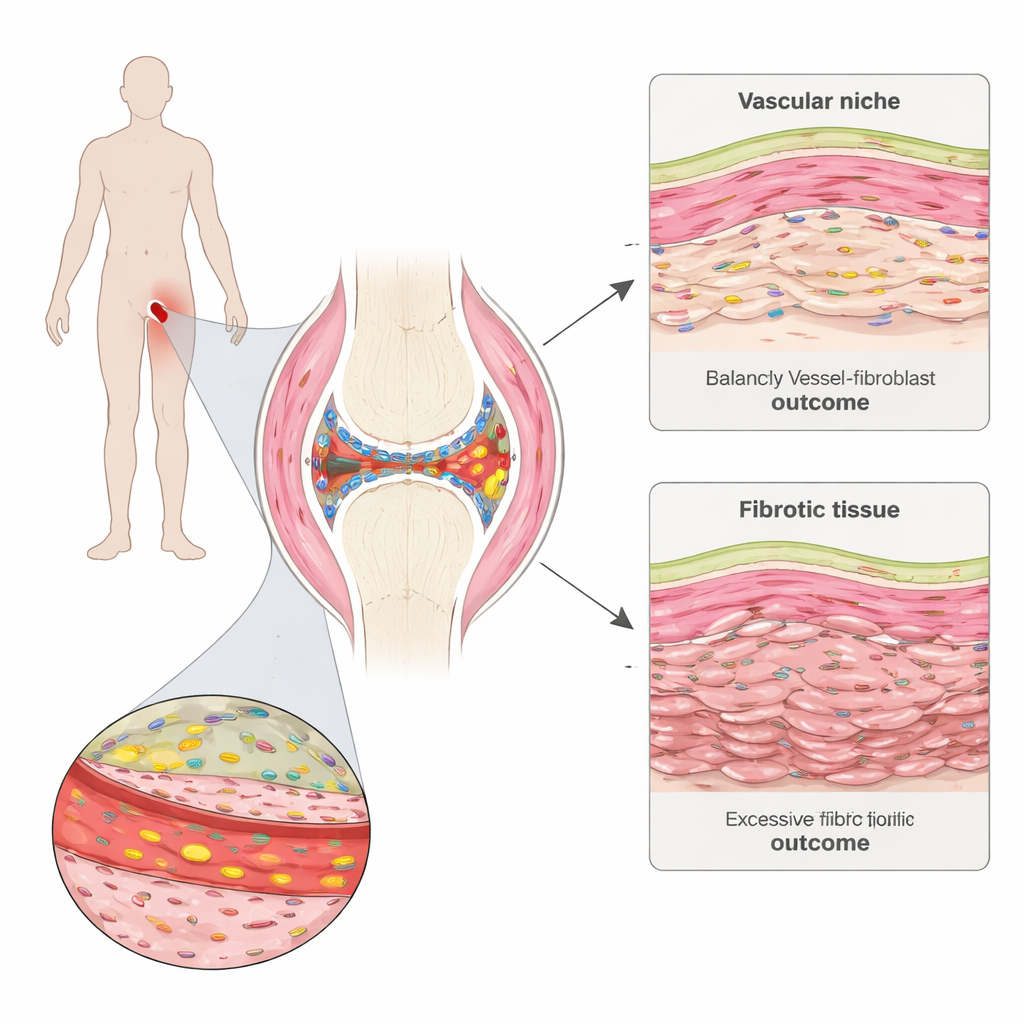
A closer look inside the aching joint
Rheumatoid arthritis attacks the synovium, a thin tissue that lines and lubricates joints. The team collected tiny synovial biopsies from people with early disease before starting therapy and again six months later. Using advanced spatial gene mapping, they could see which genes were active in thousands of individual cells while preserving where those cells sat in the tissue. They identified distinct “neighborhoods” in the synovium, including immune cell clusters, fat-rich areas, the thin inner lining, and regions packed with blood vessels and structural cells called fibroblasts.
Scar‑forming cells and treatment resistance
Comparing patients who later reached remission with those who did not, the researchers found a striking pattern: before treatment, non‑remitting patients already had stronger signals of tissue scarring, or fibrosis. A particular fibroblast program, marked by high production of an extracellular matrix protein called COMP, was expanded in these patients. These COMP‑rich fibroblasts shared features with scar‑forming cells seen in lung and skin fibrosis and were most strongly linked to poor treatment response. Over time, regions dominated by these cells tended to become densely packed with connective tissue but relatively poor in cells, suggesting that they lay down stiff matrix that can persist even after inflammation subsides.
Blood vessels as hidden conductors
The fibrogenic fibroblasts were not scattered randomly. They clustered around blood vessels, forming layered perivascular zones. The innermost layer, hugging the vessel wall, expressed different genes than the outer layer. The team showed that cells lining blood vessels send Notch signals to nearby fibroblasts, which in turn shape how these fibroblasts respond to a family of pro‑scarring cues known as TGF‑beta. Close to the vessel, Notch signaling pushes fibroblasts to make TGF‑beta but simultaneously lowers the number of TGF‑beta receptors on their surface, keeping their sensitivity in check. Farther away, where Notch influence fades, fibroblasts carry more receptors and become highly responsive, turning into COMP‑rich cells that drive fibrosis.
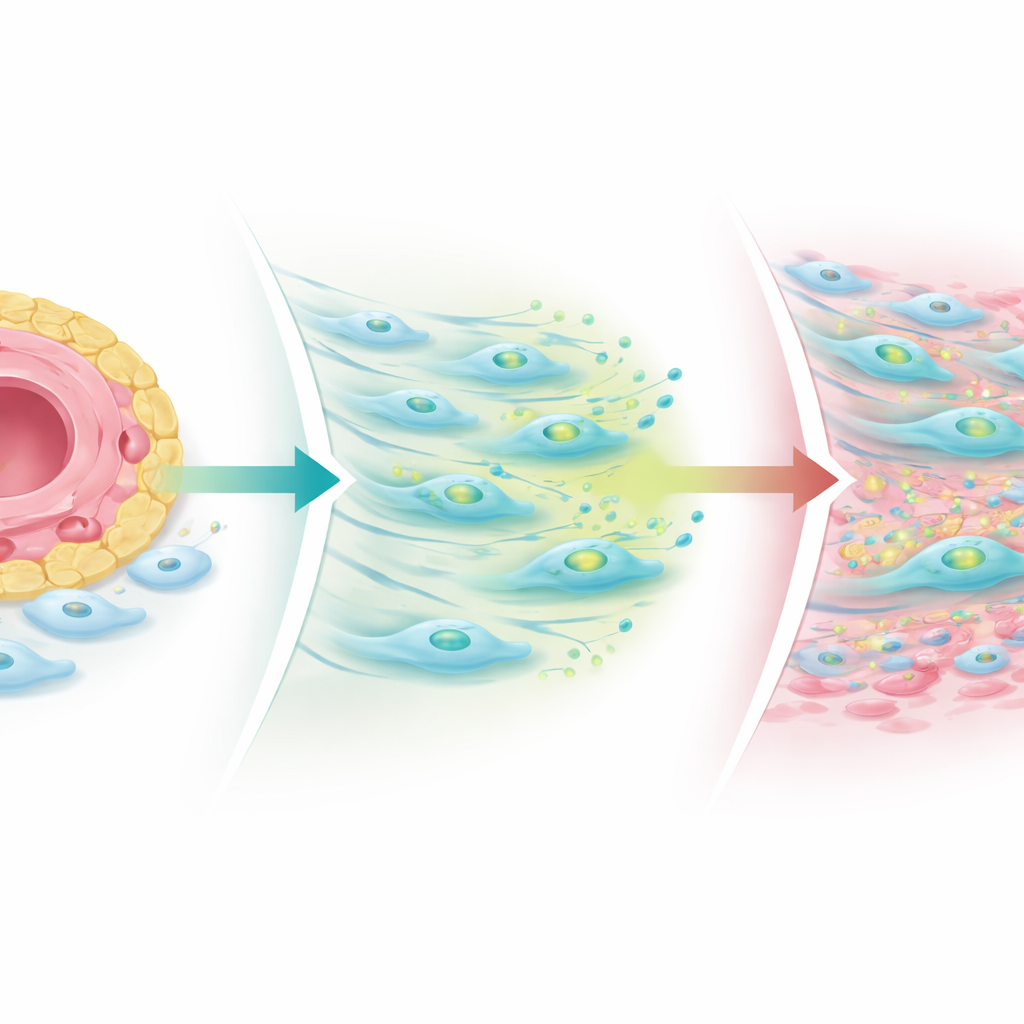
What happens when the balance is broken
The researchers built simplified joint models in laboratory dishes and three‑dimensional organoids made from patient tissue to probe this mechanism. When they strengthened Notch signals from endothelial cells, fibroblasts increased TGF‑beta production but reduced receptor levels, limiting scarring. When they blocked Notch or disturbed its steady pattern, fibroblasts regained TGF‑beta receptors—especially a co‑receptor called TGF‑beta receptor III—and COMP‑positive fibrogenic cells expanded away from vessels. In patient biopsies taken after treatment, immune cell clusters shrank in almost everyone, but fibrogenic niches, particularly COMP‑rich regions, often grew, especially in those whose joint pain persisted. This suggests that standard anti‑inflammatory drugs can drain the immune “fire” while leaving behind or even unmasking a scarring process that keeps joints stiff and sore.
New ways to calm stubborn joints
Finally, the team tested whether disrupting this fibrogenic circuit could be therapeutically useful. In patient‑derived organoids, drugs that inhibit Notch or block TGF‑beta signaling reduced production of COMP, other scar‑related proteins, and key collagens, and rewired gene activity in perivascular zones. To a lay observer, the message is that not all rheumatoid arthritis damage comes from visible inflammation: some of it is “hard‑wired” into the joint’s supporting cells and blood vessels. By targeting the Notch–TGF‑beta conversation that controls how fibroblasts lay down scar tissue, future treatments might prevent or reverse a stubborn, fibrotic form of the disease that resists today’s immune‑focused drugs.
Citation: Bhamidipati, K., McIntyre, A.B.R., Kazerounian, S. et al. Spatial patterning of fibroblast TGFβ signaling underlies treatment resistance in rheumatoid arthritis. Nat Immunol 27, 556–571 (2026). https://doi.org/10.1038/s41590-025-02386-2
Keywords: rheumatoid arthritis, fibrosis, fibroblasts, TGF-beta signaling, Notch pathway