Clear Sky Science · en
A single allosteric site merges activation, modulation and inhibition in TRPM5
How One Tiny Gatekeeper Shapes Taste and Metabolism
The foods we enjoy and how our bodies handle sugar both depend on microscopic “gatekeeper” proteins in our cells. One such gatekeeper, a channel called TRPM5, helps us sense sweet, bitter and savory flavors and helps the pancreas release insulin. This study reveals how a single small pocket on TRPM5 can act like a master control knob, turning the channel on, tuning its sensitivity and even shutting it off—insights that could guide future treatments for diabetes, obesity and gut disorders.
A Channel at the Crossroads of Taste and Blood Sugar
TRPM5 sits in the membranes of taste cells in the tongue, hormone-producing cells in the gut and insulin-secreting cells in the pancreas. When calcium levels inside these cells rise, TRPM5 opens and lets positively charged ions flow, briefly changing the cell’s voltage. In taste buds, this electrical signal tells the brain that something sweet, bitter or umami is on the tongue. In the pancreas, it helps fine-tune bursts of insulin after a meal. People and animals with impaired TRPM5 function show problems with insulin release and blood sugar control, suggesting that drugs targeting TRPM5 might one day help treat metabolic disease. Yet until recently, researchers lacked precise tools to switch this channel on or off.
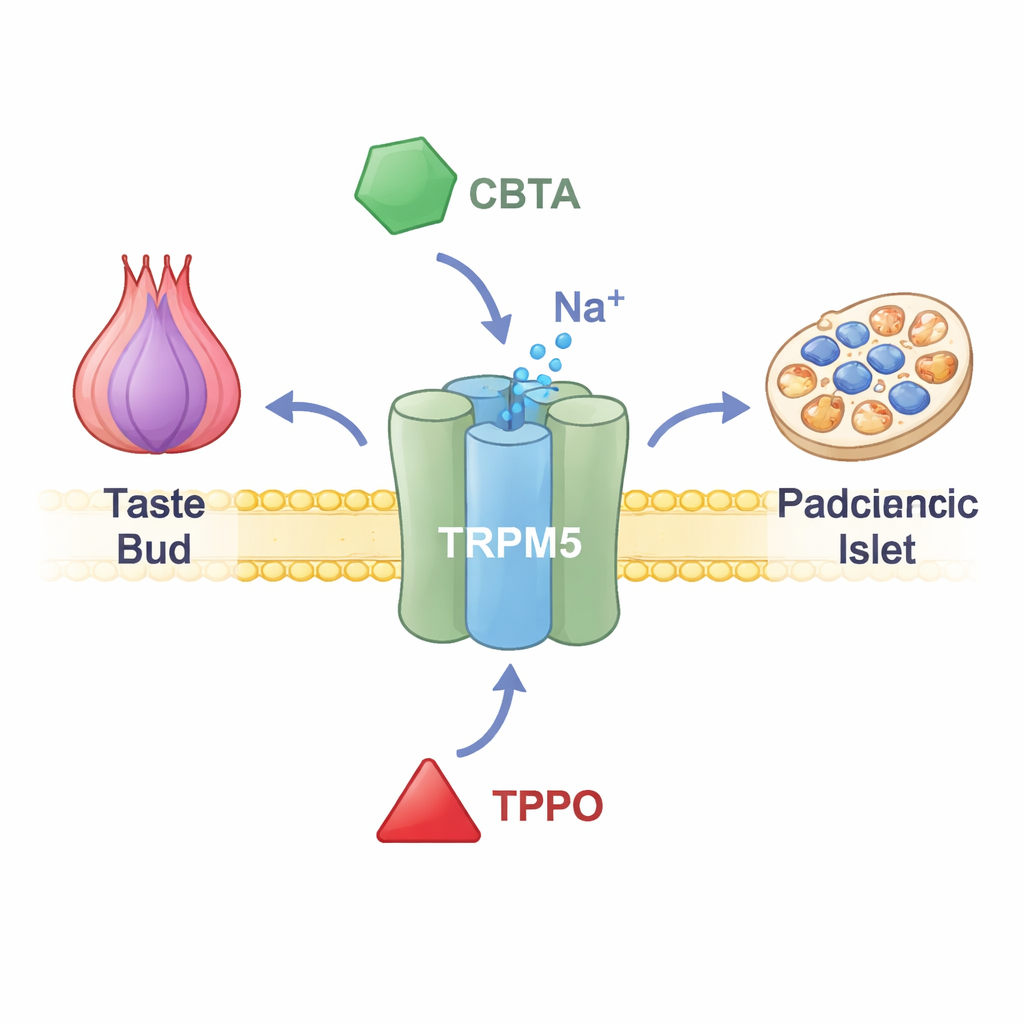
A New Chemical Switch That Supercharges the Channel
The researchers focused on a synthetic compound called CBTA, previously known to stimulate TRPM5 but not well understood. Using electrical recordings from cells engineered to produce TRPM5, they showed that CBTA can open the channel even when calcium is nearly absent, proving it acts as a true activator rather than just boosting calcium’s effects. When tiny amounts of calcium are present—levels that normally leave TRPM5 quiet—CBTA and calcium act together, generating much larger currents than either alone. This means CBTA makes TRPM5 extraordinarily sensitive, so that near-resting calcium levels suddenly become strong enough to flip the channel wide open.
A Hidden Control Pocket Revealed by Cryo-EM
To see how this works in atomic detail, the team used cryo–electron microscopy to capture three-dimensional snapshots of TRPM5 under different conditions. They discovered that CBTA nestles into a previously unnoticed cavity in the upper part of a voltage-sensing region of the channel, just above where calcium usually binds. This cavity, formed by a cluster of amino acids, acts as a precise docking site. When the scientists mutated key building blocks that line this pocket, CBTA could no longer activate TRPM5, even though calcium still worked, confirming that this tiny niche is essential for the drug’s action. Strikingly, CBTA binding subtly rearranges nearby parts of the protein in a way that makes it much easier for calcium to grab its usual spot, explaining the dramatic synergy seen in the electrical measurements.
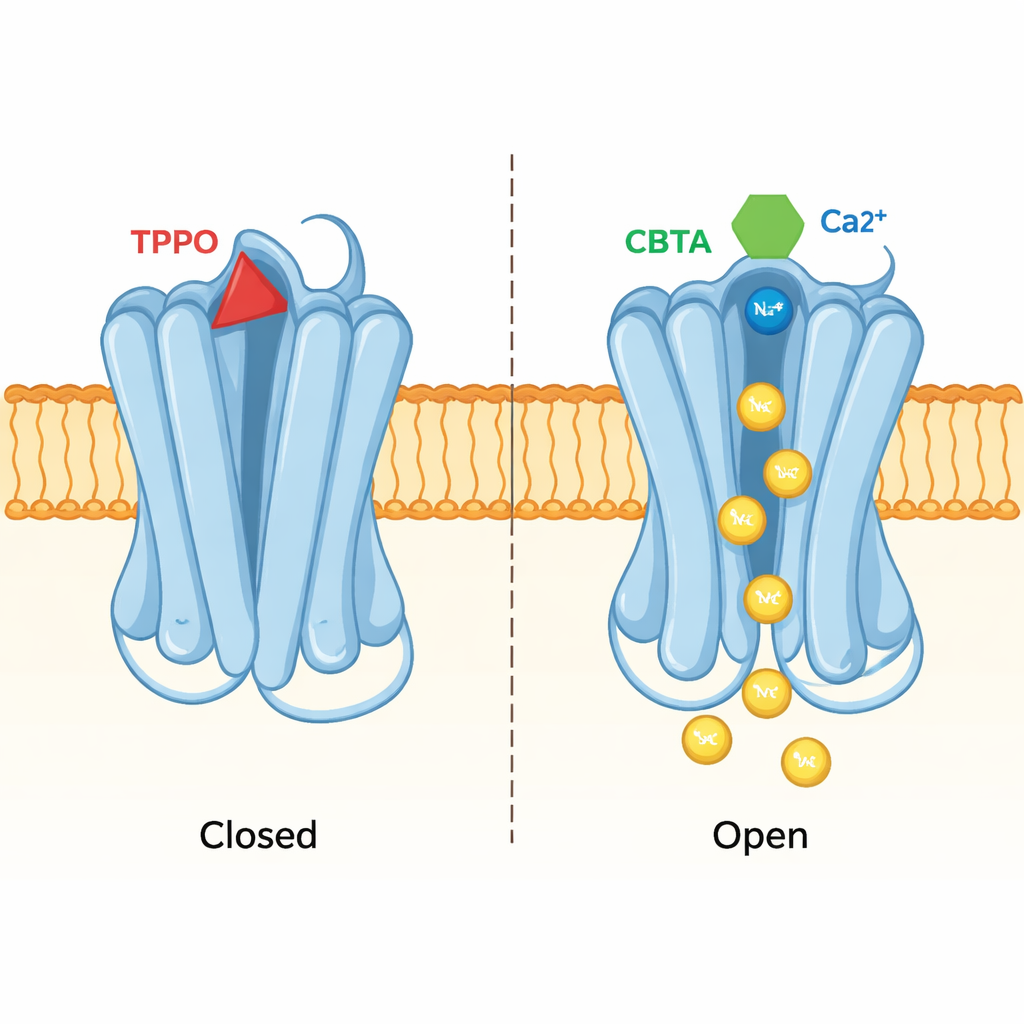
One Pocket, Two Opposite Outcomes
The same structural approach uncovered how an inhibitor called TPPO uses the exact same pocket to produce the opposite effect. When TPPO binds, both calcium sites on TRPM5 remain filled, but the central pore of the channel stays pinched shut and no ions pass through. Comparing TPPO-bound and CBTA-bound structures showed that their different shapes push on nearby protein segments in opposite directions. CBTA encourages movements that spread to the pore and open it; TPPO instead disrupts the communication between the pocket and the pore, keeping the gate closed. In essence, this one pocket can act either as an accelerator or a brake, depending on which molecule occupies it.
Rerouting the Channel’s Internal Wiring
The study also shows that this control pocket can take over jobs normally handled by a separate calcium-binding region inside the cell. In mutant channels where the usual internal calcium site is disabled, calcium alone could not open TRPM5. Yet CBTA still switched these mutants on and reshaped a key structural element that links the inner and outer parts of the channel. In other mutants that break the connection between calcium binding and pore opening, adding CBTA restored normal behavior. These results reveal that the newly identified site can reroute or repair communication between different channel domains, acting as a flexible hub for long-range signaling within the protein.
Why This Matters for Future Medicines
For non-specialists, the central message is that the researchers have found a single, drug-sensitive pocket on TRPM5 that can integrate activation, fine-tuning and shutoff. A small activator like CBTA can both mimic calcium and greatly boost the channel’s sensitivity, while another compound, TPPO, can clamp the same channel closed, all by docking in the same place. This unified picture of how TRPM5 is controlled opens the door to designing tailored molecules that either enhance or dampen its activity in specific tissues, with potential applications ranging from improving taste-based therapies and gut motility drugs to developing new strategies for managing blood sugar and metabolic disease.
Citation: Ruan, Z., Lee, J., Li, Y. et al. A single allosteric site merges activation, modulation and inhibition in TRPM5. Nat Chem Biol 22, 402–410 (2026). https://doi.org/10.1038/s41589-025-02097-7
Keywords: TRPM5 channel, taste perception, insulin secretion, ion channel gating, allosteric modulation