Clear Sky Science · en
Genome-wide association analyses of autoimmune hypothyroidism reveal autoimmune and thyroid-specific contributions and an inverse relationship with cancer risk
Why this matters to everyday health
Many people feel tired, cold, or mentally foggy without realizing that their thyroid gland may be to blame. Autoimmune hypothyroidism, in which the body’s own defenses slowly attack the thyroid, affects more than one in twenty people. This study uses genetic data from hundreds of thousands of volunteers to ask two big questions: which inherited changes in our DNA make this condition more likely, and how are these same changes linked to the risk of developing cancer? The answers reveal a surprising trade-off between an immune system that sometimes harms the thyroid but can also offer extra protection against tumors.
Looking across the whole genome
The researchers combined health records and genetic information from two large population projects, FinnGen in Finland and the UK Biobank in the United Kingdom. They focused on people who needed long-term thyroid hormone replacement and carefully excluded those whose thyroid problems were due to surgery, cancer, or other non-autoimmune causes. This produced over 81,000 cases of autoimmune hypothyroidism and more than 700,000 controls, making it the largest study of this condition so far. Scanning millions of DNA markers across the genome, they uncovered 418 independent genetic signals outside the major immune gene cluster, spread across at least 280 regions of the genome. Many of these signals involved rare or low-frequency DNA changes that alter the structure of proteins, offering direct clues to the underlying biology.
Separating general immune effects from thyroid-specific ones
Autoimmune hypothyroidism sits at the crossroads of general autoimmunity and the unique biology of the thyroid gland. To tease these components apart, the team compared their results with genetic studies of other autoimmune diseases and with blood levels of thyroid-stimulating hormone, the key clinical marker used to detect an underactive thyroid. Using a Bayesian classification method, they grouped genetic signals into those shared with broad autoimmune diseases and those more specific to thyroid hormone regulation. They estimated that roughly 38% of the signals act through general immune pathways that influence many autoimmune conditions, while about 20% act mainly through thyroid function itself. The thyroid-focused variants tended to influence hormone levels and genes active in thyroid tissue, whereas the general immune variants were more active in T cells, the white blood cells that orchestrate immune responses.
Zooming in on a key immune switch
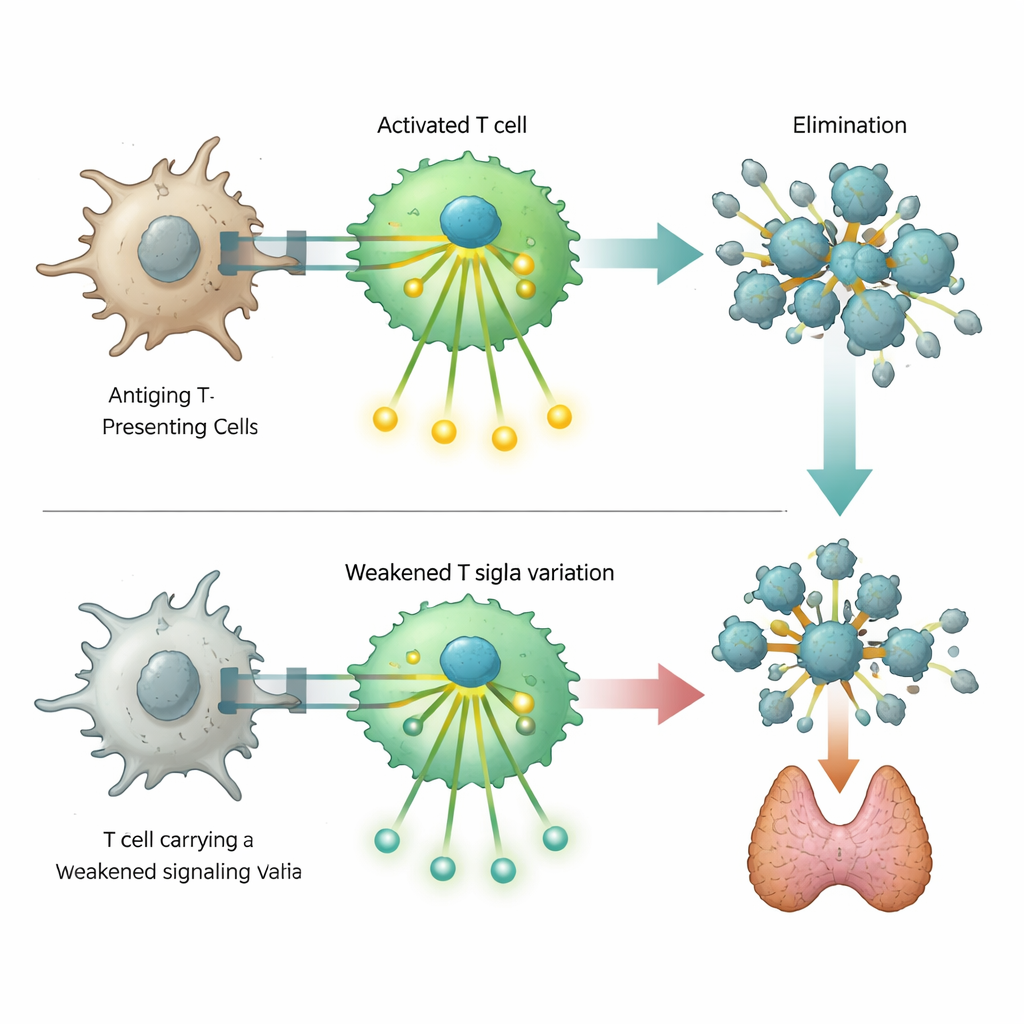
One particularly striking finding involved a rare DNA change in the gene ZAP70, which encodes a key signaling protein inside T cells. Severe defects in this protein are known to cause profound immune deficiency, but the variant identified here produces only a partial loss of function. Laboratory experiments in engineered T cells showed that this altered ZAP70 weakens, but does not completely block, the cascade of signals that normally follow recognition of a target. Cells carrying the variant were less able to switch on activation markers and downstream signaling steps. This blunted response appears to disturb the delicate balance that normally weeds out self-reactive T cells, thereby predisposing carriers to autoimmune disease while also modestly increasing their risk of certain immune deficiencies.
An unexpected link to cancer protection
Because the same immune pathways that drive autoimmunity can also attack tumors, the authors next asked how autoimmune hypothyroidism genetics relates to cancer risk. They calculated a polygenic risk score summarizing each person’s inherited tendency toward hypothyroidism and tested its association with many diseases in the Finnish data. As expected, a higher score correlated with a higher risk of various autoimmune conditions. More surprising was a consistent pattern of lower risk for several cancers, especially basal cell carcinoma and other skin cancers, but also breast and prostate cancer and an overall “all cancer” grouping. When the team looked directly at cancer genome-wide scans, about 10% of the hypothyroidism-linked sites also influenced skin cancer, with the same genetic variant generally increasing thyroid autoimmunity while decreasing skin cancer risk. These shared variants were concentrated in immune-related genes, including well-known drug targets used in checkpoint immunotherapy.
What this means for patients and medicine
Seen through a lay lens, this work shows that some people are born with immune systems tuned slightly “hotter,” making them more prone to slow, often silent damage to the thyroid gland but at the same time better equipped to spot and eliminate emerging cancers. The study disentangles which genetic changes act through general immune overactivity and which specifically affect thyroid hormone production, explaining why conditions like Hashimoto’s disease and Graves’ disease can share some risk genes yet push thyroid function in opposite directions. It also helps explain why patients who develop thyroid problems while receiving immune-boosting cancer drugs often respond better to those treatments. Overall, the findings suggest that common, natural variation in immune checkpoint and signaling genes shapes both autoimmune thyroid disease and lifetime cancer risk, offering new leads for prevention and more personalized use of immunotherapies.
Citation: Reeve, M.P., Kanai, M., Graham, D.B. et al. Genome-wide association analyses of autoimmune hypothyroidism reveal autoimmune and thyroid-specific contributions and an inverse relationship with cancer risk. Nat Genet 58, 550–559 (2026). https://doi.org/10.1038/s41588-026-02521-1
Keywords: autoimmune hypothyroidism, thyroid disease, genetic risk, immune system, cancer protection