Clear Sky Science · en
A universal concept for melting in mantle upwellings
Why deep Earth melting matters to us
Far below our feet, hot rock in Earth’s mantle slowly rises like the flow in a giant lava lamp. This hidden motion powers volcanoes, builds new seafloor, and even delivers diamonds from the deep. Yet scientists have long puzzled over a basic question: when mantle rock first begins to melt at great depth, what does that very first melt look like, and does it follow the same rules beneath oceans, islands, and continents? This study tackles that mystery and argues that a single kind of carbon-rich melt may lie at the root of most of Earth’s volcanoes.
The first drips of deep Earth “lava”
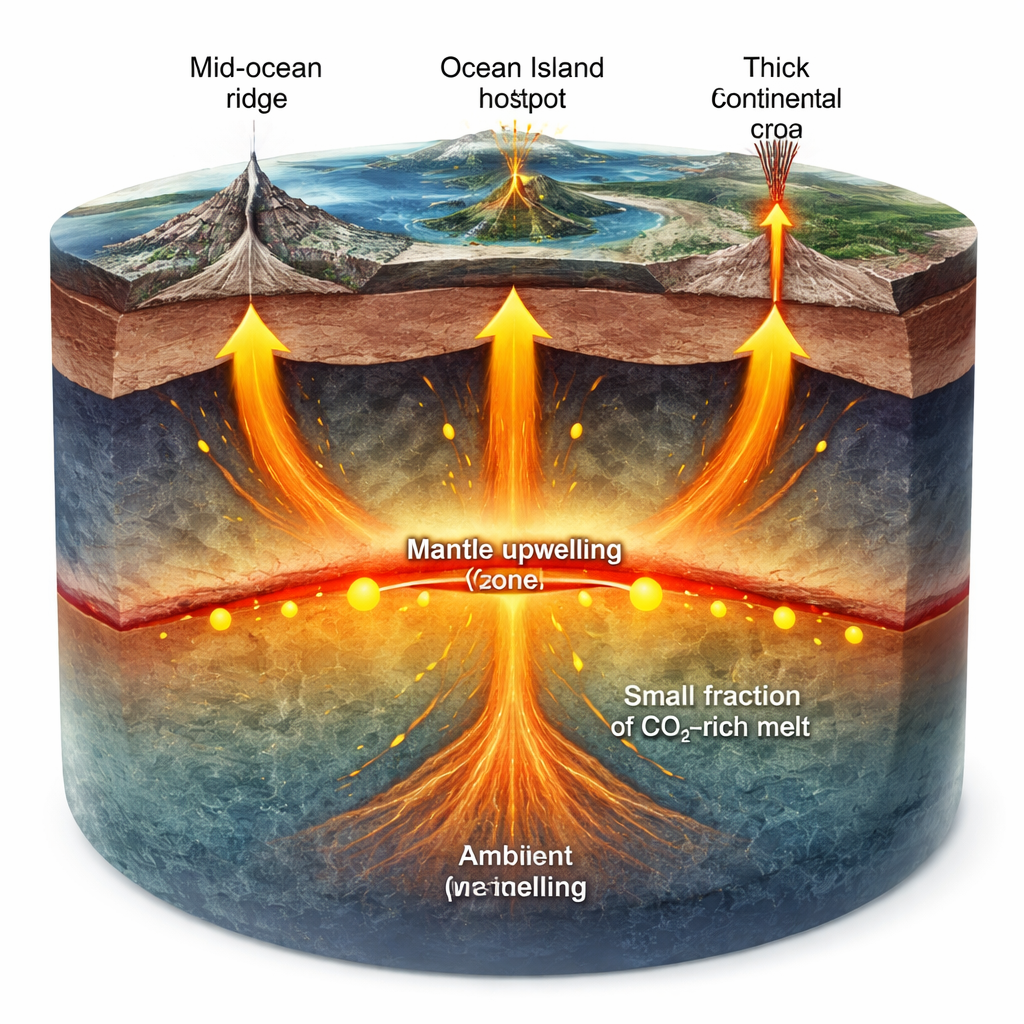
As solid mantle rises, the pressure drops, making it easier to melt. Classic models said that “dry” rock would not begin melting until it approached relatively shallow depths of about 40–70 kilometers. But real lavas collected at the surface often contain dissolved carbon dioxide (CO2) and water, which can trigger melting deeper down. The authors focus on what happens near 230–250 kilometers depth, where tiny amounts of metal and carbon in the mantle can react with iron-bearing minerals. In this reaction, solid carbon (as diamond or metal alloy) is oxidized to CO2, which in turn allows mantle rock to begin melting at temperatures hundreds of degrees lower than otherwise possible.
A universal starting recipe: carbon-rich kimberlite-like melt
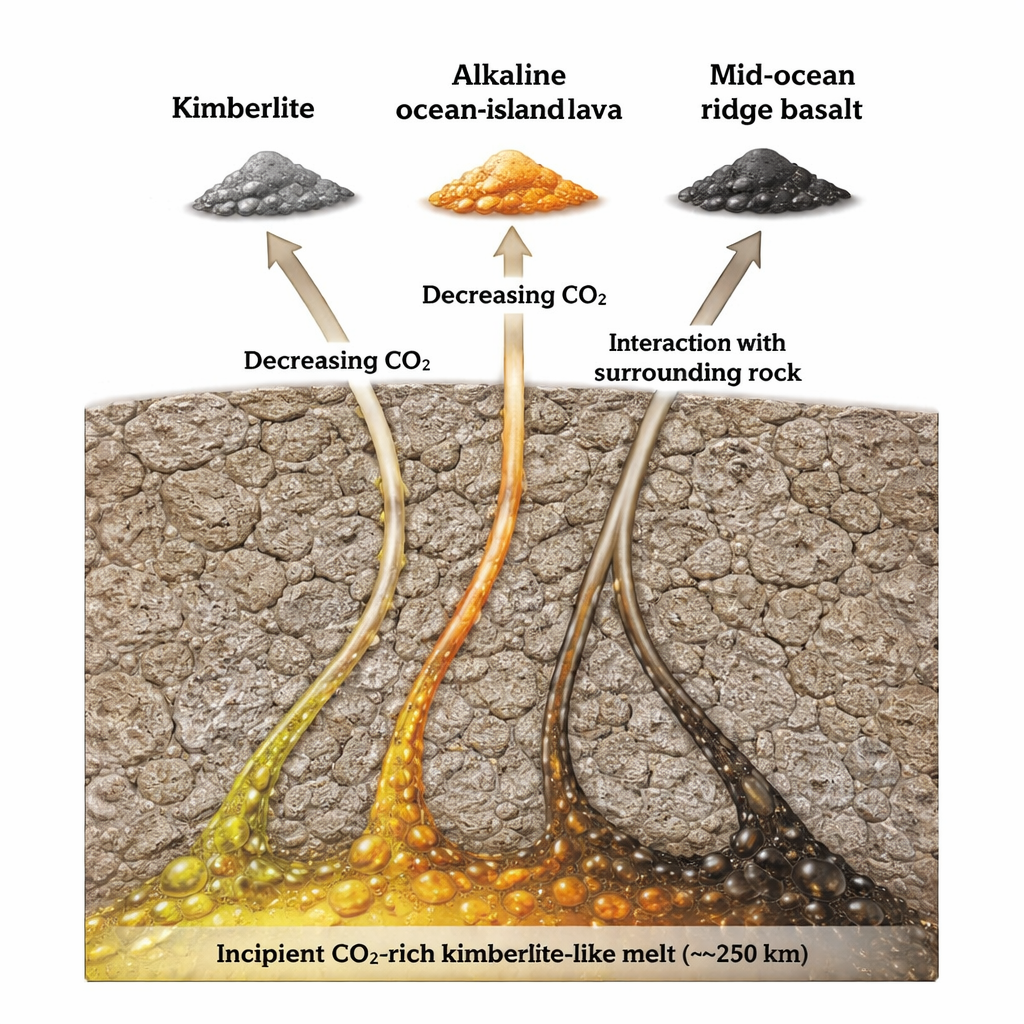
To test whether this deep “redox” melting behaves the same everywhere, the researchers carried out high-pressure experiments at about 7 gigapascals—equivalent to roughly 230 kilometers depth. They started with three very different surface lava types: kimberlites (which can carry diamonds), alkaline ocean-island basalts from hotspots, and the tholeiitic basalts that build oceanic crust at mid-ocean ridges. In the lab, they forced each of these to equilibrate with a realistic mix of mantle minerals at the right pressures and temperatures. Despite their contrasting origins, all three prototypes converged on nearly the same kind of melt: a CO2-rich, magnesium- and calcium-bearing silicate liquid with low aluminum, closely resembling natural kimberlite-like compositions. This suggests that any solid-state mantle upwelling, no matter how hot or wide, first produces broadly similar carbonated, kimberlite-style melts once it crosses the redox front.
How one melt type turns into many volcano styles
Once these first drops of carbon-rich melt form, they do not rise unchanged. The melts percolate upward through the surrounding peridotite rock, dissolving some minerals and losing some of their CO2 as pressure decreases. This process, called reactive porous flow, steadily increases the total amount of melt and pushes its composition toward higher silica and lower volatile content. Under very thick, old continental roots, the melt may be tapped near its birthplace, erupting as classic kimberlites rich in CO2 and incompatible elements. Beneath ocean islands with moderately thick lithosphere, the same incipient melt can evolve into strongly alkaline, silica-undersaturated lavas. Where the overlying plate is thin and melting continues to shallow levels, the original kimberlite-like signature becomes almost completely overprinted by larger volumes of drier, silica-richer basalt typical of mid-ocean ridges.
Clues from trace elements and seismic waves
Chemical fingerprints in lavas support this shared ancestry. Isotopes of elements such as strontium, neodymium, hafnium, and lead show that kimberlites, ocean-island basalts, and mid-ocean ridge basalts all tap similar deep mantle reservoirs, just at different degrees of melting and mixing. Trace element patterns can be explained by starting from very small melt fractions (as in kimberlites) and increasing the amount of melting toward the higher values seen beneath ridges. Seismology adds an independent line of evidence: a global low-velocity zone, usually interpreted as containing a small amount of melt, sits near 200–250 kilometers depth under ocean basins. This depth range lines up with the redox front where carbon-induced melting should begin, hinting that the same process operates worldwide.
A simple big picture beneath complex volcanoes
For non-specialists, the key message is that Earth’s most diverse lava types—from diamond-bearing kimberlites to island chains like Hawaii and the basalt that floors our oceans—may all start from essentially the same kind of deep, carbon-rich melt. The differences we see at the surface come mainly from how far those melts travel, how much they grow on the way up, and how thick the overlying tectonic plate is. In this view, carbon in the deep mantle is not just a minor ingredient: it is the switch that turns solid upwellings into melt-bearing plumes, providing a unified, planet-wide framework for how melting begins in Earth’s interior.
Citation: Schmidt, M.W., Paneva, N. & Giuliani, A. A universal concept for melting in mantle upwellings. Nature 650, 903–908 (2026). https://doi.org/10.1038/s41586-025-10065-3
Keywords: mantle melting, kimberlite, carbon dioxide in the mantle, ocean island basalts, mid-ocean ridge basalts