Clear Sky Science · en
GlycoRNA complexed with heparan sulfate regulates VEGF-A signalling
How cells fine-tune the body’s blood vessel signals
Blood vessels don’t just spring up wherever growth signals appear—they must be precisely controlled to nourish tissues without feeding tumors or causing blindness. This paper uncovers an unexpected "brake" on a major vessel-growing signal called VEGF-A. The authors show that small bits of RNA, decorating the outside of cells in partnership with sugars and proteins, can dial down VEGF-A’s message, reshaping how we think about the language cells use at their surfaces.
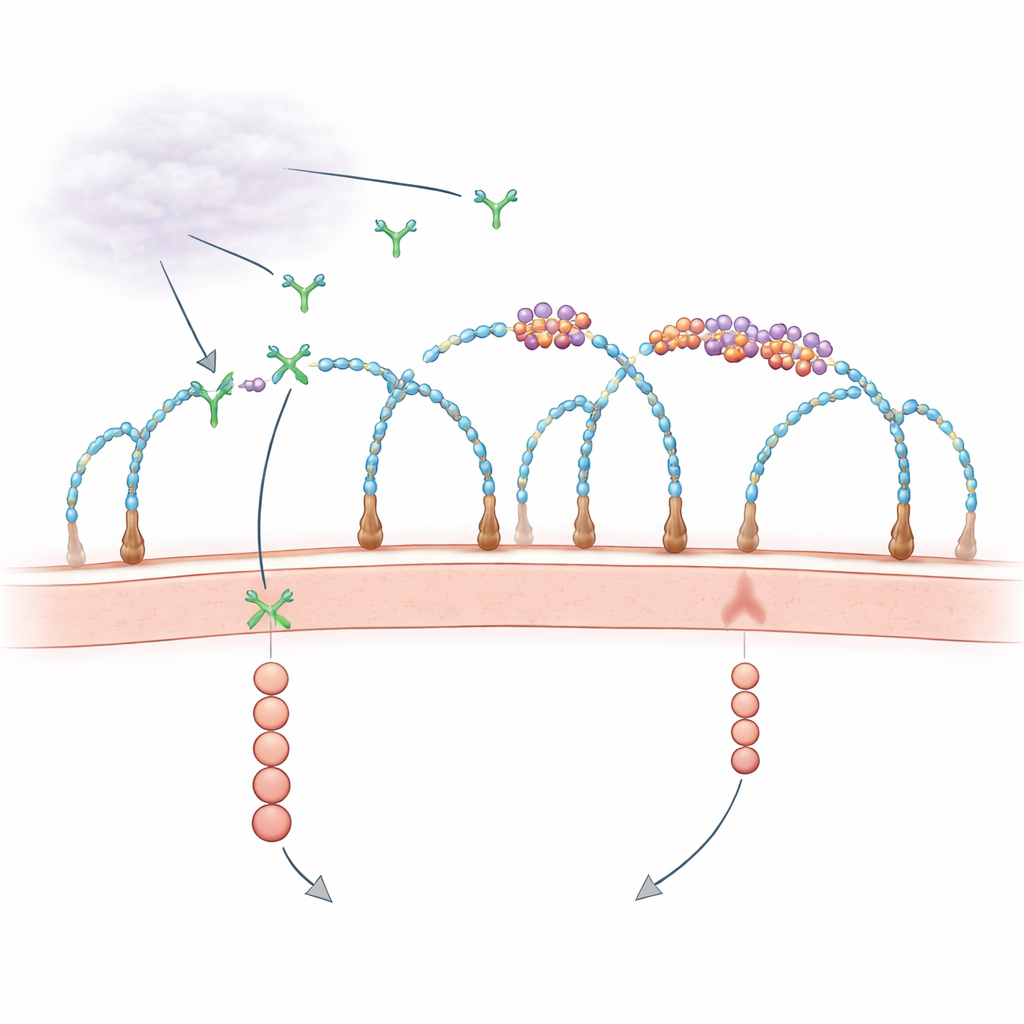
A surprising role for RNA on the cell surface
For decades, biologists have known that the cell’s outer coat, rich in sugar-decorated proteins called heparan sulfate proteoglycans, helps capture growth factors and present them to their receptors. At the same time, scattered observations hinted that RNA fragments outside cells might influence blood vessel growth, but the players and mechanisms were unclear. Recent work revealed that some small RNAs are chemically linked to complex sugars to form “glycoRNAs,” and that certain RNA-binding proteins also appear on the cell surface, clustering together with these RNAs. The new study asks a pointed question: do these tiny RNA–protein islands at the membrane actually control how growth signals like VEGF-A are received?
Building clustered RNA–protein islands with sugar chains
Using genome-wide CRISPR knockout screens and high-resolution microscopy, the researchers discovered that specific sugar chains—heparan sulfate with particular sulfate decorations—are essential scaffolds for assembling these RNA–protein clusters, which they call cell-surface ribonucleoproteins (csRNPs). When key enzymes that build or sulfate heparan sulfate were removed, the clusters of glycoRNA and their partner proteins vanished from the cell surface, even though the RNAs and proteins were still present inside the cells. Enzymes that clip heparan sulfate chains off the surface or chemically block sulfation had the same effect. These results show that intact, properly sulfated heparan sulfate chains organize csRNPs into nanoscale islands at the membrane.
RNA clusters act as brakes on a powerful vessel-growth signal
The team then turned to human endothelial cells, which line blood vessels and respond to VEGF-A. They found that these cells also display heparan sulfate–anchored csRNP clusters. Treating live cells with RNases, enzymes that degrade RNA, stripped away the RNA component without disturbing the underlying sugar chains. Under those conditions, VEGF-A’s most common, heparan-binding form (VEGF-A165) triggered much stronger activation of the downstream ERK signalling pathway, while a shorter form lacking the heparan-binding tail (VEGF-A121) was unaffected. More VEGF-A165 molecules bound the cell surface after RNA removal, yet receptor levels stayed the same. In 3D microfluidic devices, endothelial cells exposed to RNase grew farther into collagen gels and formed tube-like structures more robustly, indicating that destroying surface RNA unleashes pro-angiogenic behaviour.
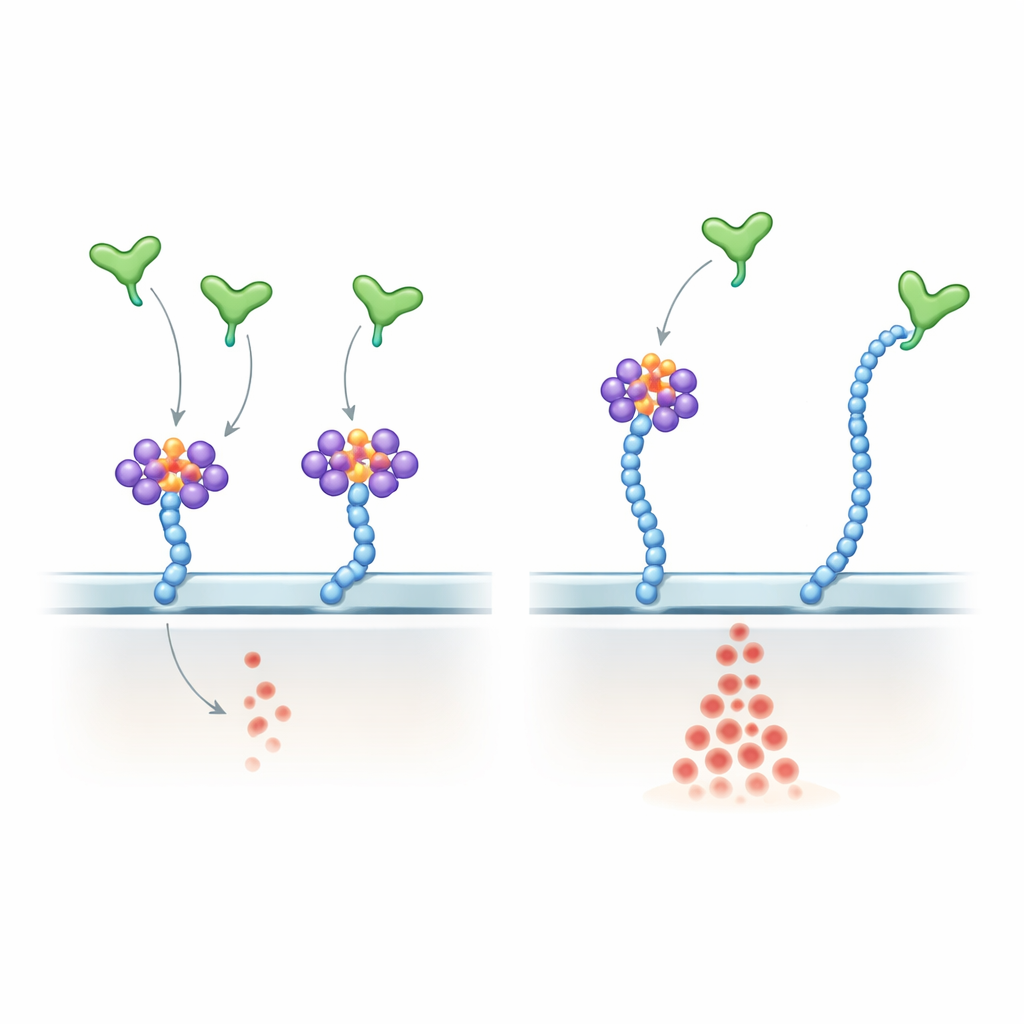
Dissecting how VEGF-A senses RNA versus sugar
To probe mechanism, the authors showed that VEGF-A165 directly binds selected small RNAs, including many known glycoRNAs, through its positively charged C‑terminal region—the same region that binds heparan sulfate. They engineered a subtle version of VEGF-A165 in which key arginine residues in this tail were swapped for lysines. This mutant kept its overall positive charge and still grabbed heparan sulfate chains, but it bound glycoRNA poorly and became largely insensitive to RNase treatment. In endothelial cells, the mutant produced stronger and more RNase-resistant ERK activation than normal VEGF-A165, closely mimicking the effect of removing RNA from the cell surface. In living mice, injecting the mutant into the eye caused more exuberant retinal vascular growth than the normal protein. In zebrafish embryos, overexpressing the mutant version of Vegfa disrupted normal vessel patterning and increased endothelial cell numbers.
From basic mechanism to broader implications
Finally, the authors showed that fusing VEGF-A’s heparan-binding tail to an unrelated signal protein, Wnt3a, could redirect Wnt activity: attaching the normal tail reduced Wnt-driven developmental changes, whereas attaching the RNA-insensitive tail boosted them. Together, these experiments support a model in which csRNP clusters, anchored by specifically sulfated heparan sulfate, bind VEGF-A and related factors to temper their activity. When the RNA component is removed or when growth factors lose their ability to sense RNA, the balance tips toward stronger signalling and altered vascular development in multiple species.
Why this matters for health and future therapies
To a non-specialist, the key message is that cells use not only proteins and sugars, but also small RNAs displayed on their surface to fine-tune powerful growth cues like VEGF-A. These sugar-tethered RNA–protein clusters act as adjustable brakes on blood vessel formation, helping ensure that new vessels sprout where and when they are needed. Disrupting this balance—by degrading surface RNA or altering growth factors’ RNA-binding properties—can either exaggerate or misdirect vessel growth. Understanding this newly revealed control layer could open the door to therapies that more precisely dial angiogenesis up or down in cancer, eye disease, wound healing, and regenerative medicine by targeting surface RNAs, their sugar scaffolds, or the RNA-sensing regions of growth factors.
Citation: Chai, P., Kheiri, S., Kuo, A. et al. GlycoRNA complexed with heparan sulfate regulates VEGF-A signalling. Nature 651, 808–818 (2026). https://doi.org/10.1038/s41586-025-10052-8
Keywords: angiogenesis, vascular endothelial growth factor, heparan sulfate, cell surface RNA, glycoRNA