Clear Sky Science · en
Tumour–brain crosstalk restrains cancer immunity via a sensory–sympathetic axis
How Nerves Can Help Tumors Hide
Cancer is usually framed as a battle between rogue cells and the immune system, but this study reveals a surprising third player: the nervous system. Researchers show that lung tumors can tap into a sensory–sympathetic nerve circuit linking the lungs and the brain, and then use that circuit to weaken the body’s natural anti-cancer defenses. Understanding this hidden “wire” between tumors and the brain could open up new ways to treat lung cancer by targeting nerves and their signals, not just the cancer cells themselves. 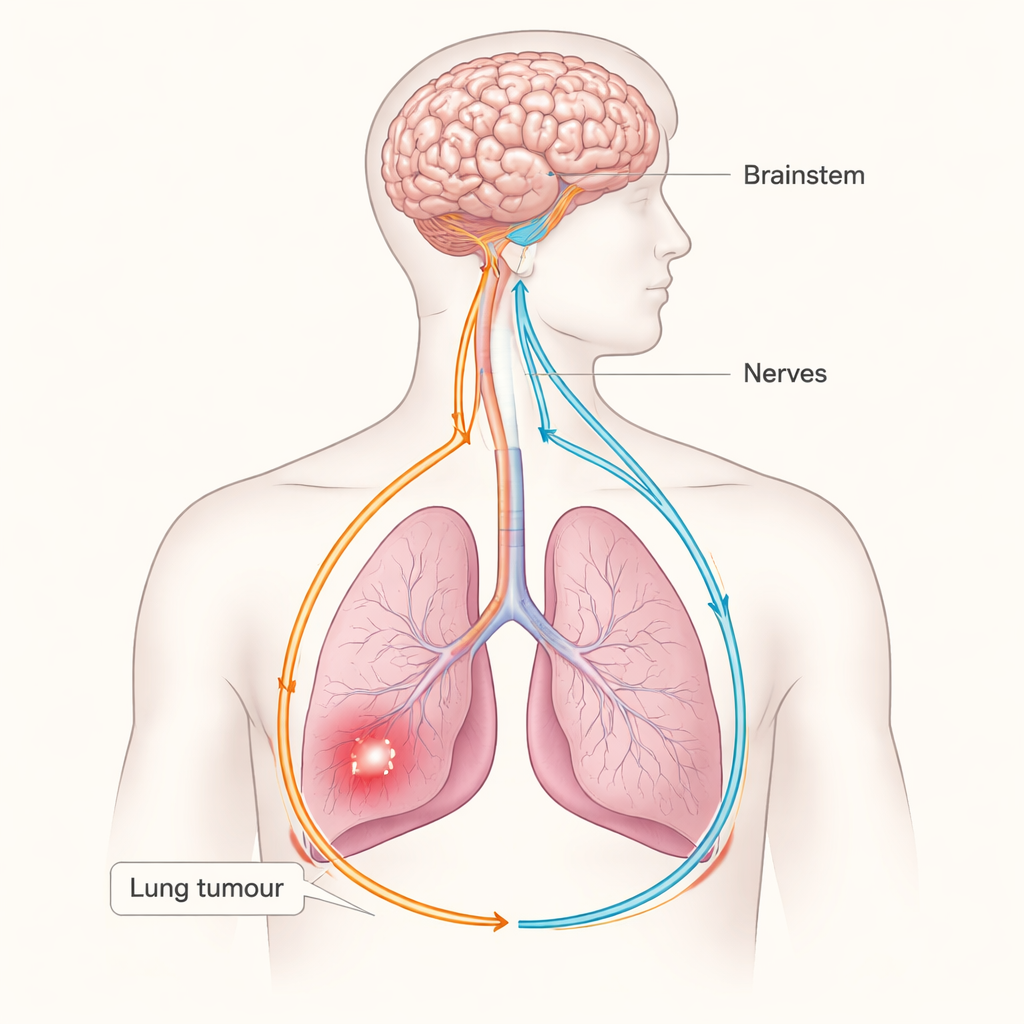
Nerves Growing Into the Tumor
The team studied lung adenocarcinoma, a common form of lung cancer, in sophisticated mouse models that closely mimic human disease. Using whole-organ 3D imaging and genetic labeling, they found that tumors in the lungs are heavily wired by nerve fibers. These nerves are not random: many come from a specific group of sensory cells in the vagus nerve, which normally carries information about organ health to the brain. Tumor cells were seen to secrete growth-promoting proteins, such as nerve growth factor, that encourage these vagal sensory fibers to sprout and invade the tumor. At the same time, the sensory neurons themselves changed their gene activity, suggesting that the tumor was reprogramming them to respond differently than they would in healthy lung tissue.
A One-Way Boost for Tumor-Friendly Signals
Not all vagus-linked sensory neurons were involved. The researchers pinpointed a subtype marked by two molecules, NPY2R and TRPV1, as the main players. These neurons were abundant inside tumor regions but largely absent from nearby normal lung. When the scientists selectively removed or silenced this NPY2R/TRPV1 group—using genetic tricks, targeted toxins, or designer receptors they could switch off with a drug—lung tumors shrank dramatically and mice lived longer, with better weight and behavior. By contrast, removing a different sensory subgroup that innervates other lung structures did not slow tumor growth, underscoring that only a specific sensory “channel” is wired into cancer control. 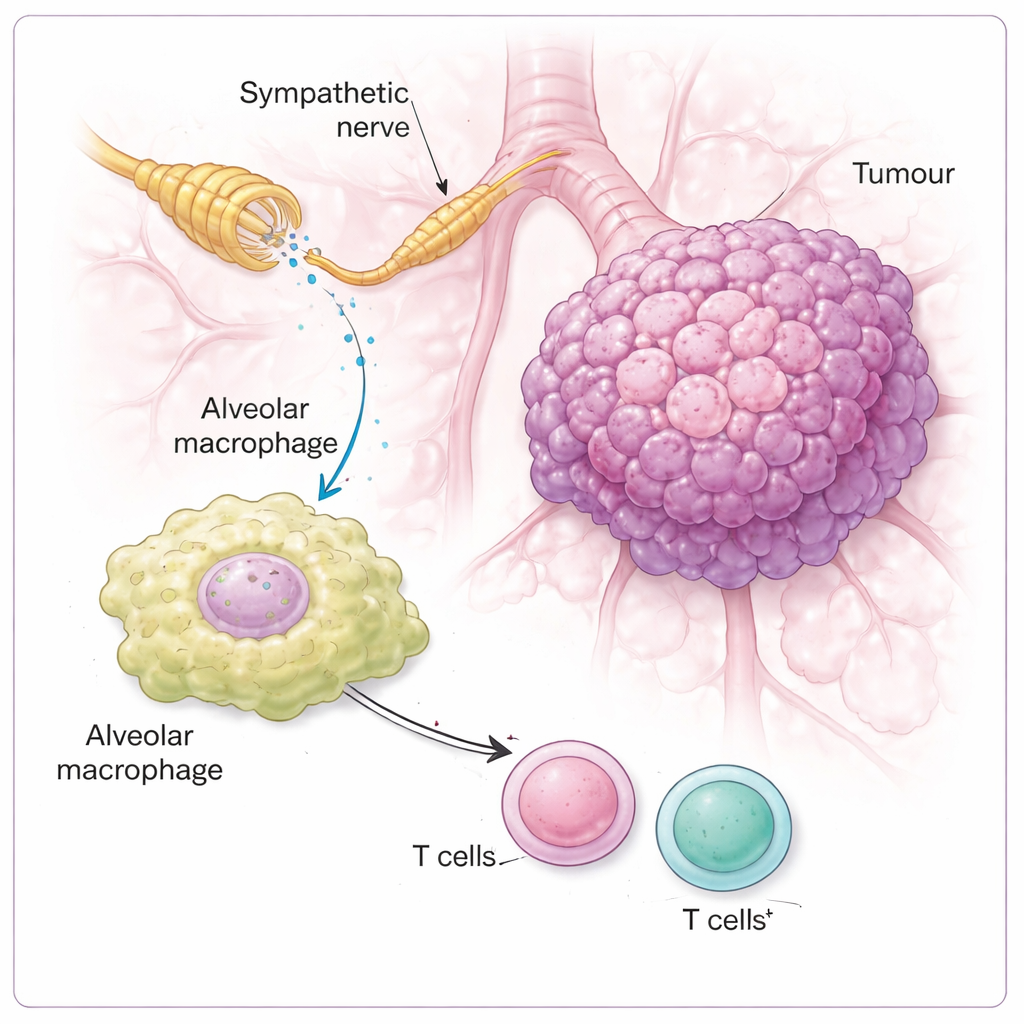
From Brainstem to Stress-Like Signals in the Lung
To see how these sensory signals are handled by the brain, the team tracked the circuit upstream. Activity markers showed that when lung tumors were present, brainstem regions that receive vagal input lit up, especially an area called the rostral ventrolateral medulla, a key hub that boosts sympathetic “fight-or-flight” output. Turning off the NPY2R/TRPV1 sensory neurons quieted this brainstem activity and reduced the number of sympathetic nerve fibers and levels of the stress messenger noradrenaline in the tumor area. Silencing the brainstem hub itself also slowed tumor growth. Together, these results outline a closed loop: the tumor stimulates vagal sensory nerves, the brainstem responds by increasing sympathetic outflow, and sympathetic nerves feed back onto the lung environment in a way that favors the cancer.
Switching Off Immune Cells Through Local Middlemen
The study then asked which cells in the tumor neighborhood actually “hear” the sympathetic signals. Surprisingly, noradrenaline had little direct effect on tumor cell growth in a dish. Instead, the key responders were immune cells, especially alveolar macrophages—sentinel cells that sit in the air sacs of the lung. These macrophages carried high levels of a receptor called β2-adrenergic receptor, which senses noradrenaline. When this receptor was missing, or when noradrenaline signaling was blocked, tumors grew less and immune attack by T cells became stronger. In mice with an intact nerve circuit, noradrenaline pushed macrophages into a more suppressive state, marked by higher levels of an enzyme called ARG1, which is known to dampen T cells. Once macrophages were reprogrammed in this way, nearby CD4 and CD8 T cells—normally the body’s front-line cancer killers—were less able to mount a robust response.
Why This Matters for Treating Cancer
Put simply, the authors show that lung tumors can “talk” to the brain through the vagus nerve and receive a harmful reply through sympathetic nerves that tell local macrophages to calm T cells down. Breaking any major link in this loop—sensory input, brainstem relay, sympathetic output, or β2-adrenergic signaling in macrophages—was enough to restore stronger anti-tumor immunity and slow cancer in mice. Human lung cancer data further suggested that patients whose tumors show higher signatures of these nerve pathways tend to fare worse and have fewer killer T cells in their tumors. The work raises the possibility that drugs already used for heart disease and anxiety, such as beta-blockers, or future therapies aimed at specific nerve circuits, could be repurposed or refined to “cut the wires” that tumors use to escape immune attack.
Citation: Wei, H.K., Yu, C.D., Hu, B. et al. Tumour–brain crosstalk restrains cancer immunity via a sensory–sympathetic axis. Nature 650, 1007–1016 (2026). https://doi.org/10.1038/s41586-025-10028-8
Keywords: cancer neuroscience, lung cancer immunity, vagus nerve, sympathetic nervous system, tumour microenvironment