Clear Sky Science · en
Population-scale sequencing resolves determinants of persistent EBV DNA
Why a common virus still matters
Most adults carry Epstein–Barr virus (EBV), often without ever knowing it. For some, though, this quiet passenger is linked to illnesses ranging from mono to cancers and autoimmune diseases. This study asks a simple but far-reaching question: can we use the massive troves of DNA data already collected from the public to see who carries more EBV in their blood, and whether their genes help determine who stays in control of the virus and who does not?
Reading a hidden viral footprint in our DNA
Modern biobanks, such as the UK Biobank and the U.S. All of Us program, have sequenced the complete DNA of hundreds of thousands of volunteers. These projects focused on human genes, but the raw data also contain stray pieces of DNA from viruses. The researchers realized that the reference human genome used for alignment includes an extra "chromosome" for EBV. By re-examining reads that mapped to this EBV segment, they could estimate how many copies of EBV’s genome were present in each person’s blood at the time of sampling. After carefully masking a few misleading repetitive regions, they defined people with clearly detectable levels as having "EBV DNAemia"—a measurable amount of EBV DNA circulating in blood.
A small minority carry a high viral load
Looking at nearly half a million people in the UK Biobank and a quarter of a million in All of Us, the team found that most adults had no detectable EBV DNA in blood, even though over 90% had evidence of past infection from antibody tests. Roughly 10% of participants, however, fell into a tail of the distribution with clearly elevated EBV DNA. These individuals tended to be older, more often male, and were slightly more likely to be taking immune-suppressing medications. Similar patterns appeared in both cohorts, and in a separate set of saliva samples the virus was much more common, highlighting that EBV sits in different reservoirs in the body and that blood measurements capture only one of them.
Links between viral persistence and disease
With this new biomarker in hand, the authors asked how persistent EBV DNA in blood relates to health. They scanned thousands of diagnosis codes and lab measurements to see which were more common in people with EBV DNAemia. Strong associations emerged with conditions already suspected to be tied to EBV, including Hodgkin lymphoma, rheumatoid arthritis, lupus, chronic obstructive pulmonary disease, and certain lung cancers. There were also signals for cardiovascular problems, kidney failure, depression, and fatigue, and suggestive links to rare neurologic disorders such as neuromyelitis optica. Many of these patterns were reproduced in the independent All of Us cohort. The study cannot prove that EBV causes these problems, but it shows that high EBV DNA in blood is a marker of broader health risks and immune disturbance.
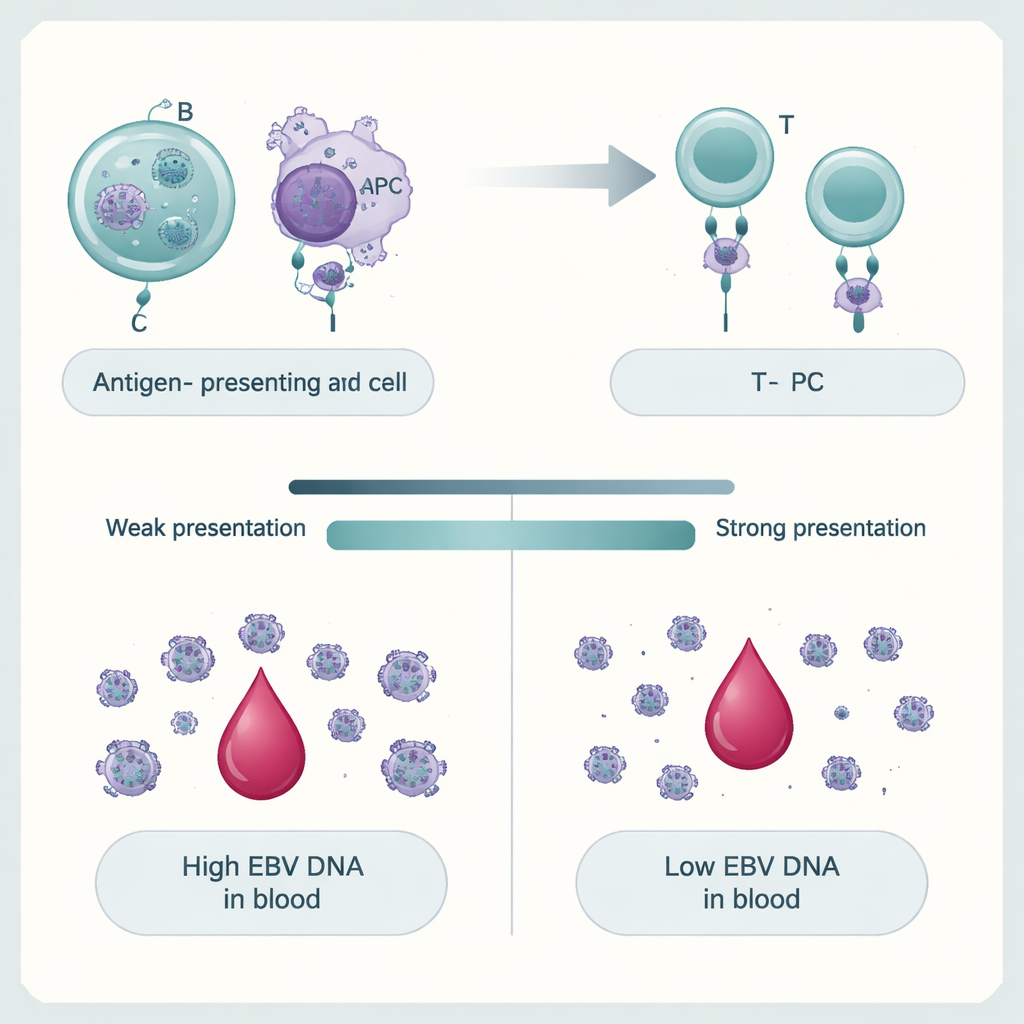
How our genes shape control of the virus
The team then asked why only some people have high EBV DNA levels. By scanning millions of genetic variants across the genome, they showed that EBV DNAemia is a polygenic trait: many small DNA differences together nudge viral levels up or down. The strongest signals clustered in and around genes of the major histocompatibility complex (MHC), which help immune cells display viral fragments to T cells. Variants that alter these proteins, particularly MHC class II molecules, were tightly linked to whether EBV DNA persisted. Genes active in B cells—the main long-term home of EBV—and other antigen-presenting cells were especially enriched. Using computer models of how different human MHC variants bind pieces of the EBV proteome, the authors found that versions predicted to present EBV fragments more strongly tended to protect against high EBV DNA, while weaker presenters were associated with persistence.
What this means for understanding infection
For non-specialists, the core message is that our long-term balance with EBV is not random. By mining existing population-scale DNA data, the study shows that a person’s genetic makeup—especially in immune genes that show viral pieces to T cells—helps determine how much EBV lingers in their blood. High levels mark a subset of people at elevated risk for a range of immune, respiratory, and possibly neurologic illnesses. The work offers a blueprint for turning old sequencing data into new insights about many viruses that share our bodies, and suggests that tailored strategies to boost or mimic effective antigen presentation could one day help keep lifelong infections like EBV better under control.
Citation: Nyeo, S.S., Cumming, E.M., Burren, O.S. et al. Population-scale sequencing resolves determinants of persistent EBV DNA. Nature 650, 664–672 (2026). https://doi.org/10.1038/s41586-025-10020-2
Keywords: Epstein–Barr virus, viral persistence, human genetics, autoimmune disease, biobank sequencing