Clear Sky Science · en
Predatory aggression evolved through adaptations to noradrenergic circuits
Why tiny predators matter
Most of us think of worms as simple, harmless creatures, but some microscopic worms are surprisingly fierce hunters. This study asks a big question with these tiny predators: how does evolution reshape the brain’s chemistry so that an animal becomes more aggressive? By dissecting the behaviour and nerve circuits of a predatory nematode, the researchers reveal how two chemical signals in the nervous system work like opposing switches that turn predatory aggression on and off.
From harmless feeder to fierce hunter
The star of this work is Pristionchus pacificus, a worm that can eat bacteria like the classic lab nematode Caenorhabditis elegans, but also attacks and kills other worms, sometimes even members of its own species. It has teeth-like structures and a powerful feeding organ that can puncture prey. Yet it does not attack every time it meets another worm, suggesting its behaviour is carefully controlled. To understand how, the authors used fluorescent markers in the feeding organ and high-speed video to track many animals at once while they roamed on either bacterial food or live prey.
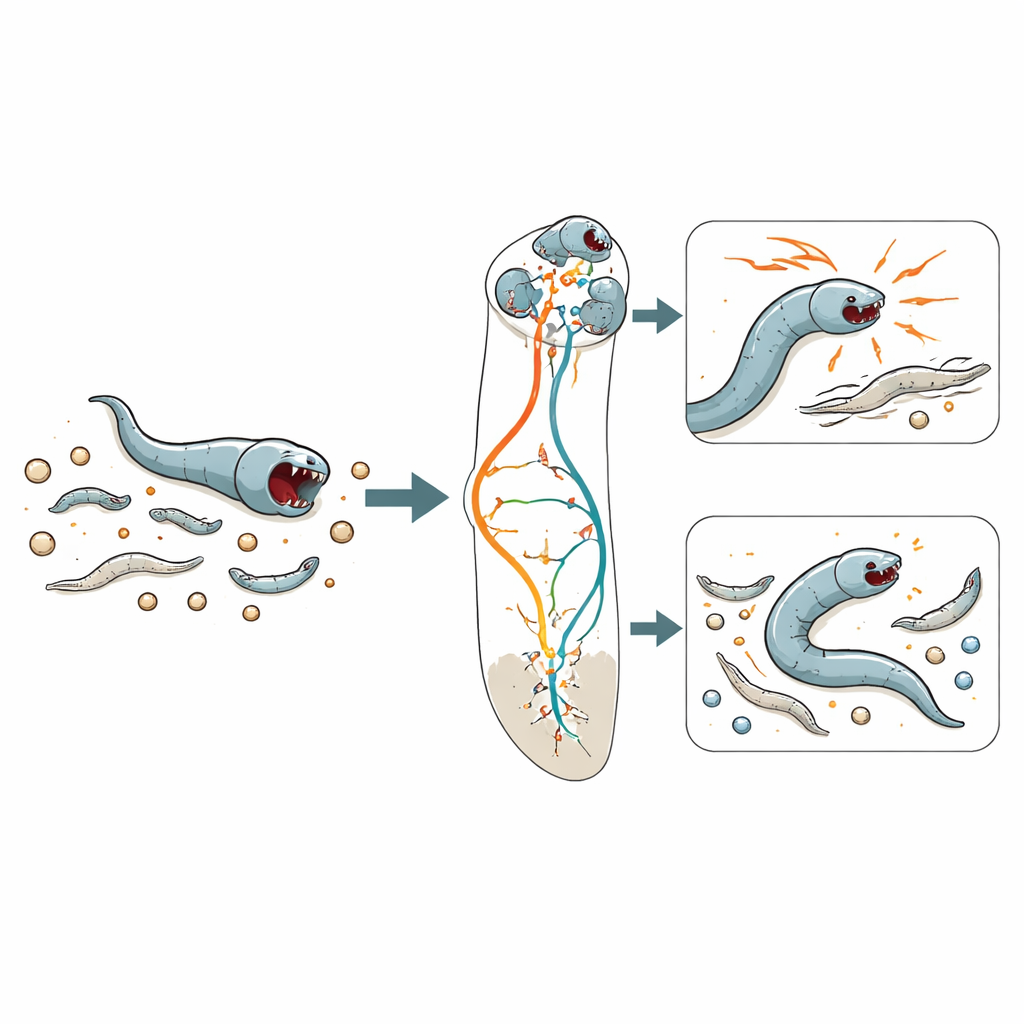
Teaching a computer to read behaviour
Instead of scoring behaviour by eye, the team trained a machine-learning system to recognize patterns in movement and feeding. They extracted features like speed, the rate of “pumping” in the feeding organ, and how much the head swings back and forth. Using modern clustering methods, the algorithm discovered six recurring “states” that worms cycle through. Some matched familiar patterns such as fast roaming and slow dwelling, known from non-predatory worms. Others were unique to situations rich in prey and were labelled predatory search, predatory biting and predatory feeding. On plates full of larvae, the worms spent much more time in these predatory states; on simple bacterial lawns, they rarely entered them. The model could predict these states in new recordings with very high accuracy, turning raw motion into a kind of behavioural weather map.
Context and the meaning of a bite
The scientists then asked when bites mean feeding and when they mean pure aggression. Using a dual-colour microscope that separately tracked predators and glowing prey, they confirmed that the “predatory biting” state coincides with nose-to-prey contact, while “predatory feeding” corresponds to swallowing fluorescent prey material. When both bacteria and larvae were available, the worms still bit other larvae just as often, but followed through with feeding less frequently. In other words, a larger fraction of bites were not about hunger at all—they were about driving away competitors from shared food, revealing an aggressive, territorial side to this tiny predator.
Chemical switches for attack and calm
Next, the authors turned to brain chemistry. They disrupted genes needed to make several signalling molecules that are chemical cousins of human noradrenaline. Two of these, octopamine and tyramine, proved crucial. When the worms could not make octopamine, they showed far fewer aggressive biting bouts and entered predatory states less often. But when both octopamine and its precursor tyramine were removed, aggression bounced back, hinting that tyramine normally pushes the animal into a calmer, non-predatory mode. Adding the pure chemicals to worms confirmed this tug-of-war: octopamine extended predatory behaviour, while tyramine promoted docile, non-hunting states. The team pinpointed specific receptors for these chemicals in head sensory neurons that sit around the mouth. Silencing one set of these sensory neurons greatly reduced predation, showing that they act as a gate: when tuned by octopamine, they help turn a simple nose touch into an attack.
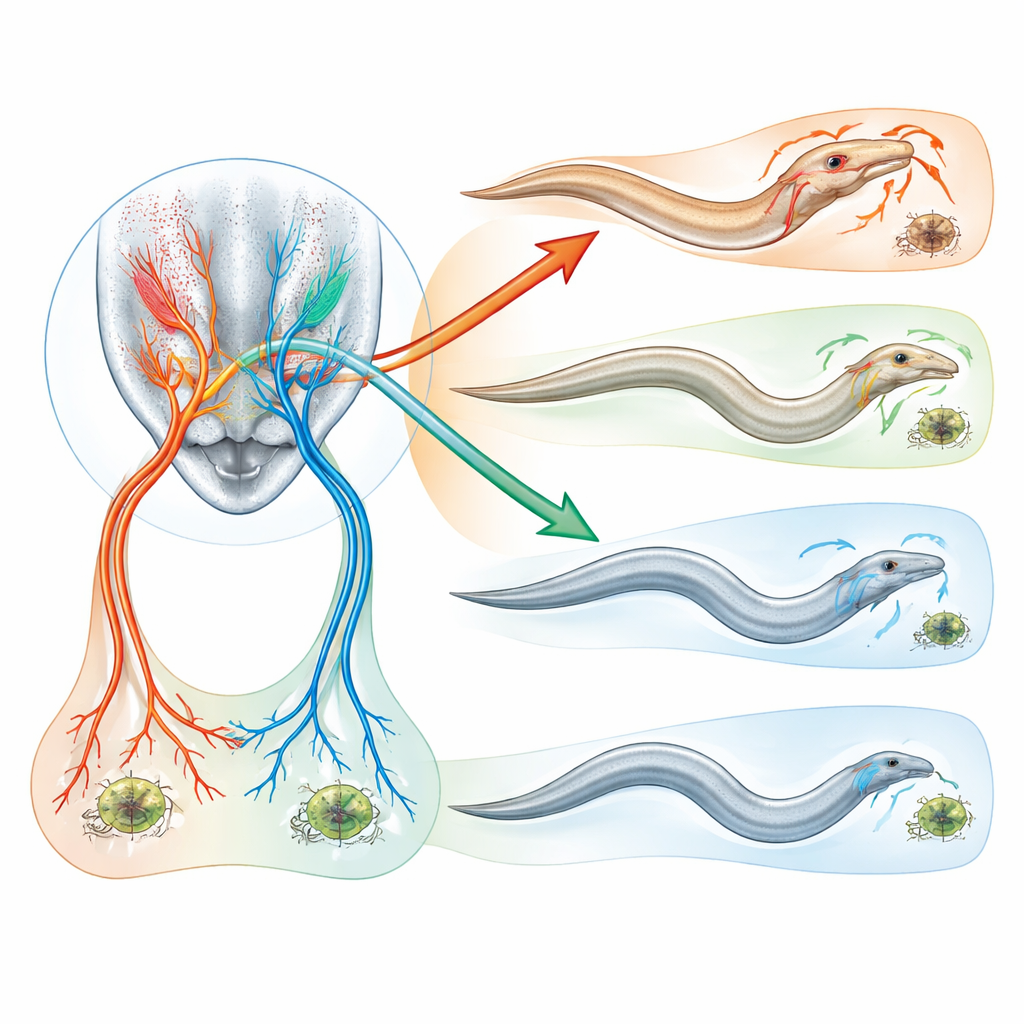
How evolution rewired tiny brains
By comparing this predatory worm with C. elegans and other relatives, the researchers found that the basic nerve cells producing octopamine and tyramine are ancient and shared. What has changed over evolution is where their receptors are placed and how their signals are interpreted. In the predatory lineage, the receptors that read these chemicals have been repositioned onto specific sensory cells in the head, linking environmental contact with a powerful aggression switch. Similar genetic changes in another toothy nematode species also reduced its tendency to kill prey, suggesting this chemical control system arose early in the group and helped enable the evolution of predation.
What this means for understanding aggression
The work paints a clear, accessible picture: in these microscopic predators, aggression is not just raw instinct but a carefully tuned state controlled by opposing brain chemicals. Octopamine acts like a “go for it” signal that primes sensory neurons to turn encounters into attacks, while tyramine provides a “stand down” signal that favours peaceful foraging. By tracing how evolution rewired this small circuit, the study offers a concrete example of how changes in neural chemistry and wiring can give rise to new, complex behaviours—insights that may echo across the animal kingdom, from worms to much larger brains.
Citation: Eren, G.G., Böger, L., Roca, M. et al. Predatory aggression evolved through adaptations to noradrenergic circuits. Nature 651, 154–163 (2026). https://doi.org/10.1038/s41586-025-10009-x
Keywords: aggression, nematode predation, neuromodulators, evolution of behavior, sensory circuits