Clear Sky Science · en
Individualized mRNA vaccines evoke durable T cell immunity in adjuvant TNBC
New hope after breast cancer treatment
For many women with an aggressive form of breast cancer called triple‑negative breast cancer, fear does not end when chemotherapy and surgery are over. This cancer has a high risk of coming back and spreading, often within just a few years. The study described here tests a highly personalized vaccine, built from each patient’s own tumor mutations, to see whether it can train the immune system to stand guard for years and lower the chance of relapse after standard treatment is finished.
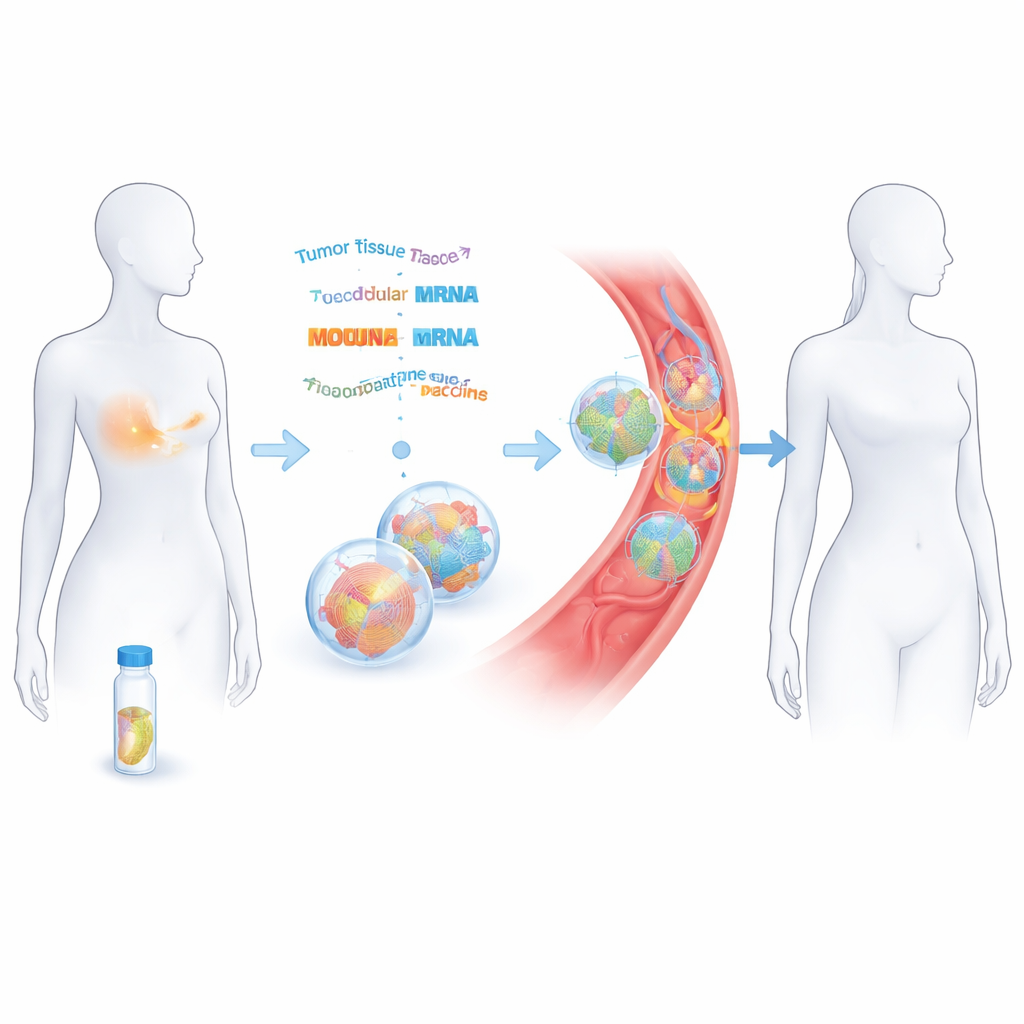
Turning a removed tumor into a custom vaccine
The researchers worked with 14 women who had early‑stage triple‑negative breast cancer and had already received surgery and chemotherapy with curative intent. From the removed tumor tissue, they read the DNA and RNA to catalog the cancer’s unique mutations. Using computer tools, they predicted which of these changes could be seen as foreign by immune cells. Up to 20 of these "neoantigens" were then stitched together and encoded into two strands of messenger RNA (mRNA), similar in principle to COVID‑19 vaccines but tailored to each patient. These mRNA strands were packaged in tiny fat bubbles and given by vein in eight doses over about two months.
How the vaccine rouses immune defenders
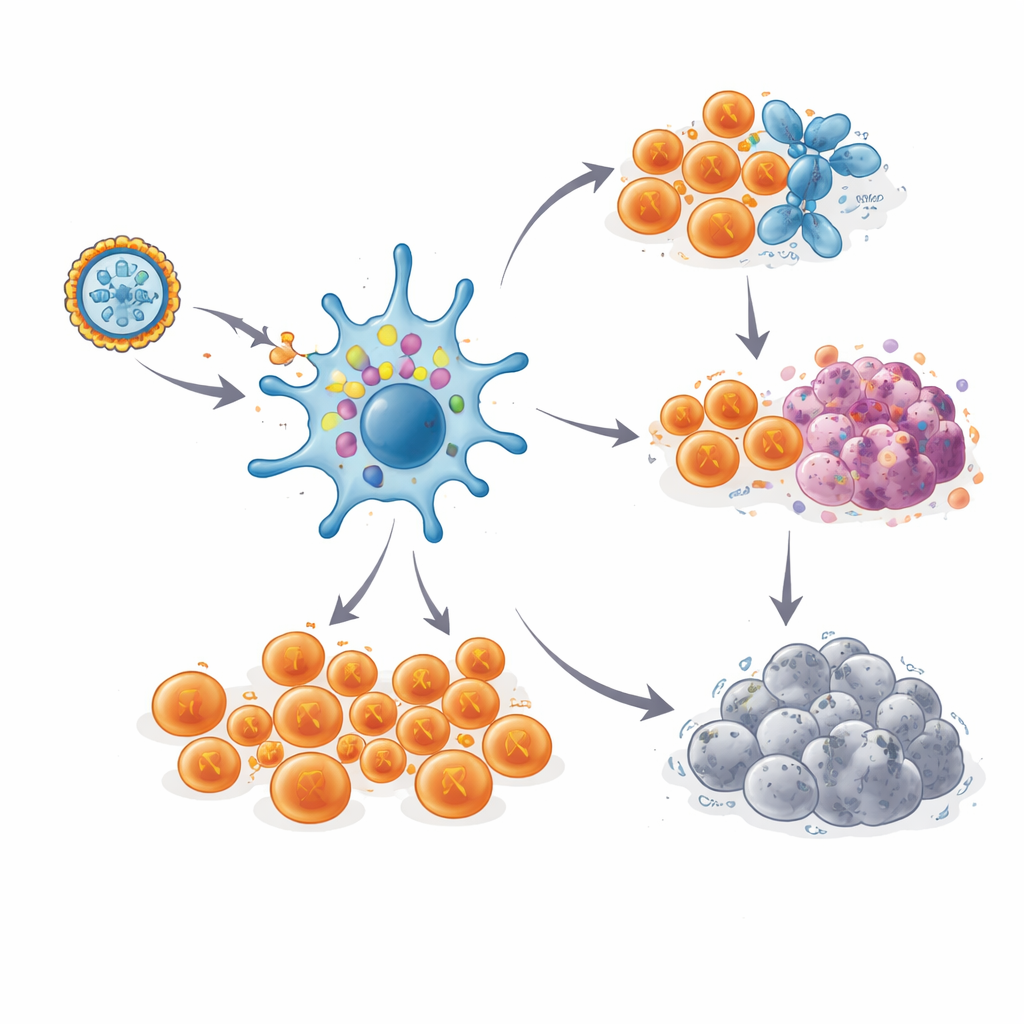
Once in the body, the mRNA instructs immune sentinels called dendritic cells to briefly make the neoantigen proteins and display fragments of them on their surface. This acts like a most‑wanted poster for T cells, the immune system’s primary cancer‑killing force. The team drew blood before vaccination and at several time points afterward to test whether neoantigen‑specific T cells appeared and how strong they were. Using sensitive lab assays, they found that every patient developed new or boosted T cell responses against at least one of her selected mutations, and most responded to several. In many cases, large fractions of circulating T cells—levels usually seen only with powerful cell therapies—were now tuned to recognize tumor‑specific targets.
Long‑lasting immune memory
Importantly, these T cell responses did not fade quickly. In most patients, strong responses peaked during the vaccination period, dipped slightly, and then remained at high levels for one to three and a half years, even without booster shots. By tracking the unique receptor "barcodes" of individual T cell clones, the scientists could follow specific neoantigen‑reactive cells for up to six years in one patient who stayed cancer‑free. Detailed single‑cell analyses showed two main fates for these cells. Many became highly armed killer T cells, poised to destroy any cell bearing the right neoantigen. Others developed into a rarer, stem‑like memory pool with markers of self‑renewal, suggesting a reservoir capable of regenerating fresh waves of fighters if cancer cells reappeared.
What happened to the patients
After a median of just over five years of follow‑up from the first vaccine dose, eleven of the fourteen women had not experienced a return of their cancer; one of these died from unrelated causes while still in remission. Three women did relapse, and their cases offer clues to why a personalized vaccine may sometimes fail. One patient mounted only a weak T cell response and later benefited from a different immunotherapy drug, an anti‑PD‑1 antibody, before eventually succumbing to widespread disease. A second patient had hereditary risk and tumors in both breasts; only one tumor was used to design the vaccine, and evidence later showed the recurrence came from the genetically distinct, unvaccinated tumor. The third patient had strong vaccine‑induced T cells that infiltrated the recurrent tumor, but the cancer cells had largely lost key molecules needed to display neoantigens on their surface, effectively hiding from immune attack.
Why this work matters for the future
This early‑phase study was small and had no control group, so it cannot prove that the vaccine alone prevented relapses. Nonetheless, it shows that building a complex, custom mRNA vaccine from a patient’s own tumor is feasible in routine hospital settings, is generally well tolerated, and can trigger powerful, long‑lived T cell armies against multiple cancer‑specific targets. The findings also highlight escape routes that tumors may use, such as losing their antigen display machinery or arising from untargeted lesions, pointing toward combinations with other immune therapies and more extensive tumor sequencing. For patients facing triple‑negative breast cancer, this work suggests that a bespoke vaccine, given after standard treatment, could one day help keep the disease from returning by turning their own immune system into a durable, highly specific line of defense.
Citation: Sahin, U., Schmidt, M., Derhovanessian, E. et al. Individualized mRNA vaccines evoke durable T cell immunity in adjuvant TNBC. Nature 651, 1088–1096 (2026). https://doi.org/10.1038/s41586-025-10004-2
Keywords: triple-negative breast cancer, mRNA cancer vaccines, neoantigen immunotherapy, T cell immunity, tumor recurrence