Clear Sky Science · en
Environmentally driven immune imprinting protects against allergy
Why everyday germs might shield us from allergies
Allergies have become strikingly common over the past century, yet our genes have barely changed. This study asks a deceptively simple question with big implications for parents, doctors and public health: can ordinary exposure to a rich, microbe-filled environment "train" the immune system in a way that protects against allergies? Using mice as stand-ins for humans, the researchers uncover how early and repeated encounters with diverse germs and foods imprint the immune system, making dangerous allergic reactions less likely later in life.
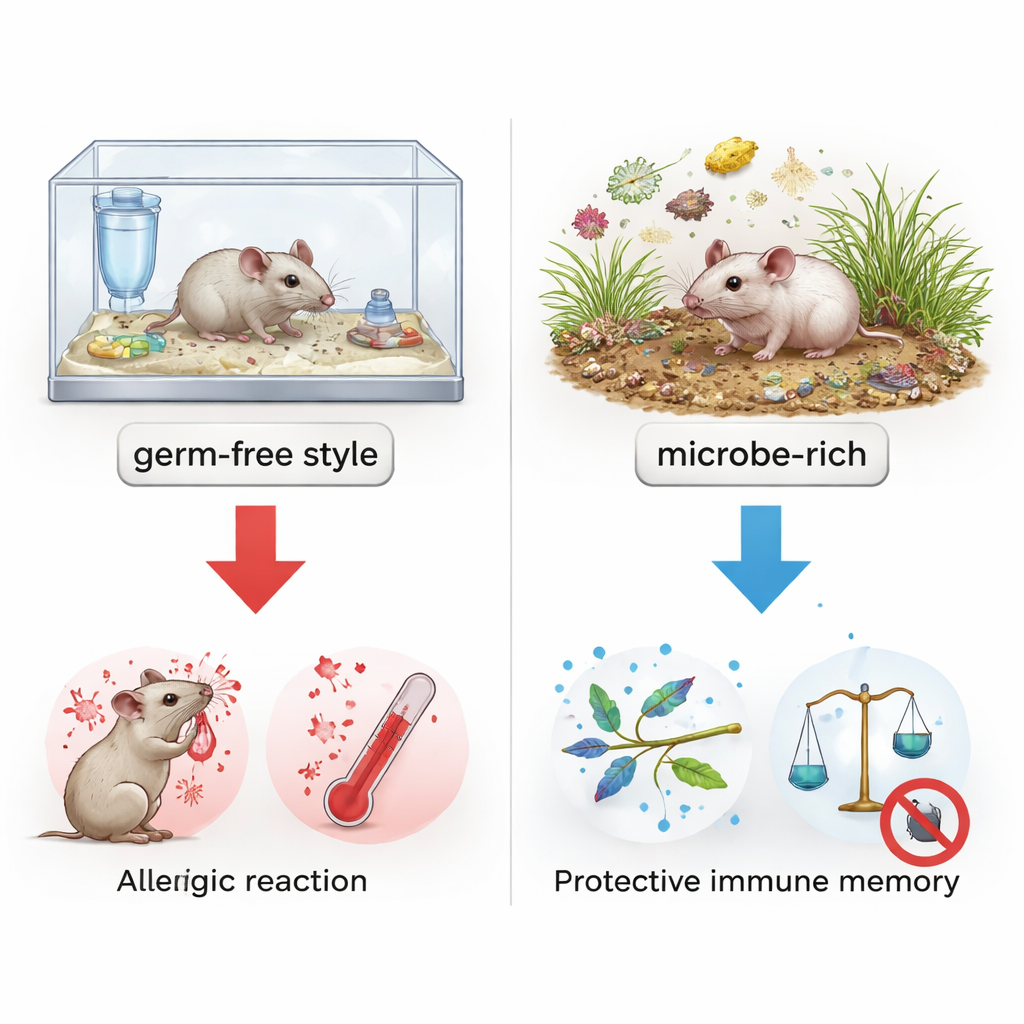
Two kinds of mice, two very different allergy outcomes
The team compared standard laboratory mice, raised in ultra-clean conditions, with "pet shop" mice that had lived in a much messier, more natural environment. Both groups were exposed to a model allergen and then challenged to trigger an allergic reaction resembling anaphylactic shock. The contrast was dramatic: clean lab mice developed severe, even life‑threatening reactions, while pet shop mice showed only mild symptoms. Yet the pet shop mice were not generally defective in their ability to react; when their mast cells—the cells that release histamine during allergy—were activated directly, they responded much like the lab mice. This pointed to a key difference not in the final execution of allergic reactions, but in the way earlier immune responses had been "set" by their environments.
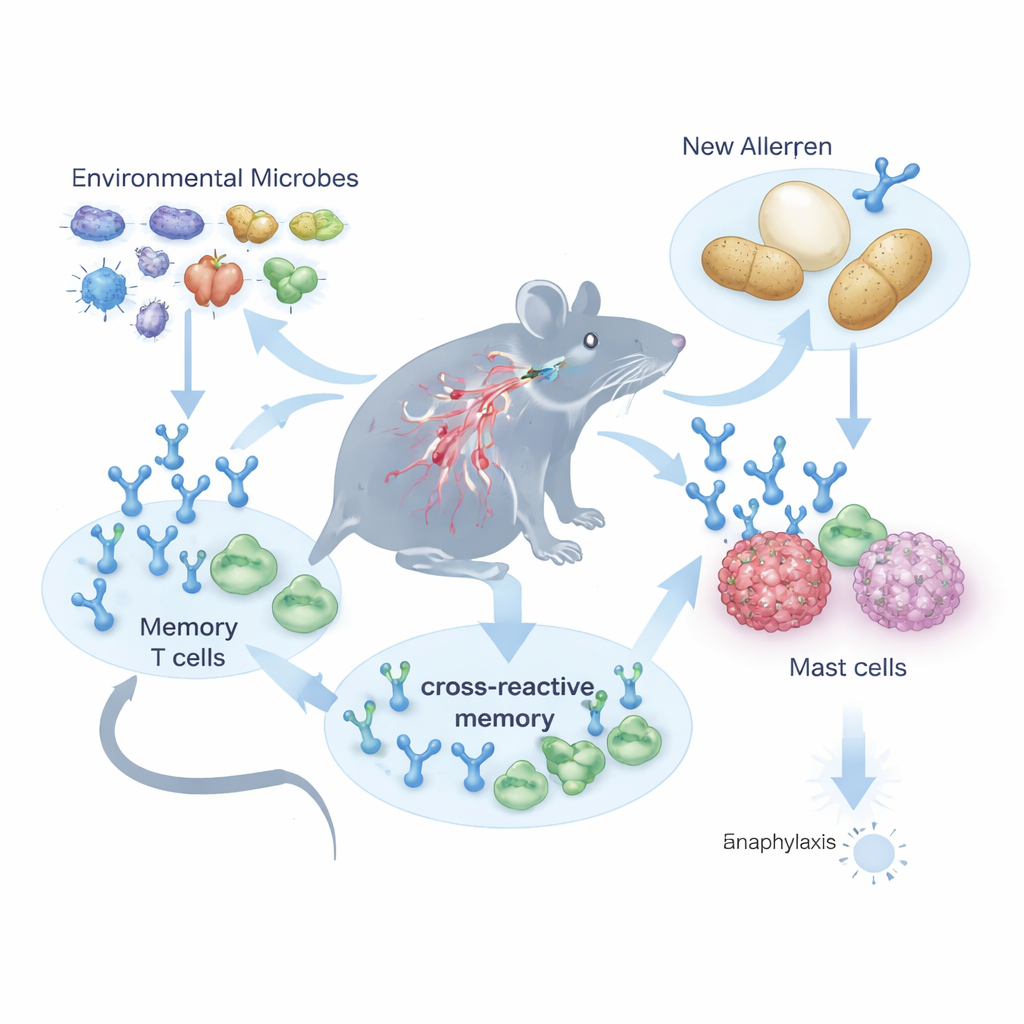
Immune memories that recognize strangers
Diving deeper, the researchers found that pet shop mice carried antibodies and T cells that reacted to the test allergen even before they had ever seen it. That sounds paradoxical, but it fits with a principle called cross‑reactivity: immune cells trained on one set of molecules can sometimes recognize related shapes on completely different proteins. The pet shop mice’s immune systems, shaped by years of exposure to a grab bag of microbes and food components, had built a broad, cross‑reactive memory pool. When these mice later encountered the allergen in an allergy‑promoting setting, this pre‑existing memory nudged their response toward producing protective antibody types rather than the IgE antibodies that drive classic allergic reactions.
A ticking clock for allergy risk
The timing of exposure turned out to be crucial. In mice descended from pet shop parents but born and raised in the laboratory, the researchers showed there is a brief early‑life window when allergic sensitization is easy to induce. If these young mice were exposed to allergen during this perinatal period, they developed strong, long‑lasting allergic responses. The same type of exposure given later in adulthood, after they had accumulated more immune experience, instead produced a balanced antibody mix and protection from anaphylaxis. Strikingly, repeated allergen exposures in an immune‑stimulating context could even reverse an already established allergic state, pushing the system away from a fragile, allergy‑prone configuration.
How similar proteins share protection
To test how far this protection extends, the team used closely related proteins, such as ovalbumin from different bird species, and complex mixtures of legume proteins from soy, peas and peanuts. When mice were first exposed to one version of a protein in an infection‑like or tolerizing (oral) context, they became harder to sensitize to other versions later, even when the sequences were only partly similar. In the case of diets, mice raised on soy‑containing feed were not only less likely to react to soy as an allergen, but also showed reduced reactions to pea and peanut extracts. This suggests that everyday eating in a diverse plant‑based diet may quietly generate a web of cross‑tolerance that dampens the risk of strong allergic responses to related foods.
What this means for the allergy epidemic
Taken together, the study supports a mechanistic twist on the “hygiene hypothesis.” Rather than simply saying that dirt is good and cleanliness is bad, the work shows that repeated, varied exposure to microbes and dietary proteins steers the immune system away from a purely allergic mode by building cross‑reactive memory and tolerance. In environments where children encounter fewer infections, fewer microbes and more restricted diets, that protective imprinting may be weaker, leaving a larger window for allergy to take hold. While these findings come from mice and cannot be applied directly as medical advice, they outline a biological pathway by which modern, sanitized lifestyles could be fueling the rise of allergies—and hint that thoughtfully timed, safe exposures to diverse microbes and foods might one day help nudge immune systems back toward a more resilient state.
Citation: Erickson, S., Lauring, B., Cullen, J. et al. Environmentally driven immune imprinting protects against allergy. Nature 650, 987–996 (2026). https://doi.org/10.1038/s41586-025-10001-5
Keywords: allergy, immune memory, cross-reactivity, hygiene hypothesis, oral tolerance