Clear Sky Science · en
Dissecting gene regulatory networks governing human cortical cell fate
How brain stem cells decide what to become
Before we are born, our brains are built from a small pool of stem-like cells that must decide whether to keep dividing or turn into one of many kinds of nerve and support cells. Tiny switches in our DNA, called gene regulators, guide these choices. This study shows, in unusually fine detail, how dozens of those switches work together to shape the developing human cortex—the brain region that supports thought, sensation and memory—and how glitches in these controls may contribute to mental and developmental disorders. 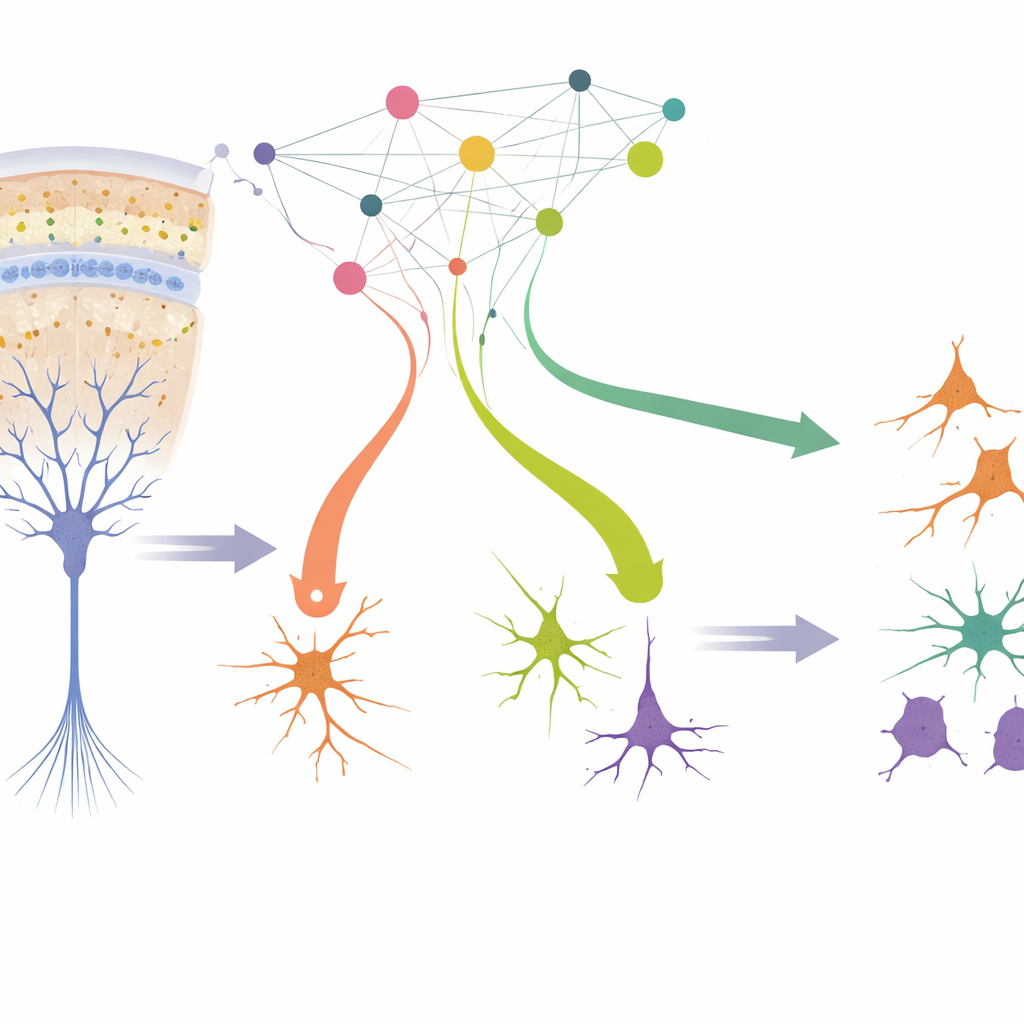
A laboratory window into the growing human cortex
The authors created a lab system that closely mimics how the human cortex develops in the womb. They started with radial glia, the main stem cells that line the brain’s inner surface during mid-pregnancy and give rise to most other cortical cells. By briefly bathing human fetal tissue in growth factors, they enriched for these stem cells and then withdrew the factors so the cells would naturally begin to specialize. Within a week, the cultures produced the major players found in the prenatal cortex: excitatory neurons that send signals, inhibitory interneurons that fine-tune activity, and glial cells that support and insulate neurons. Detailed comparisons to existing brain atlases showed that the lab-grown cells strongly resemble their in‑vivo counterparts and display less stress than cells in many organoid models.
Switching genes off one by one, cell by cell
To see how specific genes control this developmental drama, the team turned to a powerful screening method called Perturb‑seq. They used a CRISPR interference system that can reliably dim, rather than cut, chosen genes, avoiding toxic DNA damage. In more than one hundred thousand single cells, they selectively repressed 44 transcription factors—genes that act as master switches on many others—and then measured the full set of active genes in each cell. This allowed them to link the loss of each switch to both changes in gene activity and shifts in what types of cells appeared in the cultures.
Balancing cell types and timing in the developing cortex
Several of the targeted switches produced striking effects. Turning down NR2E1 pushed radial glia to stop dividing earlier and to generate more inhibitory neurons and, later, more oligodendrocytes, suggesting this factor normally slows the developmental clock. In contrast, reducing ARX had the opposite effect: it favored excitatory neurons over inhibitory ones and kept lineages in a more immature state. Another factor, ZNF219—previously not known to act in the cortex—was found to hold back neural differentiation; when repressed, it boosted production of both excitatory and inhibitory neurons, with a tilt toward excitatory cells. By combining the gene perturbations with DNA barcodes that permanently mark all descendants of individual stem cells, the researchers showed that these switches change the “fate bias” of single radial glia clones, altering how much each clone contributes to different lineages and at what developmental stage.
Shared output genes linked to brain disorders
When the team compared the gene expression changes caused by the different perturbations, they noticed that about a quarter of all affected genes were hit by more than one transcription factor. Many of these shared targets are involved in how young neurons connect, migrate and mature. Importantly, these convergent genes overlapped strongly with gene sets previously tied to conditions such as schizophrenia and major depression. For example, genes like PTPRD and IL1RAPL1, known from human and mouse studies to influence neurogenesis and behavior, sat at the crossroads of several regulatory circuits. This suggests that different genetic hits in early development can funnel into common downstream pathways that shape brain wiring and disease risk. 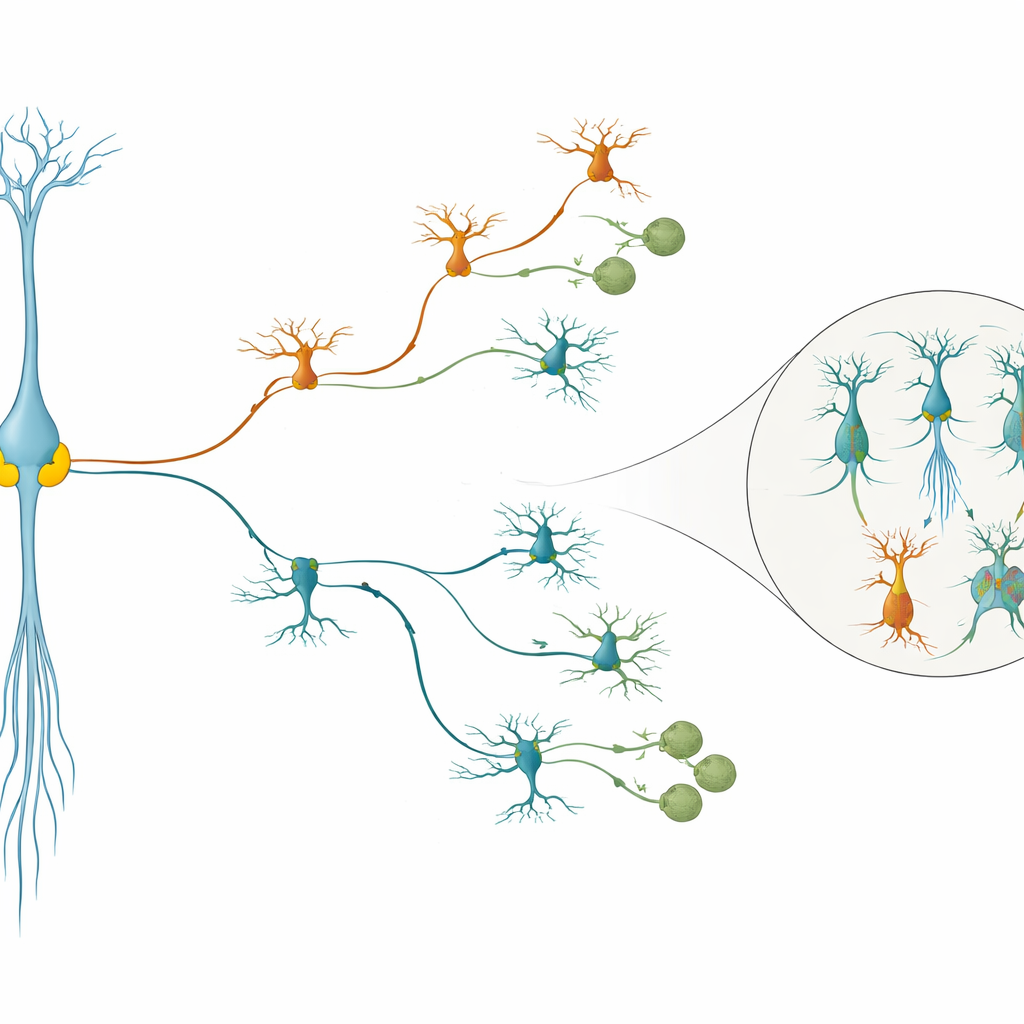
Safeguarding neuron identity after birth of the cell
Beyond deciding “what” cell a stem cell will make, some switches also guarded “which subtype” that cell would become. Within inhibitory neurons, loss of ARX produced an unusual subgroup marked by the gene LMO1 and changes in signaling pathways that normally guide cell movement and synapse formation; similar ectopic cells appeared in both human tissue slices and rhesus macaque cells. Using a double‑perturbation strategy, the authors showed that simultaneously repressing ARX and LMO1 partly erased this abnormal state, indicating that ARX normally preserves proper interneuron identity in part by keeping LMO1 in check. Notably, many of the transcription factors with the strongest effects—including ARX, NR2E1, SOX2, CTCF, NEUROD2, PHF21A and ZNF219—have been implicated in neurodevelopmental and psychiatric disorders, linking their single‑cell findings to clinical genetics.
Why these findings matter for understanding the human brain
Together, this work delivers a blueprint of how a network of gene switches in human radial glia choreographs both the mix of cell types and the pacing of cortical development, and how errors in that network can misdirect neuron identity. By using a faithful primary cell system, single‑cell readouts and lineage tracing, the authors provide a versatile framework for probing additional genes and pathways in human and primate brain development. For non‑specialists, the key takeaway is that many different genetic changes can converge on shared developmental programs that shape how our brains are built—and when those programs are disturbed, the consequences can echo as cognitive and psychiatric disorders later in life.
Citation: Ding, J.W., Kim, C.N., Ostrowski, M.S. et al. Dissecting gene regulatory networks governing human cortical cell fate. Nature 651, 732–742 (2026). https://doi.org/10.1038/s41586-025-09997-7
Keywords: cortical development, radial glia, single-cell CRISPR, neurogenesis, neuropsychiatric disorders