Clear Sky Science · en
Ageing promotes microglial accumulation of slow-degrading synaptic proteins
Why Our Brain’s Protein Cleanup Matters as We Age
Most of us hope to stay sharp as we grow older, yet age is the biggest risk factor for Alzheimer’s and other dementias. This study asks a deceptively simple question: what happens to the day‑to‑day “housekeeping” of proteins inside brain cells as we age? By building new molecular tools in mice to follow freshly made neuronal proteins over time, the researchers trace how proteins are renewed, how they clump into aggregates, and how the brain’s immune cells, microglia, help clear the mess. Their findings reveal a broad slow‑down in protein cleanup that centers on synapses—the junctions where neurons communicate—and may help explain age‑related memory loss.
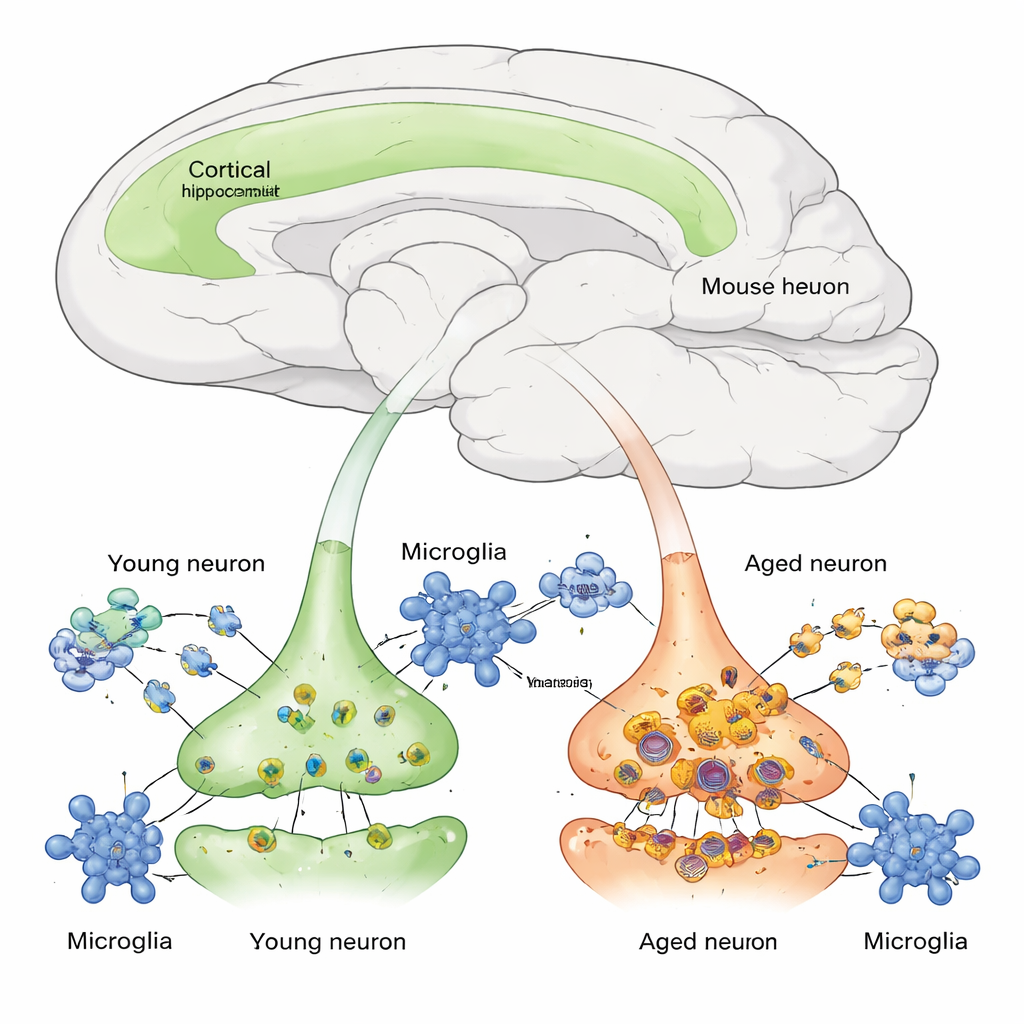
Tracking New Proteins in Living Brain Cells
Proteins in our cells are constantly being made and broken down, a balance known as proteostasis. Until now, it has been hard to measure this process in specific cell types inside a living mammalian brain. The authors engineered mice with a genetic “tagging” system called BONCAT that causes only certain neurons—those using the Camk2a promoter, mostly excitatory cortical and hippocampal neurons—to insert a special, clickable amino acid into newly made proteins. After giving mice this amino acid for a short period, the team could pull down and identify only the freshly synthesized neuronal proteins using mass spectrometry, and then watch how their levels fell over time as the proteins were degraded.
How Aging Slows Protein Turnover
Using a viral version of the tagging tool, the researchers labeled neurons in young (4‑month‑old), middle‑aged (12‑month‑old), and aged (24‑month‑old) mice. They then followed thousands of proteins across several brain regions—sensory and visual cortex, hippocampus, and hypothalamus—during a two‑week “chase” period after labeling stopped. By modeling these decay curves, they estimated each protein’s half‑life, or how long it takes for half of it to be removed. On average, neuronal protein half‑lives roughly doubled between young and aged mice, with most of the slow‑down emerging after middle age. The effect varied by region, and certain sets of proteins degraded in tightly coordinated fashion, indicating that whole pathways—such as those controlling synaptic signaling—share similar, age‑dependent kinetics.
Clumping Proteins and Vulnerable Synapses
A slower breakdown rate raises the risk that proteins will misfold and stick together. The team isolated detergent‑insoluble aggregates from aged mouse brains and, using their neuronal tag, defined an “aggregome” of 1,726 neuronal proteins that end up in these clumps. Nearly half of the proteins that showed age‑reduced degradation also appeared in aggregates, and many were linked by previous genetic work to neurodevelopmental or neurodegenerative disorders. Synaptic proteins stood out: components of pre‑ and postsynaptic machinery, cell junctions, and mitochondrial elements at synapses were highly enriched among both slow‑degrading and aggregating proteins. Microscopy confirmed that specific proteins such as RTN3 and SRSF3 formed aggregate‑like puncta in aged but not young mouse hippocampus. This synapse‑focused proteostasis failure fits with long‑standing evidence that synaptic loss tracks closely with cognitive decline.
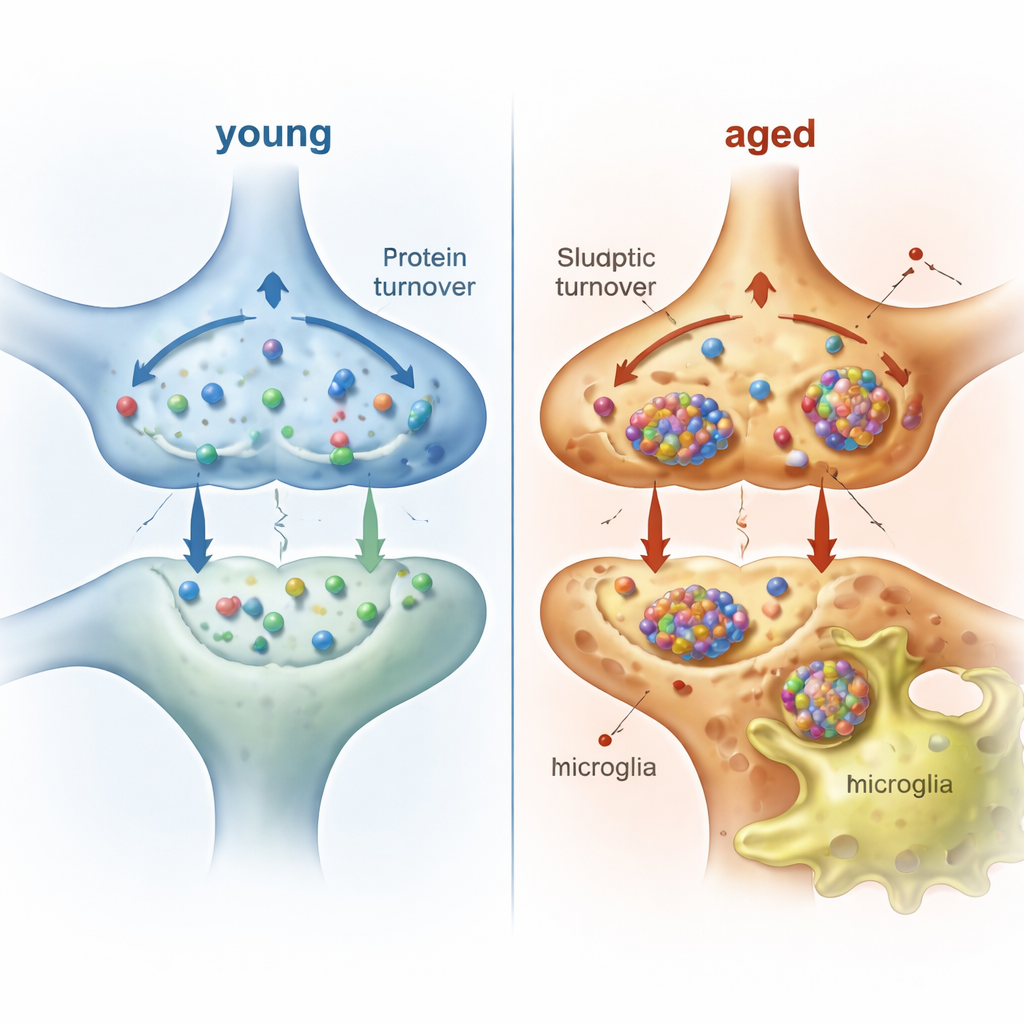
Microglia as the Brain’s Cleanup Crew
Neurons do not handle this burden alone. Microglia, the brain’s resident immune cells, constantly survey and prune synapses. To see what neuron‑derived material microglia actually take up in living animals, the authors labeled neuronal proteins for a week in young and aged mice, then purified hundreds of thousands of microglia and pulled down any tagged proteins inside them. They detected hundreds of neuronal proteins within microglia, with strong representation of synaptic, membrane, and mitochondrial components. Many of these proteins carry signal sequences or are known exosome cargo, suggesting secretion as one route of transfer, while others are consistent with microglial engulfment of synaptic elements. In aged mice, microglia contained more kinds and higher amounts of neuron‑derived proteins than in young mice, yet the balance of pre‑ vs. postsynaptic components remained similar.
When Cleanup Falters, the Brain Pays the Price
By overlapping three datasets—proteins whose degradation slows with age, proteins found in neuronal aggregates, and proteins that accumulate in microglia from aged brains—the study identified 166 proteins that sit at the intersection of these processes. More than half of all neuron‑derived proteins enriched in aged microglia showed some kind of age‑related proteostasis problem. Many are encoded by genes previously flagged as risk factors for Alzheimer’s, Parkinson’s, and other brain disorders. Together, these results outline a cascade: with age, neuronal protein turnover slows, especially at synapses; vulnerable proteins are more likely to misfold and aggregate; and microglia increasingly remove these damaged components, possibly by engulfing stressed synapses. In the short term, this may protect neurons, but over decades it could contribute to synapse loss and impaired cognition. Understanding and eventually restoring healthy neuronal proteostasis may therefore be a key strategy for maintaining brain resilience in later life.
Citation: Guldner, I.H., Wagner, V.P., Moran-Losada, P. et al. Ageing promotes microglial accumulation of slow-degrading synaptic proteins. Nature 650, 930–941 (2026). https://doi.org/10.1038/s41586-025-09987-9
Keywords: brain aging, protein turnover, synapses, microglia, neurodegeneration