Clear Sky Science · en
Common variation in meiosis genes shapes human recombination and aneuploidy
Why embryo chromosome mistakes matter
Many pregnancies end before they are even recognized, often because an embryo carries the wrong number of chromosomes. This study asks a deceptively simple question with profound consequences for fertility and reproductive health: how do ordinary DNA differences between people influence the way egg and sperm cells shuffle and divide chromosomes, and how does that, in turn, affect the chances that an embryo will have too many or too few? By peering into more than a hundred thousand in vitro fertilization (IVF) embryos, the authors reveal how common genetic variation in a handful of genes can subtly tilt the odds toward or away from these chromosome errors.
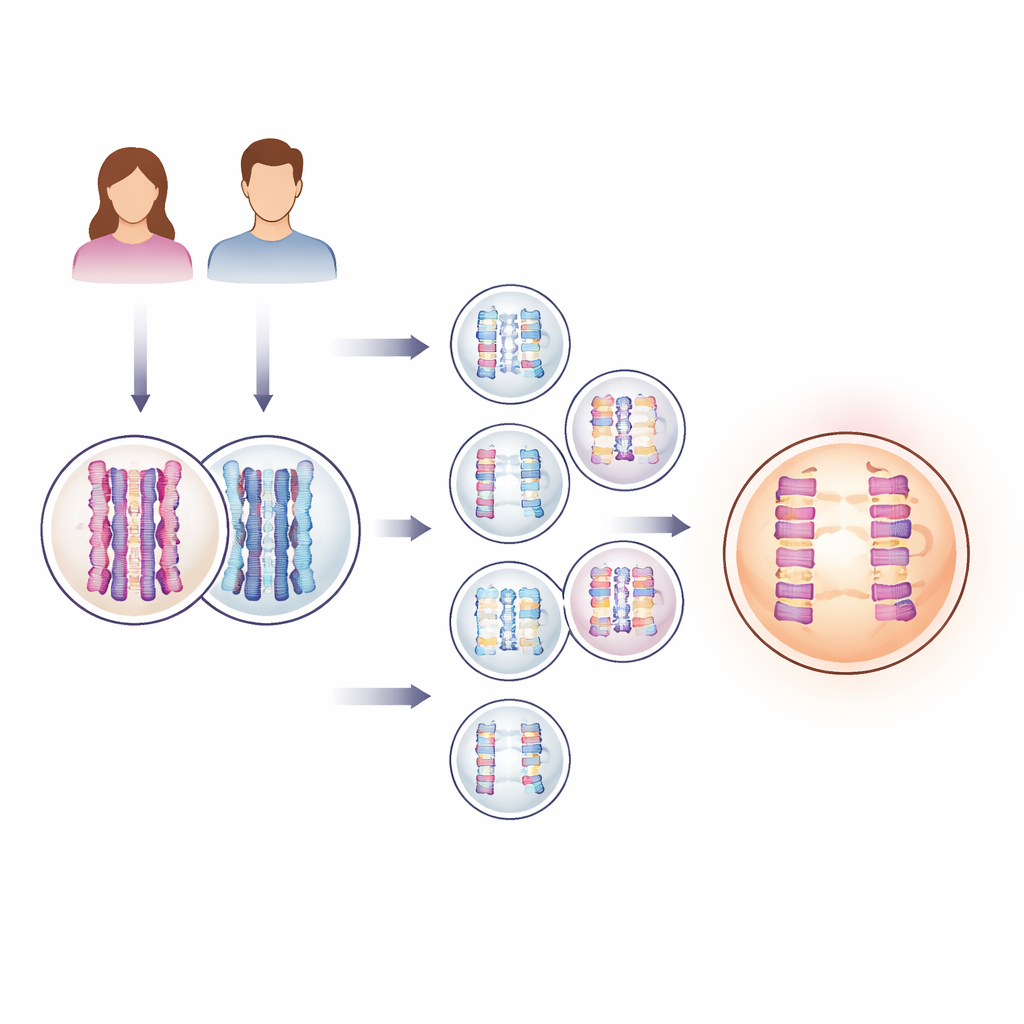
Looking inside thousands of IVF embryos
To tackle a problem that usually unfolds invisibly inside the human body, the researchers turned to genetic testing data routinely collected during IVF. Clinics often biopsy a few cells from an embryo a few days after fertilization to check for chromosome problems before transfer. Here, the team analysed DNA from 139,416 such embryos, together with DNA from nearly 23,000 pairs of biological parents. With a statistical method that tracks blocks of parental DNA as they pass into embryos, they could both count the "crossovers"—the points where parental chromosomes exchange segments—and detect when an embryo had gained or lost an entire chromosome, a condition called aneuploidy.
How common are chromosome errors and what shapes them?
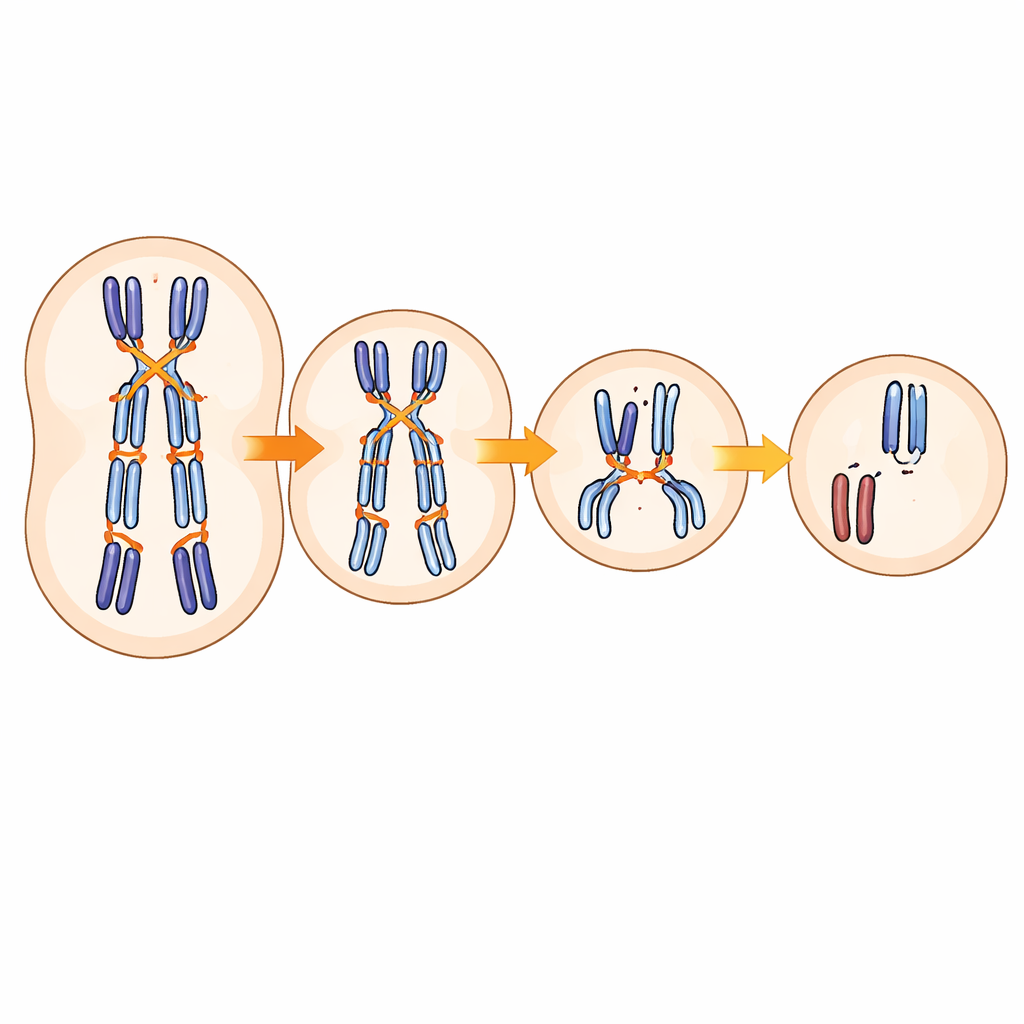
The survey showed that almost 30% of embryos carried at least one aneuploid chromosome. Most of these errors came from the mother rather than the father and often involved specific chromosomes that are already known to be trouble-prone. As expected from clinical experience, the fraction of embryos with maternal chromosome errors rose steeply with maternal age, while paternal age had little effect. Crucially, when the team compared embryos with normal chromosome counts to those with errors, they found that the error-prone embryos tended to have fewer crossovers on their otherwise normal chromosomes. This supports the idea that crossovers do double duty: they generate new genetic combinations, but they also act like physical ties that help chromosomes separate cleanly during egg formation.
Everyday genetic differences in key genes matter
The authors then searched the mothers’ genomes for common DNA variants that tracked with how often their embryos were aneuploid. A standout region lay near a gene called SMC1B, which makes part of a ring-shaped protein complex that holds sister chromosomes together in eggs. Women carrying a particular version of this DNA stretch had a slightly higher fraction of embryos with maternal chromosome errors, and this risk grew with age. Follow-up analyses in large gene-expression datasets showed that the risky version of the DNA sequence is linked to lower activity of SMC1B, apparently through subtle changes in how a transcription factor binds to its promoter. In other words, a common, non-disruptive tweak to a regulatory switch can dial down a cohesion protein just enough to measurably increase the odds of chromosome mis-segregation.
A shared genetic thread across fertility traits
Beyond SMC1B, the study highlighted other genes that help align and connect chromosomes during meiosis, including components of the scaffold that brings paired chromosomes together and enzymes that mark where crossovers will occur. Variants in these genes not only influenced recombination patterns and aneuploidy risk in embryos, but also showed links to the timing of puberty and menopause in large population studies. Yet, taken together, common variants explained only a small slice of the overall variation in embryo chromosome errors. This suggests that environment, chance events during egg development, and rarer genetic changes likely play large roles, even as many of these influences converge on the same molecular machinery.
What this means for human fertility and evolution
For non-specialists, the key message is that the same genetic processes that create healthy diversity in our DNA can, when finely tuned in different ways, also increase the risk of early pregnancy loss. This work shows that ordinary inherited differences in a few meiotic genes can modestly shift the balance, especially as women age, but no single variant seals anyone’s reproductive fate. By combining massive embryo datasets with population genetics, the study also wrestles with an evolutionary puzzle: why variants that nudge up the risk of non-viable embryos can remain common in humans. The authors argue that because many social and environmental factors blur the link between embryo number and actual family size, natural selection may not efficiently weed out such variants. Together, these insights deepen our understanding of why chromosome errors are so frequent in human reproduction and point toward biological pathways that could eventually inform counselling or interventions.
Citation: Carioscia, S.A., Biddanda, A., Starostik, M.R. et al. Common variation in meiosis genes shapes human recombination and aneuploidy. Nature 651, 146–153 (2026). https://doi.org/10.1038/s41586-025-09964-2
Keywords: aneuploidy, meiosis, human fertility, chromosome recombination, SMC1B