Clear Sky Science · en
CFAP20 salvages arrested RNAPII from the path of co-directional replisomes
Keeping Our Genetic Traffic Flowing
Every time a cell divides, it must copy its entire DNA while still using those same DNA instructions to make RNA. This is like trying to repave a busy highway while traffic is still moving. If the copying machinery and the reading machinery collide, the DNA can be damaged, which in the long run contributes to aging and disease, including cancer. This study uncovers a small but crucial protein, called CFAP20, that helps prevent such pileups and keeps the flow of genetic information smooth and safe.
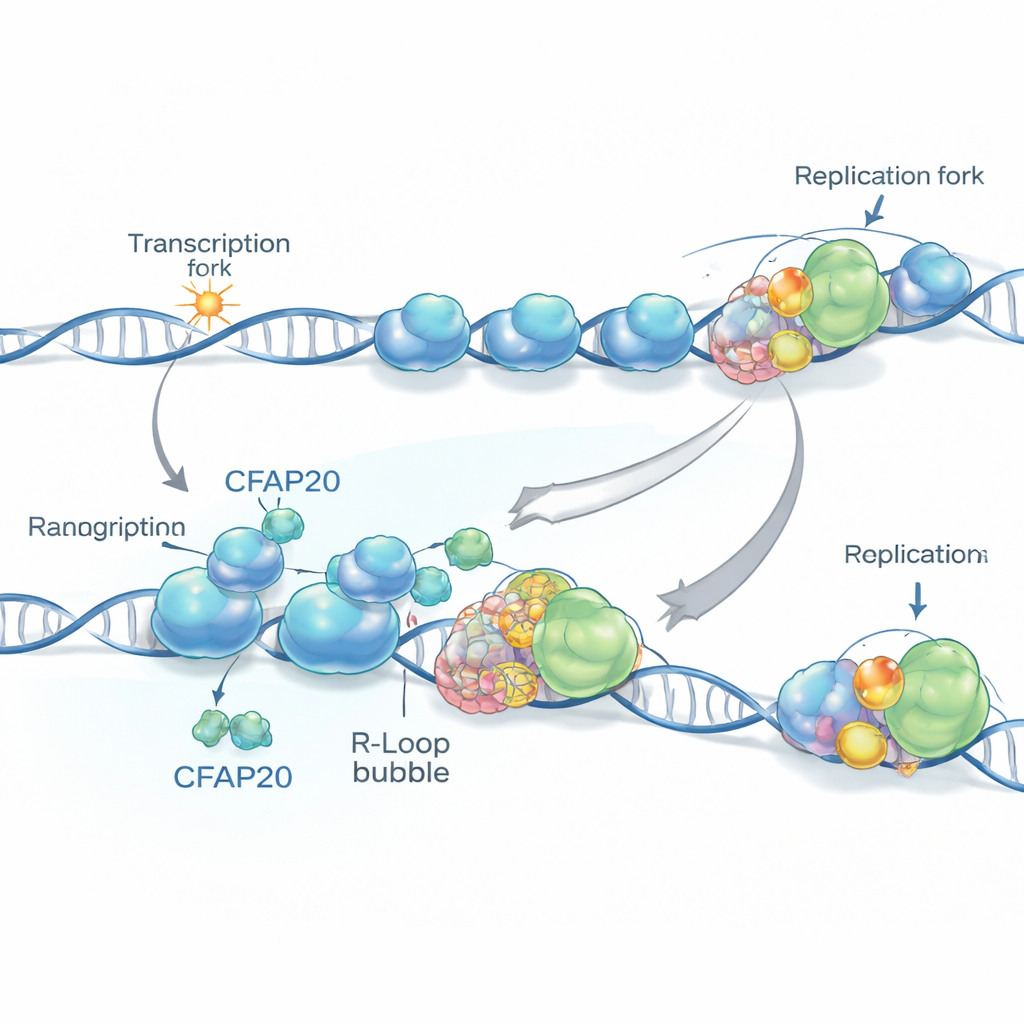
Two Jobs on One DNA Highway
Inside our cells, two major activities share the DNA “road.” One is replication, in which specialized complexes copy DNA before a cell divides. The other is transcription, where RNA polymerase II reads DNA to produce RNA, the first step toward making proteins. Often, both machines move in the same direction along genes, especially near gene starting points called promoters, where reading begins and DNA copying often starts nearby. This arrangement sounds orderly, but it easily becomes congested: RNA polymerase frequently pauses or stalls near promoters, and these stalled complexes can block the DNA-copying machinery coming up from behind.
Dangerous DNA–RNA Knots
When RNA polymerase slows down, the freshly made RNA can loop back and stick to the DNA it came from, creating three-stranded knots called R-loops. These structures are natural and sometimes useful, but in excess they become hazardous obstacles. Using genome-wide mapping tools, the researchers showed that R-loops are especially common near promoters that lie close to DNA replication starting points and are oriented in the same direction as replication. In these spots, a paused reading machine plus an R-loop can present a formidable roadblock to the copying machinery, raising the risk of breaks and gaps in the DNA.
A Small Protein with a Big Protective Role
To find factors that help cells cope with stress on both the reading and copying sides, the team used large-scale CRISPR gene knockout screens. CFAP20, previously known mainly for its role in tiny hairlike cell structures called cilia, emerged as a surprising hit. When they removed CFAP20 from human cells, R-loops piled up near promoters, DNA replication forks sped up abnormally between troubled regions, and fewer replication starting points fired overall. The result was an uneven replication pattern, with some forks stalling while neighboring ones raced ahead and left vulnerable single-stranded gaps behind. A cancer-associated mutant version of CFAP20 failed to prevent these problems, highlighting the protein’s specific protective role in the nucleus, distinct from its function in cilia.
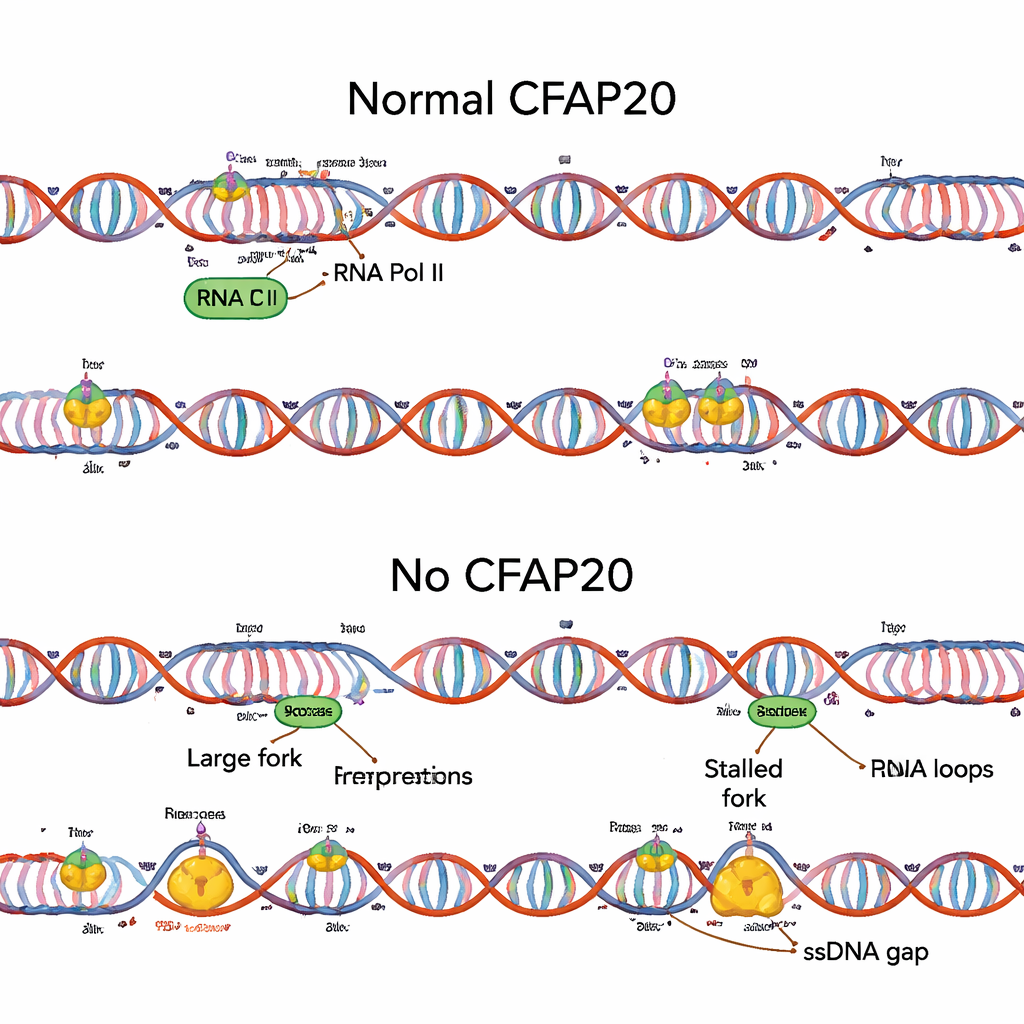
Balancing Strong Transcription with Safe Replication
The study also examined how CFAP20 interacts with Mediator, a large complex that boosts the activity of RNA polymerase II at promoters. Without CFAP20, Mediator-driven high transcription becomes a liability: R-loops surge and replication is disturbed. Intriguingly, when the researchers disabled a Mediator subunit that helps drive this strong transcription, many of the harmful effects of CFAP20 loss disappeared. R-loops fell, replication fork speeds normalized, and DNA copying patterns became more regular. Additional experiments showed that CFAP20 physically associates with RNA polymerase II and helps resolve slow or arrested polymerases tangled with R-loops, effectively clearing the track before the DNA-copying machinery arrives.
Why This Matters for Health and Disease
In simple terms, CFAP20 acts as a traffic manager on our DNA, rescuing stalled reading machines so they do not impede copying and do not generate dangerous gaps. When CFAP20 is missing or defective, local jams near promoters trigger compensatory speeding elsewhere, which paradoxically weakens genome stability. Because many tumors experience heightened transcription and replication stress, they may be particularly dependent on this safeguard protein. Understanding how CFAP20 coordinates these two essential processes not only clarifies a fundamental aspect of cell biology but may also point to new vulnerabilities that could be targeted in certain cancers.
Citation: Uruci, S., Boer, D.E.C., Chrystal, P.W. et al. CFAP20 salvages arrested RNAPII from the path of co-directional replisomes. Nature 650, 1025–1034 (2026). https://doi.org/10.1038/s41586-025-09943-7
Keywords: DNA replication, transcription, R-loops, genome stability, CFAP20