Clear Sky Science · en
Disease tolerance and infection pathogenesis age-related tradeoffs in mice
Why some infections are deadlier as we age
Why do older adults so often fare worse during severe infections, even when they are exposed to the same germs as younger people? This study uses mice to explore that puzzle, focusing on sepsis—a life‑threatening reaction to infection. The researchers show that the body’s own protective programs can flip with age: genes that shield the heart and other organs from damage in youth can actually drive harm later in life. Understanding this age‑dependent switch could change how we design treatments for infections in children, adults and seniors.
Different paths from the same infection
To mimic a common human emergency, the team infected young and aged mice with a mixture of two frequent sepsis‑causing bacteria, Escherichia coli and Staphylococcus aureus. They carefully chose a dose that killed about half the animals (an LD50), so that some mice would survive and others would not, despite having the same starting exposure. Surprisingly, both age groups carried similar amounts of bacteria in their organs. What differed was how their bodies coped with the damage. Young and aged mice followed distinct “health journeys” during sepsis: body temperature, visible sickness and the timing of decline or recovery all separated into clear survivor and non‑survivor trajectories. 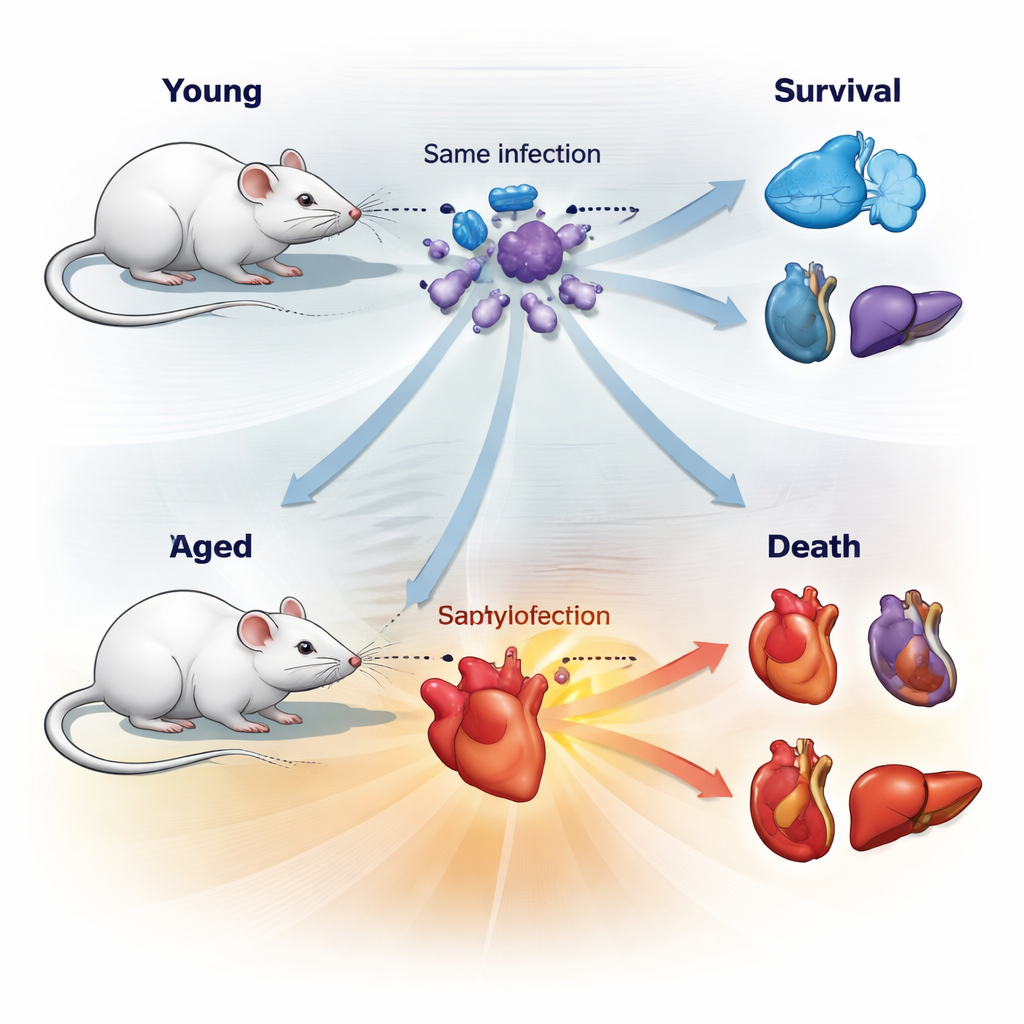
Heart changes tell two age‑specific stories
When the scientists examined organs, the heart emerged as a central player. Young mice that died of sepsis developed enlarged, swollen hearts with stretched chambers—signs of severe strain—and showed high levels of blood markers used in hospitals to flag heart injury and heart failure. Their kidneys and livers were also hurt, but less dramatically. In contrast, aged mice that died showed the opposite heart pattern: their hearts became smaller and wasted, even as kidney injury and loss of kidney function were more pronounced. Aged survivors, interestingly, often had enlarged hearts but less damage overall. Gene‑activity profiling of heart tissue confirmed that young and aged mice were engaging different remodeling programs: younger hearts switched on pathways linked to growth and structural change, whereas older hearts activated pathways tied to protein breakdown and cell atrophy.
A protective circuit in young hearts
Digging into these molecular signatures, the researchers highlighted a control gene called Foxo1 and one of its targets, Trim63, which encodes a protein known as MuRF1 involved in tagging muscle proteins for recycling. In young survivor hearts, Foxo1 and Trim63 were strongly turned on compared with young mice that died or were never infected. Blocking FoxO1 with a drug, or deleting it specifically in heart muscle cells, made young mice much more likely to become sick and die, even though their bacterial levels and inflammatory signals stayed similar. Likewise, young mice lacking Trim63 suffered worse heart swelling, more leakage of heart‑damage markers into the blood, and broader injury to liver and kidney. These results indicate that, in youth, the FoxO1–Trim63 pathway acts as a disease‑tolerance system: it does not fight the microbes directly, but instead helps the heart adapt to stress and prevents multi‑organ failure.
When yesterday’s protector becomes today’s threat
The same genes told a different story in old age. In aged mice, survival no longer depended on ramping up FoxO1 in the heart. Instead, turning FoxO1 off—either genetically in muscle cells or with a drug—actually protected older animals from sepsis‑induced sickness and death. Aged mice lacking Trim63, or treated with a MuRF1 inhibitor, also fared better: they had enlarged but structurally healthier hearts, lower levels of blood markers that signal heart strain, and less damage to kidneys and livers. In other words, the very circuit that fosters healthy adaptation to infection in young hearts now drove harmful remodeling and organ injury in older hearts. This age‑flip of function fits a concept from evolutionary biology called “antagonistic pleiotropy,” where traits that boost fitness early in life carry hidden costs later on. 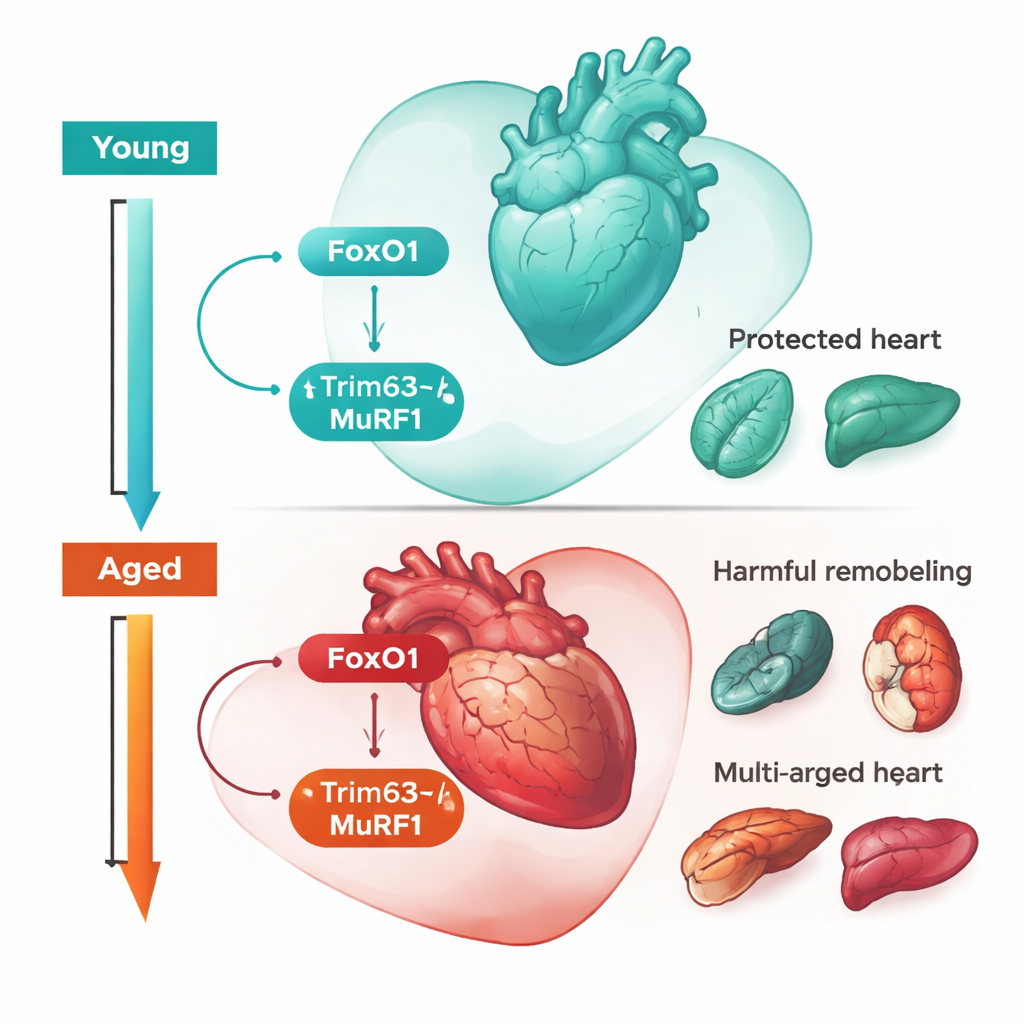
What this means for treating infections across the lifespan
To a lay reader, the key message is that combating severe infections like sepsis is not just about killing germs—it is also about how our organs weather the storm. This study shows that the heart’s stress‑response machinery, built around FoxO1 and Trim63, helps young bodies tolerate infection but can turn into a liability with age. As a result, a drug that blocks this pathway might be life‑saving for an older patient while being dangerous for a younger one. The work underscores that therapies for infectious disease may need to be age‑tailored, taking into account that the same gene can wear two faces: guardian in youth, troublemaker in old age.
Citation: Sanchez, K.K., McCarville, J.L., Stengel, S.J. et al. Disease tolerance and infection pathogenesis age-related tradeoffs in mice. Nature 650, 727–735 (2026). https://doi.org/10.1038/s41586-025-09923-x
Keywords: sepsis, ageing, disease tolerance, heart remodeling, FoxO1 Trim63 pathway