Clear Sky Science · en
Microbiota-induced T cell plasticity enables immune-mediated tumour control
How Friendly Gut Germs Might Help Fight Cancer
Cancer immunotherapy has transformed treatment for many patients, yet only a fraction see their tumours shrink and stay away. This study asks a deceptively simple question with big implications: can specific gut bacteria "coach" immune cells in ways that make checkpoint-blocking drugs, such as anti–PD‑1 therapy, work better? By following the journey of immune cells from the small intestine to tumours in mice, the authors uncover a direct, testable link between a single intestinal microbe and successful tumour control.
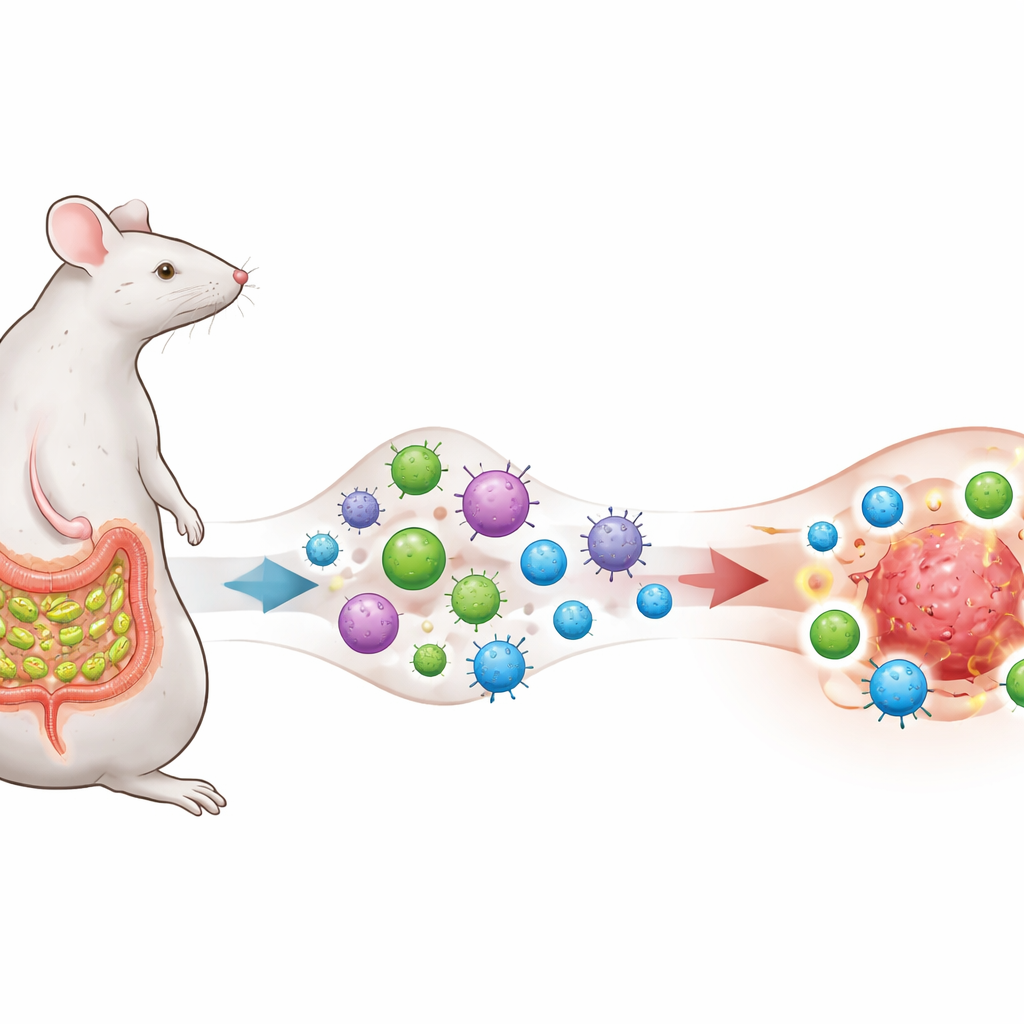
A Microbe That Trains the Immune System
The researchers focus on a gut bacterium called segmented filamentous bacteria (SFB), known to live close to the lining of the small intestine in mice and to provoke a strong but normally home‑building T helper 17 (TH17) cell response. They engineer melanoma, lung and colon cancer cells so that they display a protein fragment also found in SFB, creating an intentional "antigen overlap" between the microbe and the tumours. Mice either remain free of SFB or are colonized with it, and all receive anti–PD‑1 therapy, a widely used checkpoint-blocking antibody that aims to release the brakes on T cells.
Gut-Tutored Cells Turn Tumours from Cold to Hot
Anti–PD‑1 treatment alone, or SFB alone, does little to slow tumour growth. But when tumours carry the SFB‑derived antigen and the mice are colonized with SFB, anti–PD‑1 suddenly becomes highly effective: tumours shrink, survival improves, and surviving animals reject a later tumour re‑challenge without extra therapy, indicating lasting immune memory. In these successful mice, the tumours fill with aggressive CD8 “killer” T cells producing interferon‑γ and other toxic molecules, while suppressive regulatory T cells make up a smaller share of the immune population. In short, the local environment around the cancer is remodeled from relatively "cold" and unresponsive to "hot" and inflamed.
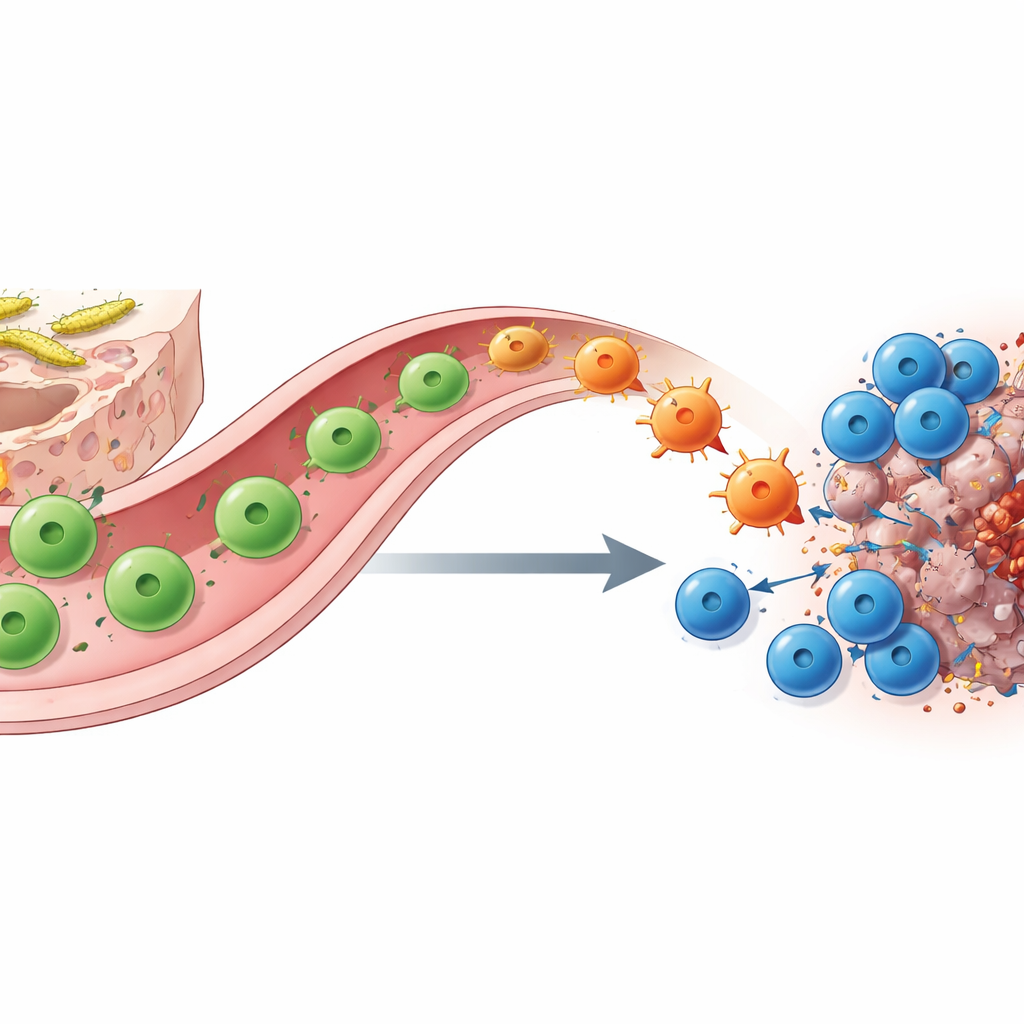
Shape-Shifting Helper T Cells Bridge Gut and Tumour
How does SFB colonization bring about this shift? Using molecular barcoding, single‑cell sequencing and clever genetic tracing, the authors show that the key players are SFB‑specific CD4 helper T cells. In the small intestine these cells start out as TH17 cells that support barrier function and normally produce the molecule IL‑17A. When they encounter matching antigen in a tumour under checkpoint blockade, they migrate from the gut to the cancer site and “re‑specialize” into TH1‑like cells that instead secrete large amounts of interferon‑γ and TNF. These converted “ex‑TH17” cells boost antigen presentation and secrete chemical attractants, drawing in and empowering CD8 killer T cells. Removing either helper or killer T cells, or genetically deleting the IL‑17A‑lineage cells that serve as precursors, largely abolishes the benefit of anti–PD‑1, underscoring their partnership.
Not All Microbes Give Helpful Instructions
To test whether any gut bacterium with overlapping antigens would do, the team repeats the experiment with a different commensal microbe, Helicobacter hepaticus. This organism also drives strong T‑cell responses but predominantly expands regulatory‑type cells that dampen inflammation. When tumours are engineered to display an H. hepaticus antigen and mice are colonized with this bacterium, anti–PD‑1 therapy does not improve. Although many H. hepaticus‑specific helper T cells travel to the tumour, they retain a regulatory character and produce little interferon‑γ, failing to energize killer T cells. This contrast shows that the quality and plasticity of microbe‑induced T‑cell programs—not just antigen matching—determine whether the microbiota will help or hinder cancer immunotherapy.
What This Could Mean for Future Cancer Care
Altogether, the work provides direct proof in animals that a defined gut bacterium can pre‑train helper T cells in the intestine, send them to antigen‑sharing tumours and, under checkpoint blockade, have them switch roles to become potent tumour‑fighting cells. By clarifying this gut‑to‑tumour circuit, the study suggests that carefully chosen or engineered microbes could one day be used alongside immunotherapy to convert more patients’ tumours into treatable, inflamed targets—provided those microbes drive the right kind of flexible, pro‑inflammatory T‑cell response rather than a calming, regulatory one.
Citation: Najar, T.A., Hao, Y., Hao, Y. et al. Microbiota-induced T cell plasticity enables immune-mediated tumour control. Nature 651, 201–210 (2026). https://doi.org/10.1038/s41586-025-09913-z
Keywords: gut microbiota, cancer immunotherapy, T cell plasticity, checkpoint blockade, tumor microenvironment