Clear Sky Science · en
Oral 4′-fluorouridine rescues nonhuman primates from advanced Lassa fever
Why this new pill matters
Lassa fever is a deadly viral disease that strikes tens of thousands of people each year in West Africa and occasionally reaches Europe and the United States. Doctors currently have no approved, reliable medicine to treat it, and existing options are hard to deliver in the remote settings where outbreaks occur. This study tests an experimental antiviral pill called 4′-fluorouridine in monkeys with severe, late-stage Lassa fever, asking a simple but crucial question: can a drug taken by mouth still save a patient when the infection is already raging?
The threat of a quiet killer
Lassa virus spreads to humans mainly from infected rodents and can cause high fever, bleeding, organ failure and death. As cities grow and land use changes, hundreds of millions of people may become exposed in coming decades. Current care usually means fluids, oxygen and blood transfusions, while the long-used drug ribavirin has mixed evidence for benefit and serious side effects, especially in pregnancy. Promising antibody therapies and another antiviral, favipiravir, must be given by vein in high-level hospitals—an impractical solution for rural clinics. A simple, stable pill that could be shipped and taken anywhere would be a game changer for outbreak control.
Putting the pill to a hard test
The researchers worked with African green monkeys, a species whose response to Lassa virus closely mimics severe human disease. They first confirmed that a modern, highly dangerous strain of the virus from Togo reliably caused lethal infection in these animals, matching what had been seen in another monkey species. The animals developed high amounts of virus in their blood and organs, along with fever, bleeding problems and severe damage to the liver, lungs, spleen, adrenal glands and brain—mirroring the multi-organ failure seen in fatal human cases.
Treating when the animals were already very sick
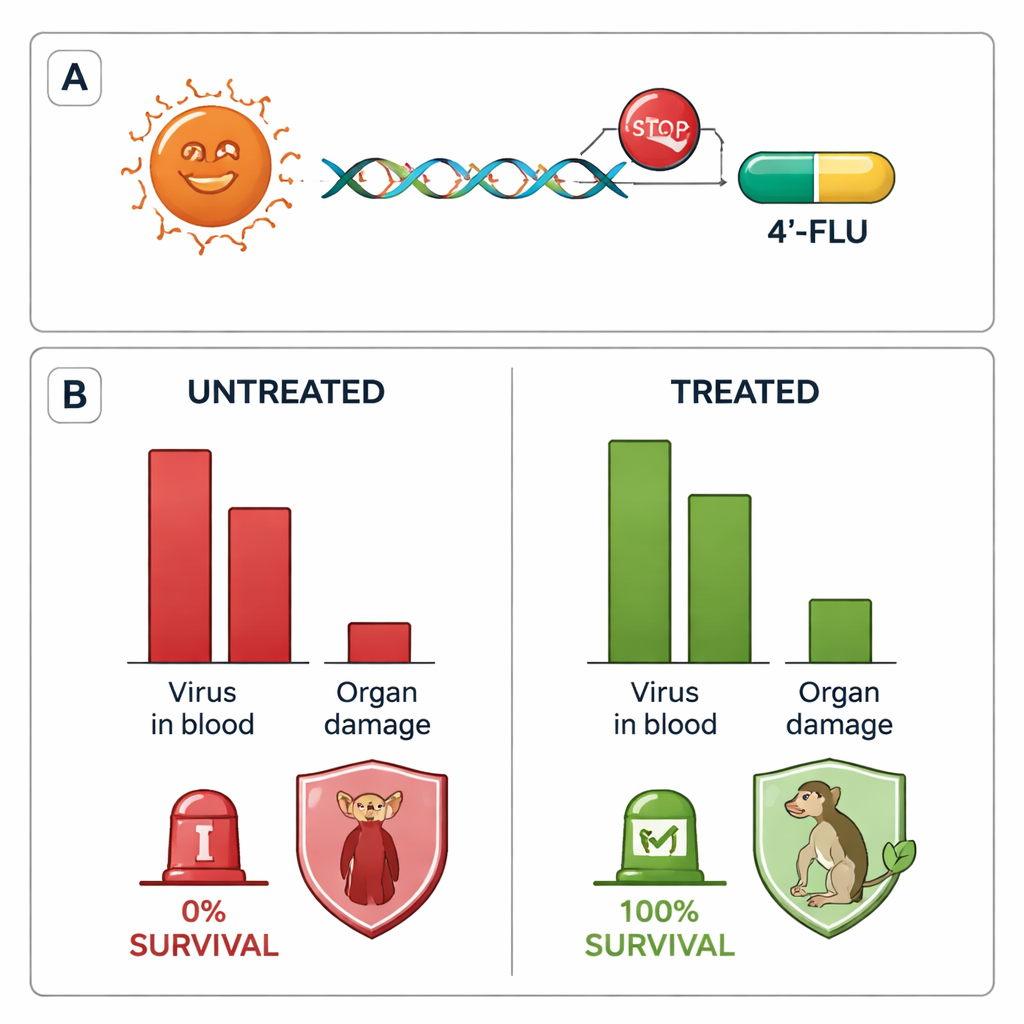
Next, the team infected a new set of monkeys with the same Togo strain and then deliberately waited until day six—when the animals already had clear symptoms and measurable virus in their blood—before starting treatment. Five monkeys received a once-daily oral dose of 4′-fluorouridine for ten days; a sixth was left untreated for comparison. All six were ill at the time therapy began, with fever, poor appetite, abnormal blood counts and high viral loads. The untreated monkey died on day eleven, but every treated monkey survived to the planned end of the study more than a month later, and their fevers and obvious sickness resolved within days of starting the pill.
Clearing the virus and limiting hidden damage
Repeated blood tests showed that in four of the five treated monkeys, infectious virus in the bloodstream dropped rapidly to undetectable levels, while the remaining animal still showed falling levels by the end of monitoring. When the researchers looked at tissues after death, the untreated animals had enormous amounts of virus and widespread organ injury, while treated monkeys had little or no detectable virus and no gross signs of organ destruction. Most tissues that still contained bits of viral genetic material did not yield live virus when tested, suggesting that only harmless remnants remained as the immune system cleaned up. Blood analyses also revealed that treated animals developed virus-specific antibodies and a balanced immune response that avoided the runaway inflammation seen in fatal cases.
How the body’s defenses were reshaped
To understand what was happening inside the animals’ immune systems, the team measured activity of hundreds of genes in blood cells over time. In untreated monkeys, genes linked to severe inflammation, leaky blood vessels and “cytokine storms” stayed highly active until death. In contrast, once treated with the pill, monkeys shifted toward a calmer but effective response involving platelets, monocytes, natural killer cells and T cells—cell types that help clear infection and repair tissues. This pattern suggests that quickly lowering the amount of virus with 4′-fluorouridine allows the body’s own defenses to regain control, instead of being driven into a damaging overreaction.
What this could mean for people
For a layperson, the key message is that an experimental antiviral pill was able to rescue monkeys from a form of Lassa fever that is normally almost always fatal, even when given late in the course of illness. The drug sharply cut the amount of live virus, limited organ damage and gave the immune system room to finish the job. Although more work is needed to test doses, timing and safety in humans, and to watch for any lingering virus in hard-to-reach tissues, this study strongly supports further development of 4′-fluorouridine. If it proves safe and effective in people, health workers in remote clinics could one day hand out a simple pill that turns a frequently deadly infection into a treatable disease.
Citation: Cross, R.W., Turcinovic, J., Prasad, A.N. et al. Oral 4′-fluorouridine rescues nonhuman primates from advanced Lassa fever. Nature 650, 961–969 (2026). https://doi.org/10.1038/s41586-025-09906-y
Keywords: Lassa fever, antiviral pill, 4′-fluorouridine, hemorrhagic virus, outbreak preparedness