Clear Sky Science · en
Insights into DNA repeat expansions among 900,000 biobank participants
Hidden Patterns in Our DNA
Every person’s DNA contains countless tiny stutters—short sequences of letters that repeat over and over again. These repeated stretches can quietly change in length as we grow and age. Sometimes the changes are harmless; other times they trigger serious diseases. This study takes advantage of whole-genome data from nearly 900,000 volunteers to ask a simple, far-reaching question: how do these repeats behave over a lifetime, and what do they mean for our health?
Repeated DNA as a Moving Target
Rather than focusing on single genetic “typos,” the researchers zeroed in on short tandem repeats—runs of one to six DNA letters, such as the famous CAG pattern linked to Huntington’s disease. Using specialized computer methods, they scanned hundreds of thousands of genomes from the UK Biobank and the All of Us Research Program to detect where these repeats had stretched or shrunk. They examined how often repeats changed between generations (inherited changes) and within an individual’s blood cells over time (somatic changes). 
DNA Repair Genes as Repeat “Speed Controllers”
By comparing people who carried the same type of repeat but showed different degrees of expansion in their blood, the team conducted genome-wide association studies to find genetic “modifiers” of repeat behavior. They found 29 regions in the genome where inherited variants consistently sped up or slowed down repeat growth in blood cells. Many of these regions lie within or near known DNA repair genes—molecular machines that patrol our chromosomes for damage. Intriguingly, the same modifier could have opposite effects on different repeats: variants in the MSH3 gene, for example, tended to dampen expansion of a repeat in the TCF4 gene while promoting expansion of the Huntington’s disease–related repeat in HTT. This suggests that repeat instability is governed by a complex interplay between general repair machinery and the local DNA landscape around each repeat.
Everyday Genomes with Aging Repeats
The study shows that repeat instability is not just a rare family tragedy tied to a single disease. Common repeat variants in several genes, including TCF4 and ADGRE2, were found to gradually expand in many people’s blood as they age. For some TCF4 repeats, more than 1% of blood cells in typical 55-year-olds already carry a longer version of the repeat. These age-linked changes reveal that many of us harbor DNA elements that continue to mutate throughout life. The degree of expansion can differ sharply between body tissues and is heavily influenced by a person’s overall mix of modifier variants, captured in a so‑called polygenic score. In some cases, individuals at the high end of this score had repeat expansion rates roughly four times higher than those at the low end.
When Expanding Repeats Tip into Disease
Most expanding repeats in this study did not obviously harm health, but one stood out. A CAG repeat in the 5′ untranslated region of the GLS gene—previously linked only to extremely rare childhood disorders when both gene copies are disrupted—showed a striking pattern in adults who carried very long expansions (around 100 or more repeats). These individuals had strongly elevated markers of liver and kidney damage and a much higher risk of severe chronic kidney disease and other liver problems. Notably, people who carried traditional loss-of-function mutations in GLS did not show the same liver and kidney signs, even though both repeat expansions and these mutations raised blood glutamine levels. 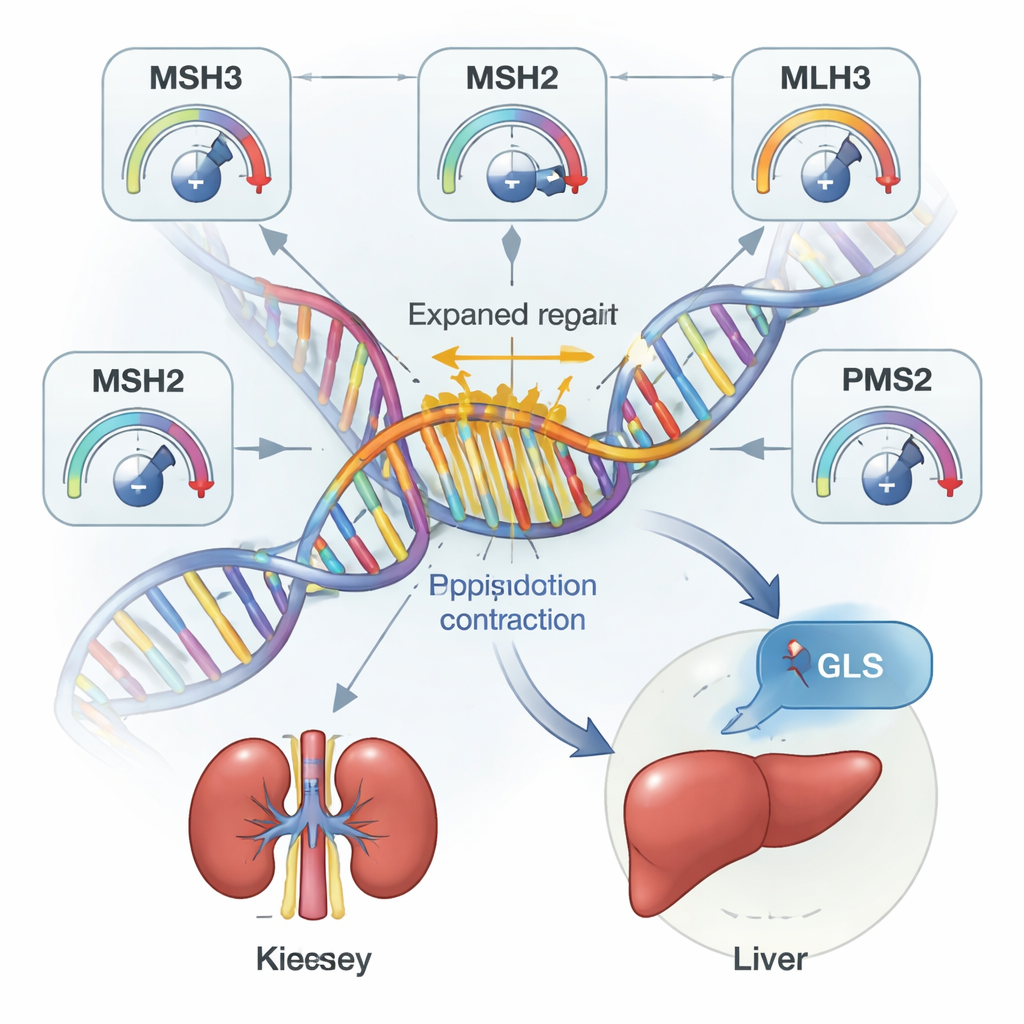
What This Means for Health and Medicine
For non-specialists, the takeaway is that dynamic DNA repeats are a widespread, lifelong feature of our genomes—more like shifting sand dunes than fixed landmarks. Our built-in DNA repair systems and environmental factors such as smoking help determine how fast these dunes move, and at certain sites, extreme shifts can push organs like the kidney and liver toward disease. The work highlights both a caution and an opportunity: blood-based measures of repeat length do not always mirror what happens in the brain or other organs, yet they may serve as practical readouts for drugs designed to slow repeat expansion. As large biobanks continue to grow, they will provide an increasingly powerful lens on how subtle genetic motions shape health across the human lifespan.
Citation: Hujoel, M.L.A., Handsaker, R.E., Tang, D. et al. Insights into DNA repeat expansions among 900,000 biobank participants. Nature 650, 920–929 (2026). https://doi.org/10.1038/s41586-025-09886-z
Keywords: DNA repeat expansion, short tandem repeats, genetic modifiers, genome biobank, kidney and liver disease