Clear Sky Science · en
Biomimetic vesicles engineered from modified tumour cells act as personalized vaccines for post-surgical cancer immunotherapy
Turning Tumor Leftovers into a Second Line of Defense
For many people with solid tumors, surgery is only the first round in a long fight. Even when most of a tumor is removed, tiny clusters of cancer cells can slip away and later seed deadly metastases. This study explores an inventive way to turn a patient’s own tumor cells into a personalized vaccine, designed to hunt down these leftovers after surgery and help the immune system keep cancer from coming back.
Why Leftover Cancer Cells Are So Hard to Erase
Standard treatments struggle with the cells that remain after a surgeon removes the main tumor mass. These scattered cells often hide in distant organs and are shielded from drugs and immune attack. Existing immune-based vaccines try to train the body to recognize tumor markers using specialized immune cells called dendritic cells, but these living vaccines are hard to manufacture, do not travel efficiently to key immune organs, and have shown modest benefits in the clinic. A central challenge is that every patient’s cancer carries a unique blend of mutations, so one-size-fits-all vaccines usually miss important targets.
A Hidden Switch that Makes Cancer Cells More Noticeable
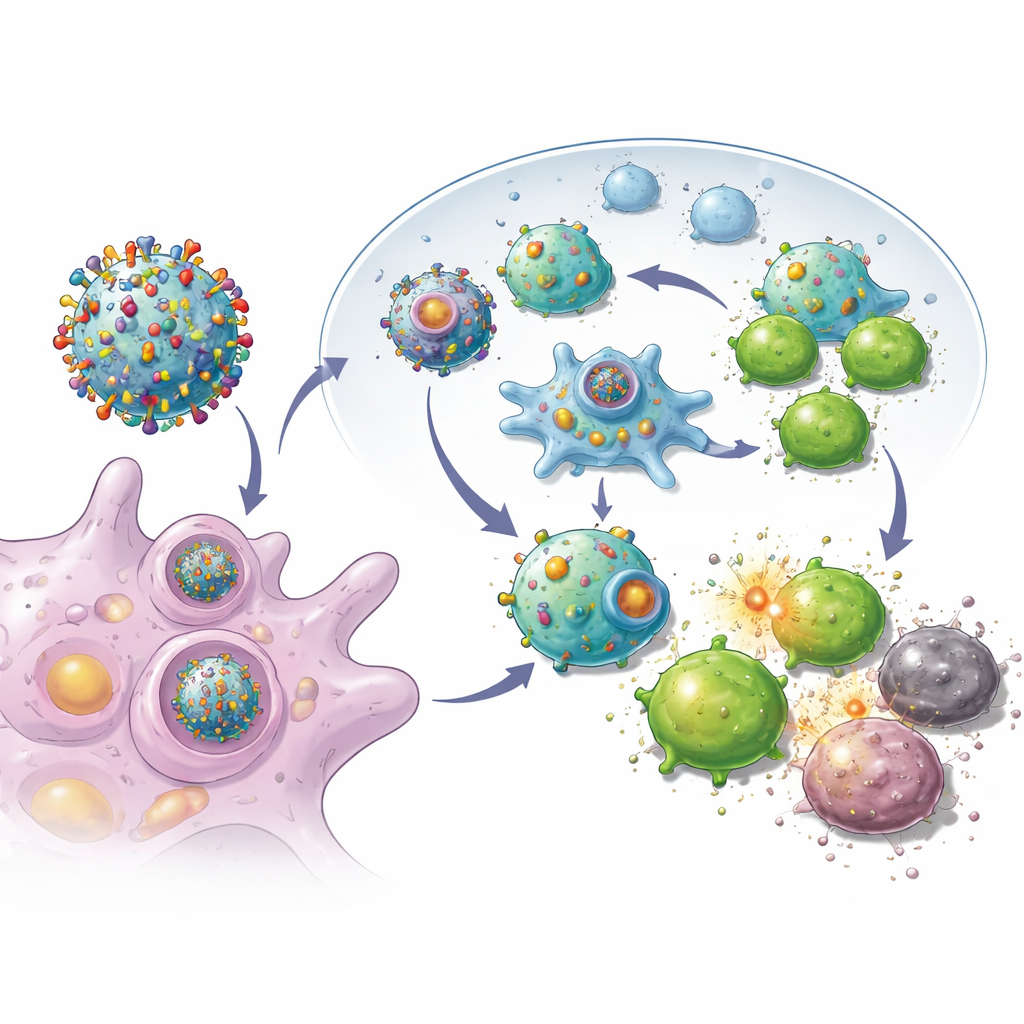
The researchers began by searching for natural switches in the immune system that make cells better at showing their internal contents to immune sentinels. They homed in on a protein called syntaxin 11, normally abundant in immune cells but scarce in many tumors. By boosting this protein in aggressive breast cancer cells from mouse models, they found that the cancer cells started to behave more like professional immune sentinels: they displayed higher levels of molecular "name tags" and helper signals on their surface. In effect, the tumor cells were reprogrammed to reveal more of their unique features to the immune system instead of hiding them.
Building Tiny Imitation Immune Messengers
Armed with these reprogrammed tumor cells, the team stripped off their outer membranes and used them to coat biodegradable nanoparticles loaded with a small immune-stimulating drug. The result was a swarm of nanoscale bubbles, each carrying the full collection of that tumor’s markers plus built-in "go" signals for killer T cells. When injected under the skin of mice after surgery, these vesicles traveled efficiently to nearby lymph nodes, where immune cells are trained. There they could both engage killer T cells directly and be swallowed by natural immune sentinels, which then further boosted the response. Experiments showed that T cells exposed to these vesicles multiplied more vigorously and were better at destroying cancer cells than those primed by simpler nanoparticle designs.
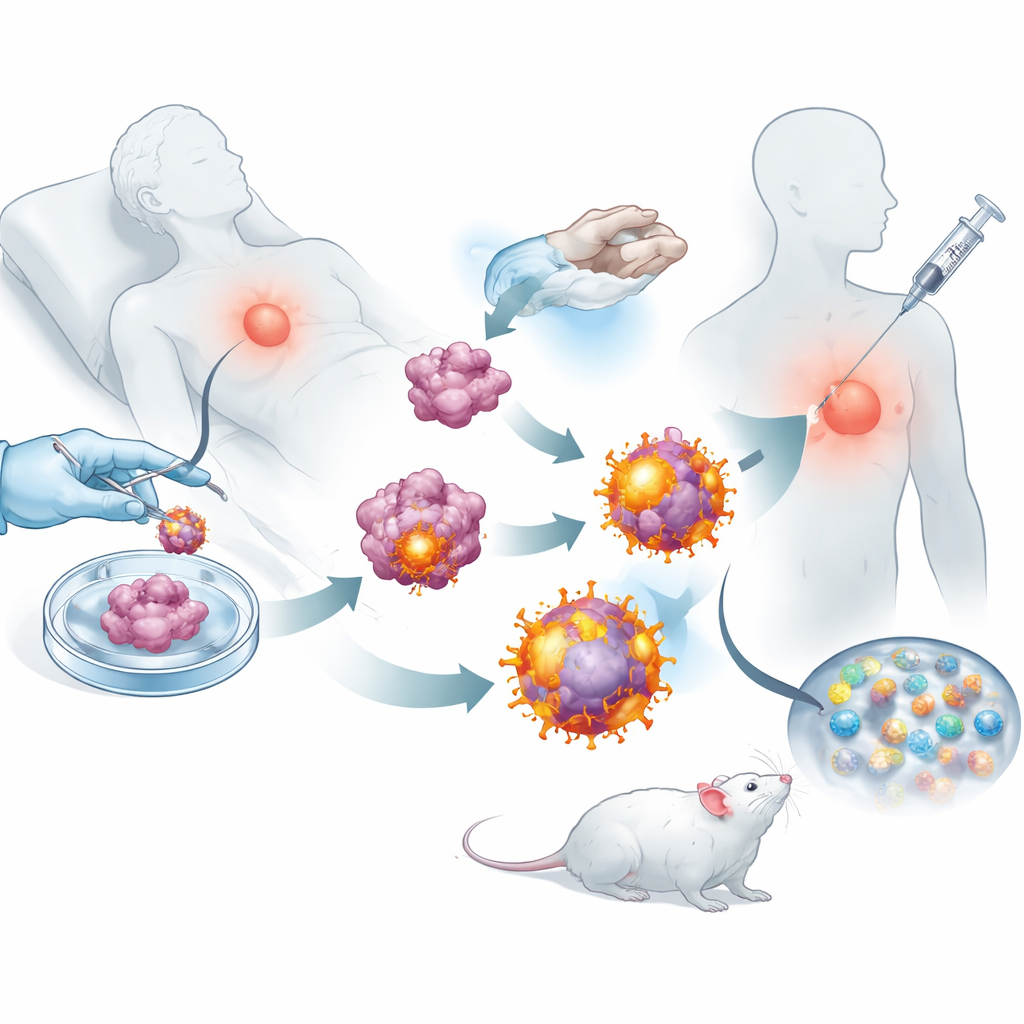
Keeping Cancer at Bay After Surgery
Using several mouse models of breast cancer and melanoma, the researchers tested whether this tailored vaccine could actually change the course of disease. In animals where most of a breast tumor had been surgically removed, vaccinated mice developed smaller recurrences and far fewer lung metastases than untreated animals. Lymph nodes, spleen, and tumors from treated mice teemed with active killer T cells and long-lived memory cells, suggesting that the immune system had been both awakened and trained for the long term. When combined with an approved checkpoint-blocking antibody that prevents tumors from paralyzing T cells, the vaccine led to durable tumor control and, in many mice, complete regression and long-term survival.
Speeding Up a Personalized Treatment
One obstacle to turning this concept into a real-world therapy is time: gene-based methods to reprogram tumor cells can be slow. To tackle this, the team screened natural compounds and discovered deoxypodophyllotoxin, a small molecule that quickly raises levels of the key switch protein and the same immune-stimulating surface markers. Using this shortcut, they built similar vesicle-coated nanoparticles in less time while preserving their ability to rouse strong, tumor-specific immune responses. Tests showed that vaccines made with this drug were just as effective as those produced with the gene-based method, and their benefits depended on recognizing the correct tumor type rather than on any general poisoning of cancer cells.
What This Could Mean for Future Cancer Care
To a non-specialist, the take-home message is that a patient’s own tumor can be reworked into a miniature training tool for the immune system. By converting cancer cells into sources of rich, personalized information and packaging that information onto sturdy nanoscopic carriers, this approach aims to sweep up cells that surgery leaves behind and prevent new growths from taking hold. While many steps remain before such vaccines could be routinely offered to people—including proving safety and effectiveness in human tumors and building reliable manufacturing pipelines—this work sketches a path toward highly individualized post-surgical treatments that enlist the body’s defenses to finish what the surgeon started.
Citation: Yu, P., Jin, Z., Meng, L. et al. Biomimetic vesicles engineered from modified tumour cells act as personalized vaccines for post-surgical cancer immunotherapy. Nat. Nanotechnol. 21, 443–454 (2026). https://doi.org/10.1038/s41565-025-02113-w
Keywords: cancer vaccine, immunotherapy, nanomedicine, postoperative treatment, personalized oncology