Clear Sky Science · en
Superionic composite electrolytes with continuously perpendicular-aligned pathways for pressure-less all-solid-state lithium batteries
Why this new battery material matters
Rechargeable batteries power our phones, cars and an increasing share of the electric grid. Many researchers see all-solid-state lithium batteries as a safer, higher-energy successor to today’s liquid-filled cells, but their solid electrolytes tend to be either fast at moving lithium ions or mechanically flexible—not both. This paper reports a new composite material that breaks that trade-off, pointing toward solid-state batteries that are both powerful and practical to manufacture.
The challenge of solid batteries
Conventional lithium-ion batteries use flammable liquid electrolytes that shuttle lithium ions between electrodes. Replacing the liquid with a solid could improve safety and allow the use of energy-dense lithium metal anodes. Unfortunately, most inorganic solid electrolytes, which move ions quickly, are brittle and make poor contact with the electrodes unless they are squeezed under very high pressure. Polymer electrolytes, by contrast, are soft and conformal but conduct ions slowly at room temperature. Composite electrolytes that mix inorganic particles into polymers usually inherit some of each problem, forcing engineers to choose between speed and robustness.
A layered shortcut for ions
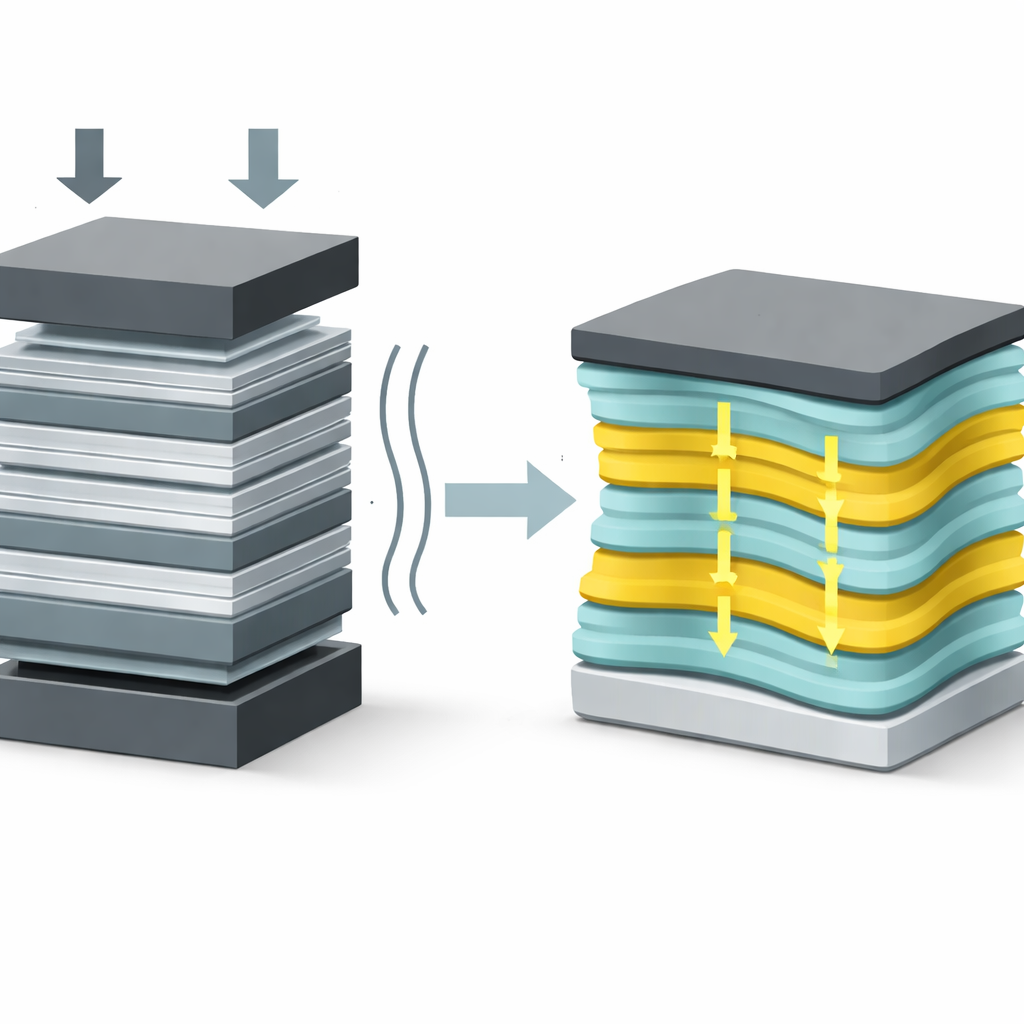
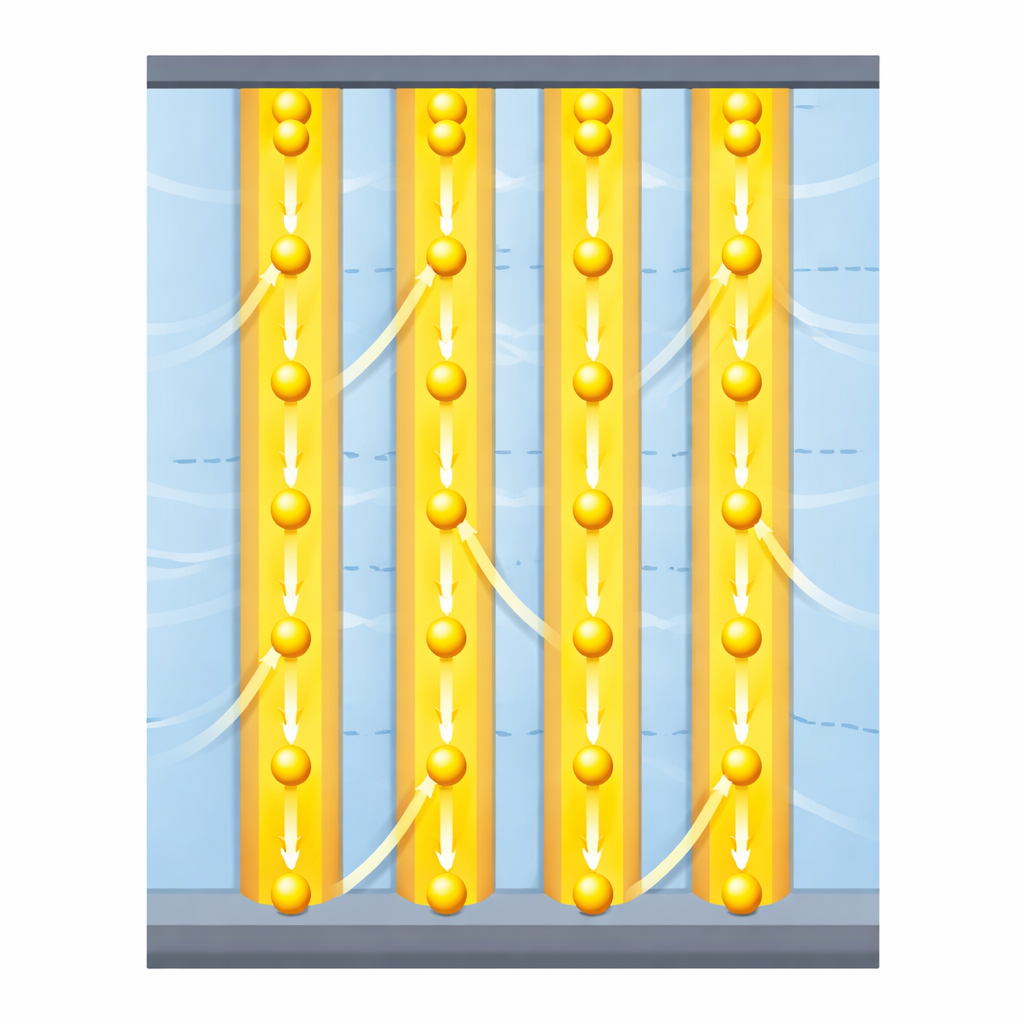
The authors tackle this problem by building a composite with a very deliberate internal structure. They use ultra-thin sheets of a sulfide material called LiMPS (where M is cadmium or manganese) that naturally conducts lithium ions extremely fast within the plane of each sheet but much more slowly across it. Instead of dispersing these sheets randomly in a polymer, they stack them into continuous layers and alternate them with layers of a flexible polymer, polyethylene oxide (PEO). Then they slice the block so that the LiMPS sheets stand perpendicular to the battery electrodes, forming straight, continuous, two-dimensional “highways” for ions through the thickness of the electrolyte.
Borrowing design tricks from nature
This architecture is inspired by biological materials such as the hinge of the bivalve shell Cristaria plicata, which combines stiff mineral fibers with soft organic layers to create a structure that bends without breaking. In the new electrolyte, tightly packed LiMPS layers carry most of the ion traffic, while the softer PEO layers absorb mechanical stress and help the solid maintain intimate contact with the electrodes as the battery charges and discharges. Additives in the polymer increase its flexibility and adhesion, so the layered stack behaves more like a tough plastic film than a brittle ceramic slab, even though it contains a high fraction of the inorganic conductor.
Performance that rivals liquids
By aligning the superionic LiMPS layers, the researchers achieve room-temperature ionic conductivities of 10.2 millisiemens per centimeter for the cadmium-based version and 6.1 millisiemens per centimeter for the manganese-based one—values comparable to or better than many liquid electrolytes and far above typical polymer or composite solids. Measurements and computer simulations show that lithium ions preferentially follow the LiMPS layers, confirming that the layered structure funnels ions along fast paths. At the same time, the membranes can be stretched to large strains without fracturing, and they maintain their structure and conductivity after days of exposure to moist air, unlike many sulfide electrolytes that quickly release toxic hydrogen sulfide gas.
From lab material to working cells
When built into lithium-metal coin cells, the new electrolyte supports long-term cycling with very small voltage losses, even at relatively high current densities. Lithium||LiNi0.8Co0.1Mn0.1O2 cells retain about 92 percent of their original discharge capacity after 600 cycles at room temperature, with nearly perfect charge–discharge efficiency. Crucially, the mechanical design allows these solid-state cells to operate with little to no external pressure, including in practical pouch-cell formats—something most high-conductivity inorganic electrolytes cannot do. The team also demonstrates a manganese-based variant that replaces the scarcer cadmium, improving prospects for scale-up.
What this means for future batteries
In simple terms, the researchers have built a solid electrolyte that lets lithium ions race along dedicated express lanes while a flexible backbone keeps everything in gentle, reliable contact. By decoupling ion transport from mechanical strength, their biomimetic layered design addresses several key roadblocks to real-world solid-state lithium batteries: conductivity, safety, air stability and pressure-free operation. While further engineering and manufacturing development are needed, this work outlines a general recipe for constructing flexible, superionic pathways inside solids, bringing commercial solid-state batteries a step closer.
Citation: Lan, X., Li, Z., Zhao, C. et al. Superionic composite electrolytes with continuously perpendicular-aligned pathways for pressure-less all-solid-state lithium batteries. Nat. Nanotechnol. 21, 388–396 (2026). https://doi.org/10.1038/s41565-025-02106-9
Keywords: solid-state batteries, lithium electrolytes, nanocomposites, energy storage, battery safety