Clear Sky Science · en
Tryptophanase disruption promotes insect–bacterium mutualism
How Tiny Gut Partners Can Make or Break an Insect
Many animals, including humans, rely on trillions of microbes living in their guts. These hidden partners can help digest food, make vitamins, or in some cases make us sick. This study looks at a simple but surprising genetic tweak in bacteria that turns an ordinary microbe into a life‑saving partner for a plant‑feeding stinkbug. By zooming in on a single bacterial gene, the work reveals how a microscopic change can kick‑start a stable friendship between very different forms of life.
A Bug, a Bacterium, and an Unlikely Alliance
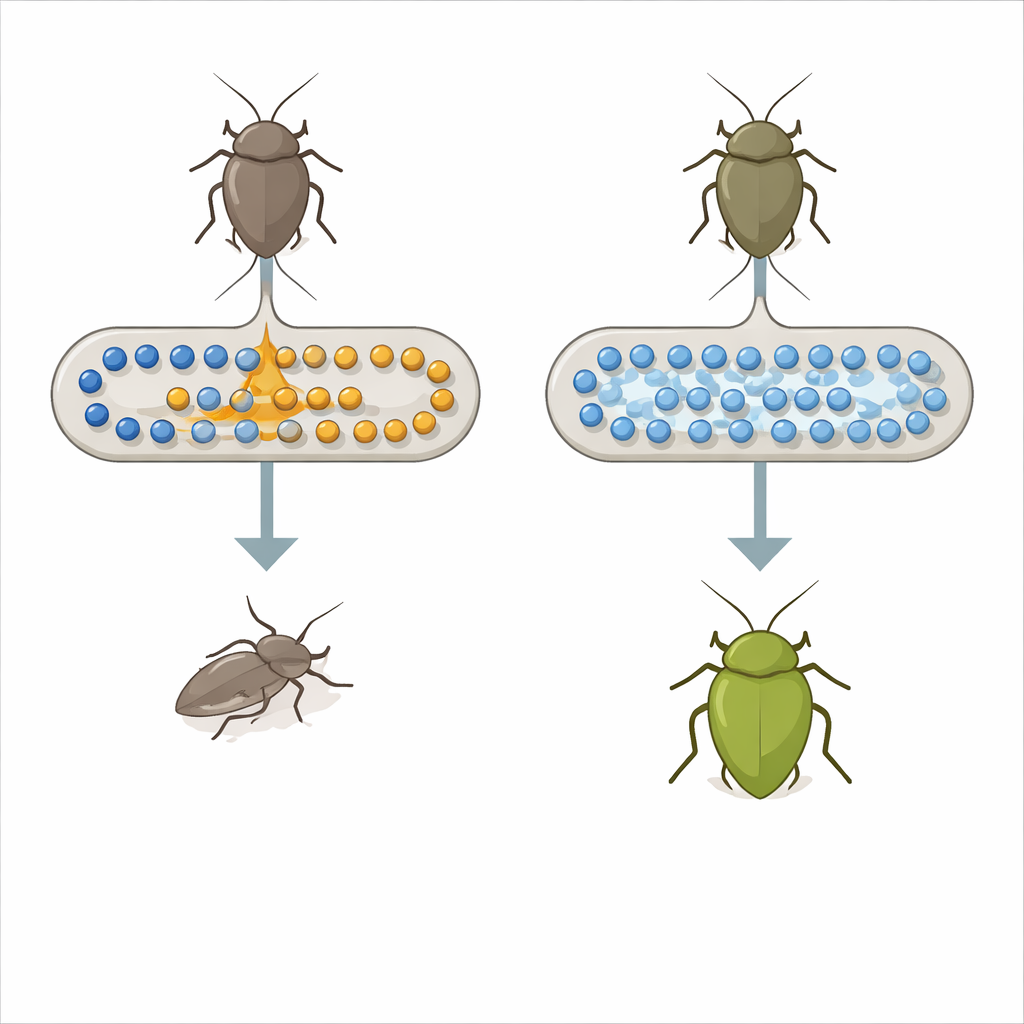
The stinkbug Plautia stali depends on special bacteria living in a pocket of its midgut to grow and survive. In nature, these partners are usually members of the genus Pantoea, which supply nutrients the insect cannot get from plant sap alone. Laboratory work had already shown that a common lab strain of Escherichia coli, a typical mammalian gut bacterium, can quickly evolve into a helpful partner for this stinkbug after just a single mutation in a global control system called carbon catabolite repression. That earlier finding raised a big question: is such a drastic regulatory change really how helpful symbioses arise in nature, or is there a more focused genetic switch at work?
Following the Trail of a Single Building Block
To answer this, the researchers compared stinkbugs carrying ordinary E. coli with those carrying evolved, helpful E. coli mutants. They measured many small molecules in the insect blood and gut. One essential amino acid, tryptophan, stood out: its levels were more than ten times higher in insects hosting the helpful mutants than in those hosting normal E. coli. Among dozens of bacterial genes affected by the earlier regulatory mutation, two were linked to tryptophan processing. One, called tnaA, makes an enzyme that breaks down tryptophan into a compound named indole along with other by‑products; the other helps transport tryptophan into the cell. When the team deleted the tnaA gene from E. coli, the stinkbugs suddenly did much better, with higher survival and a rich green body color characteristic of healthy, well‑nourished insects.
When Less Destruction Means More Help
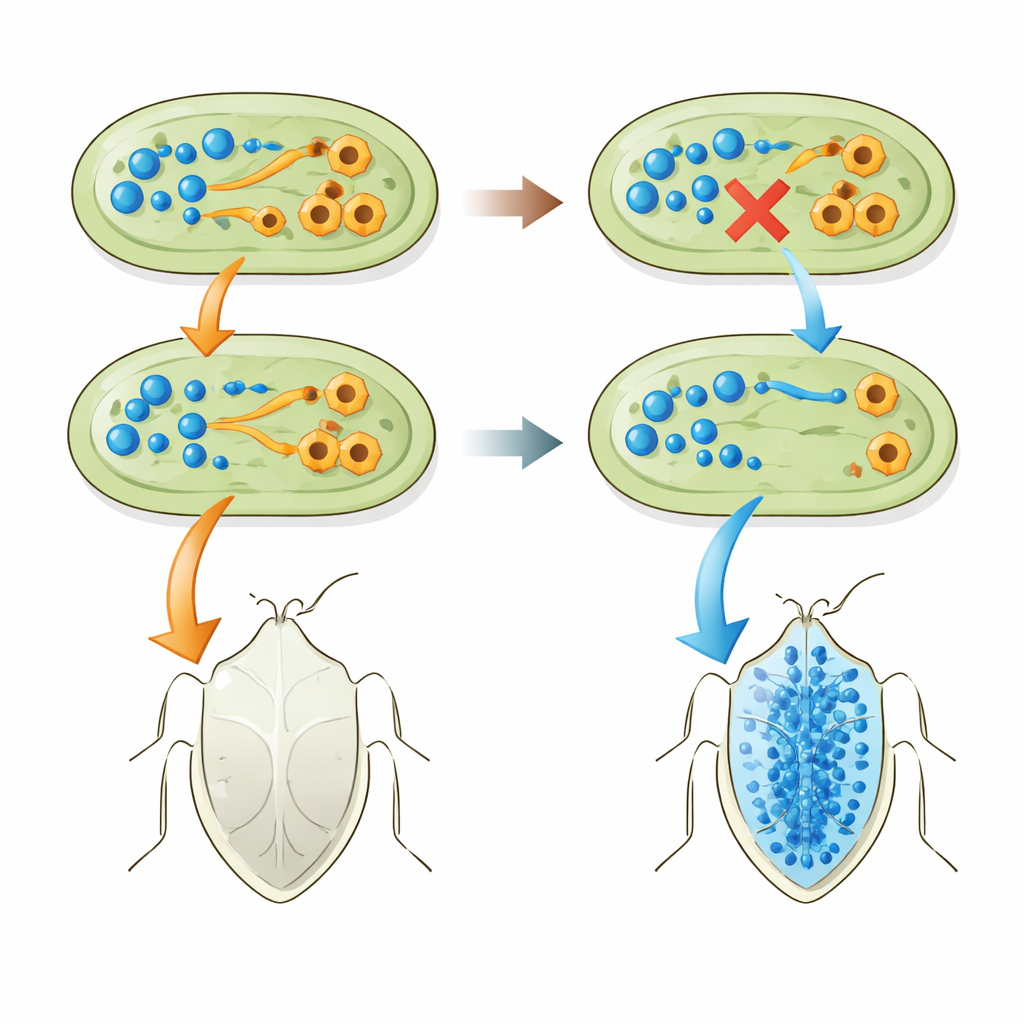
The key change turned out not to be that the bacteria made more tryptophan, but that they stopped destroying it and stopped making excess indole. Insects carrying tnaA‑deficient E. coli had high tryptophan and almost no indole circulating in their bodies. In contrast, insects carrying normal E. coli had low tryptophan and much higher indole. Feeding experiments supported this picture: giving indole in drinking water harmed the insects, especially those with indole‑producing bacteria, while extra tryptophan was only harmful when bacteria could convert it into more indole. A separate E. coli strain engineered to overproduce tryptophan gave the bugs a modest boost, reinforcing the idea that more of this building block, and less toxic breakdown product, improves the insect’s health.
Natural Partners Share the Same Genetic Loss
The team then asked whether this same gene loss shows up in real‑world symbioses. They sequenced genomes of many Pantoea bacteria living inside P. stali and related stinkbugs across the Ryukyu Islands of Japan, as well as Pantoea strains isolated from soil that could be coaxed into helping the bugs. Strikingly, every successful or potentially successful symbiont lacked the tnaA gene and showed no activity of the tryptophan‑breaking enzyme. By contrast, several free‑living Pantoea ananatis strains that still carried tnaA could not support stinkbug development at all. When the researchers knocked out tnaA in one of these P. ananatis strains, its ability to help the insects improved, though not to the level of natural partners. And when they forced a natural symbiont to carry an active tnaA operon, the insects fared worse and had lower tryptophan and higher indole in their blood.
What This Means for Life’s Hidden Partnerships
Taken together, the results point to a simple but powerful rule: bacteria that stop breaking down tryptophan, and thus avoid flooding their host with indole, are more likely to become dependable partners for plant‑feeding stinkbugs. In the lab, this can be triggered by a mutation in a global regulatory system; in nature, it shows up as a direct loss of the tryptophan‑cleaving gene itself. Either way, shutting off that one enzyme helps align the interests of insect and microbe. The study suggests that the road from loose association to tight mutualism may sometimes hinge on surprisingly small genetic steps, and that similar losses of function in bacterial metabolism may underlie many other hidden alliances across the tree of life.
Citation: Wang, Y., Moriyama, M., Koga, R. et al. Tryptophanase disruption promotes insect–bacterium mutualism. Nat Microbiol 11, 759–769 (2026). https://doi.org/10.1038/s41564-026-02264-z
Keywords: insect microbiome, bacterial mutualism, tryptophan metabolism, symbiosis evolution, Pantoea symbionts