Clear Sky Science · en
mRNA vaccination overcomes haemozoin-mediated impairment of whole-parasite malaria vaccines in mice
Why past malaria makes vaccines less powerful
Malaria kills hundreds of thousands of people every year, and one of the most promising vaccines uses whole, weakened parasites to train the immune system. Yet curiously, this "gold standard" approach works much better in people who have never had malaria than in those who grew up in regions where the disease is common. This study, done in mice, asks a simple but crucial question: what is it about previous malaria infections that makes a highly effective vaccine suddenly falter—and can a new kind of mRNA vaccine fix the problem?
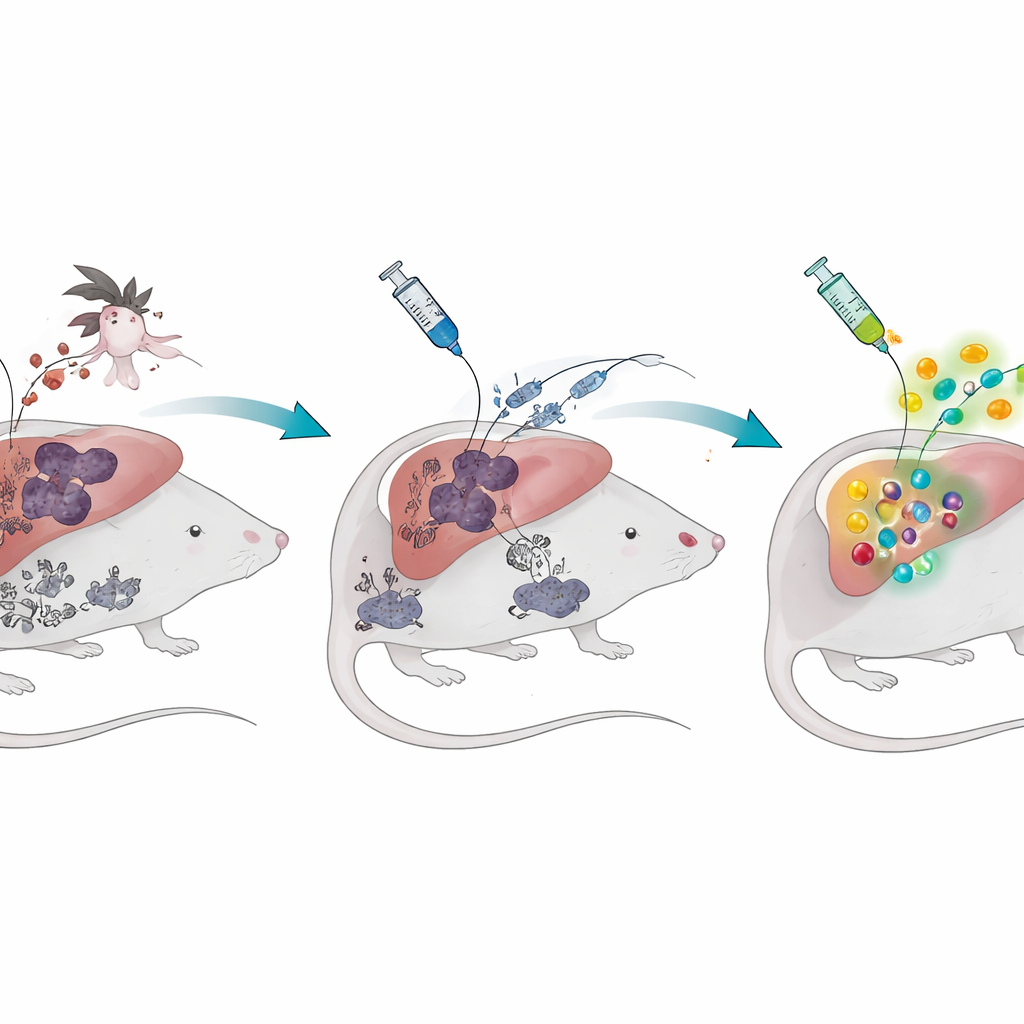
A tale of two stages of malaria
The malaria parasite has two major stages in the human body. First, after a mosquito bite, it quietly infects the liver. Later, it invades red blood cells, causing fever and illness. Whole-parasite vaccines called radiation-attenuated sporozoites target the silent liver stage: the parasites enter liver cells but cannot complete their life cycle, giving the immune system a safe preview. In malaria-naive mice, this strategy creates large numbers of specialized killer T cells that park in the liver and can wipe out parasites before they cause disease. But when the researchers first gave mice a blood-stage malaria infection and let it clear, the same vaccine produced far fewer protective T cells, and the animals were much less protected when challenged with live parasites.
A lingering pigment that won’t go away
The team then asked what was different in these "malaria-experienced" mice. One standout suspect was haemozoin, a dark, crystal-like pigment that the parasite makes inside red blood cells to detoxify the iron-rich waste from digesting hemoglobin. This pigment doesn’t disappear when the infection clears; it can sit in the liver, spleen and bone marrow for many months. The scientists found that even more than a year after a blood-stage infection had ended, mice still showed both stored haemozoin and a weakened T cell response to the liver-stage vaccine. Giving healthy mice purified synthetic haemozoin alone—without any live parasites—was enough to mimic this long-lasting vaccine weakness and reduce protection against later malaria challenge.
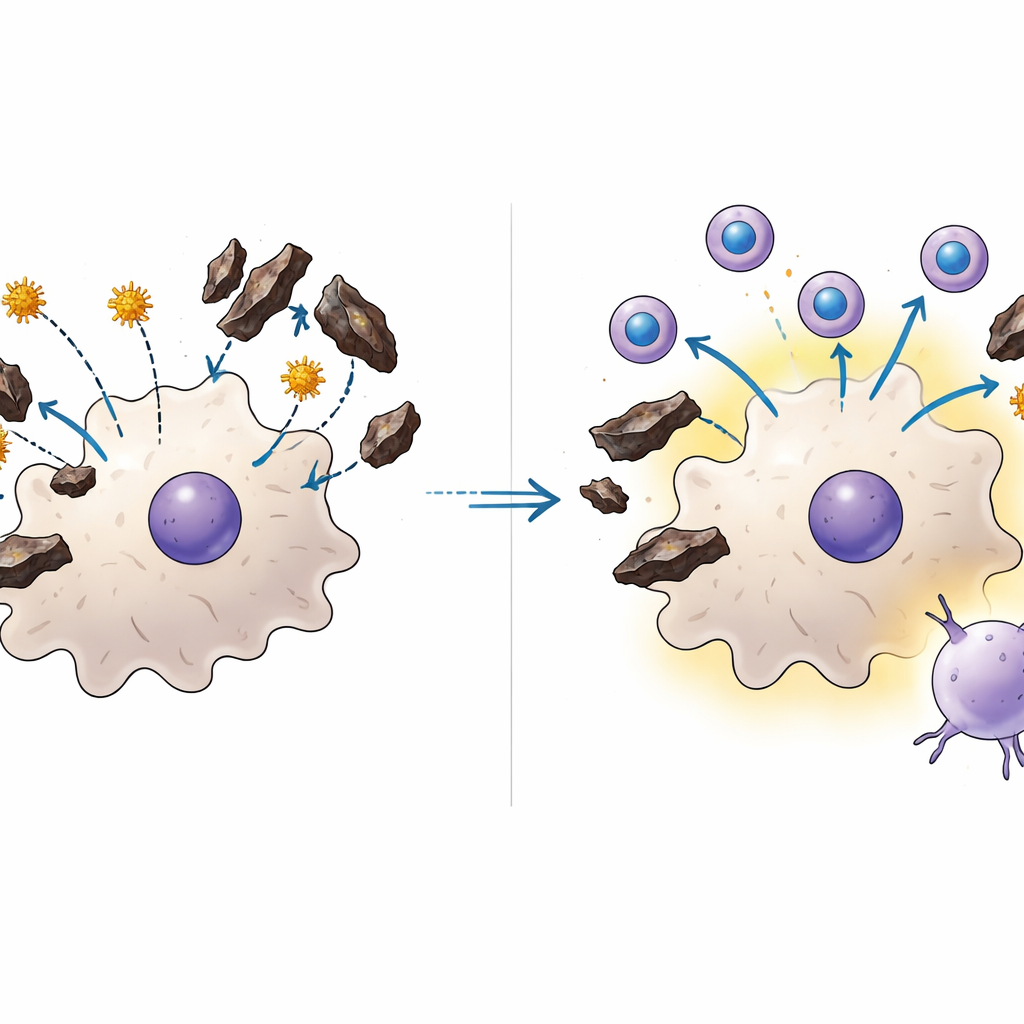
How haemozoin trips up immune sentinels
Diving deeper, the researchers focused on dendritic cells, the immune system’s sentinels that pick up bits of pathogens and present them to T cells. For the whole-parasite liver vaccine to work, dendritic cells must capture parasite material coming from infected liver cells and show it to killer T cells. In cell culture experiments, the team demonstrated that haemozoin directly interferes with this pick-up step: the more haemozoin dendritic cells had been exposed to, the less efficiently they swallowed protein antigens. This defect appeared in both mouse cells and human immune cells made from blood donors, and it did not depend on one well-known inflammatory sensor, suggesting haemozoin itself acts as a physical or biochemical roadblock to normal antigen handling.
mRNA vaccines find a way around the roadblock
To see if a different vaccine format could dodge this problem, the scientists designed an mRNA vaccine packaged in lipid nanoparticles. Instead of delivering parasite proteins from the outside, this approach gives host cells genetic instructions to make short pieces of several parasite proteins inside the cell. In mice with or without prior malaria, the mRNA vaccine generated similarly strong killer T cell responses and improved control of liver infection. Strikingly, when the mRNA vaccine was combined with a single dose of the whole-parasite liver vaccine, mice built especially large pools of T cells that took up long-term residence in the liver—a cell population known to be key for rapid protection. In dishes, haemozoin did not block the uptake or function of the mRNA-loaded nanoparticles, explaining how this strategy can succeed where the older vaccine alone fails.
What this means for future malaria shots
In everyday terms, the study shows that past malaria infections leave behind microscopic mineral-like debris that clogs how certain immune cells normally learn from vaccines, weakening a leading whole-parasite malaria shot. But by switching to, or combining with, an mRNA-based vaccine that sneaks the instructions for parasite pieces directly into cells, scientists can bypass this debris and restore strong protection in malaria-experienced hosts—at least in mice. The work points to haemozoin as an important obstacle for vaccines in people who live in endemic regions, and suggests that carefully designed mRNA or mixed vaccine regimens could be a promising path toward more effective, widely useful malaria immunization.
Citation: Hassert, M., Drewry, L.L., Pewe, L.L. et al. mRNA vaccination overcomes haemozoin-mediated impairment of whole-parasite malaria vaccines in mice. Nat Microbiol 11, 718–730 (2026). https://doi.org/10.1038/s41564-026-02263-0
Keywords: malaria vaccines, hemozoin, mRNA vaccine, T cell immunity, Plasmodium infection