Clear Sky Science · en
Skin androgens regulate Staphylococcus aureus pathogenicity via quorum sensing
Why hormones on our skin matter
Men tend to get worse skin infections than women, but the reasons have been murky. This study shows that hormones sitting right on the skin’s surface, especially the male hormone testosterone, can switch the behavior of a common bacterium from quiet neighbor to dangerous invader. Understanding this hidden conversation between our own hormones and microbes not only explains a long-standing sex difference in infection risk, it also points to an unexpected way to disarm antibiotic‑resistant germs without relying on traditional drugs.
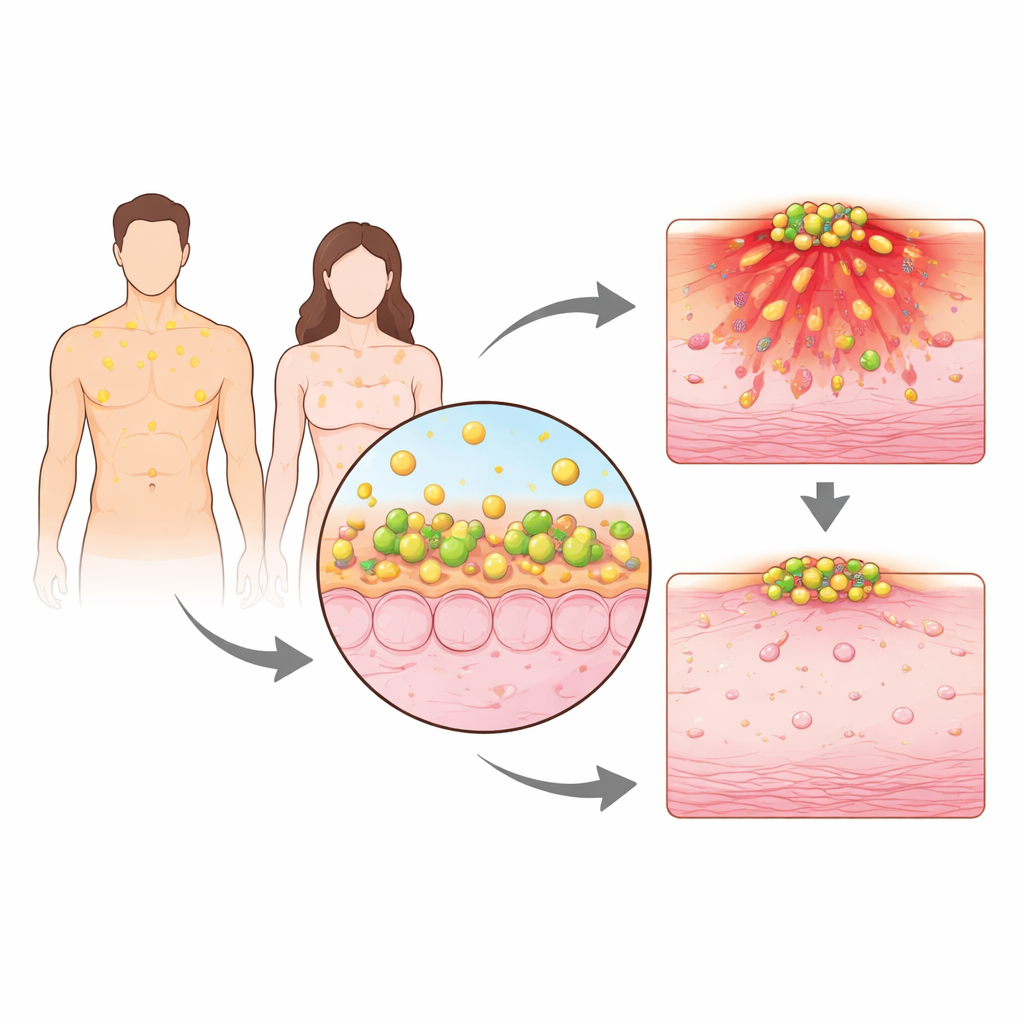
Everyday hormones bathing the skin
Our skin does more than shield us from the outside world; it also makes and releases its own cocktail of hormones. Using tiny adhesive strips that soak up skin oils, the researchers measured testosterone on the faces of healthy men and women over several days. Both sexes produced steady, nanomolar amounts of testosterone, but men consistently had more at the skin surface. In mice, males likewise had higher levels of testosterone and its close relative dihydrotestosterone in their skin secretions. These higher androgen levels went hand‑in‑hand with more severe infections when the animals were exposed to Staphylococcus aureus, a major cause of skin and soft tissue infections in people.
Turning down skin hormones protects against infection
To test whether skin‑made hormones truly matter, the team engineered mice whose skin cells lacked a key enzyme needed to produce testosterone locally, while leaving hormone levels in the bloodstream unchanged. These "androgen‑deficient" mice had normal skin structure and barrier function but released much less testosterone, progesterone and dihydrotestosterone onto their skin. When infected with methicillin‑resistant Staphylococcus aureus (MRSA), these mice showed weaker bacterial signals, milder redness and swelling, less tissue damage and better‑preserved barrier function than normal mice. Remarkably, when female androgen‑deficient mice received a dab of testosterone on the skin, their infections worsened and began to resemble those seen in males. This showed that skin‑surface androgens, rather than whole‑body hormone status, help set the stage for S. aureus disease.
How bacteria eavesdrop on our hormones
S. aureus uses a communication system called quorum sensing to decide when to produce toxins that damage host cells. The heart of this system is a sensor pair, AgrC and AgrA, that normally responds to small bacterial peptides. When the researchers bathed different S. aureus strains in tiny amounts of testosterone or dihydrotestosterone, the bacteria ramped up genes that are normally switched on during quorum sensing, including those encoding potent toxins. This happened across multiple clinical strains, including MRSA and isolates from people with atopic dermatitis. Testosterone boosted bacterial killing of human red blood cells, skin cells and neutrophils, yet did not increase bacterial growth itself. In mice with low skin androgens, a quorum‑sensing reporter strain showed much weaker activation, confirming that skin hormones drive this bacterial "group decision‑making" in living tissue.
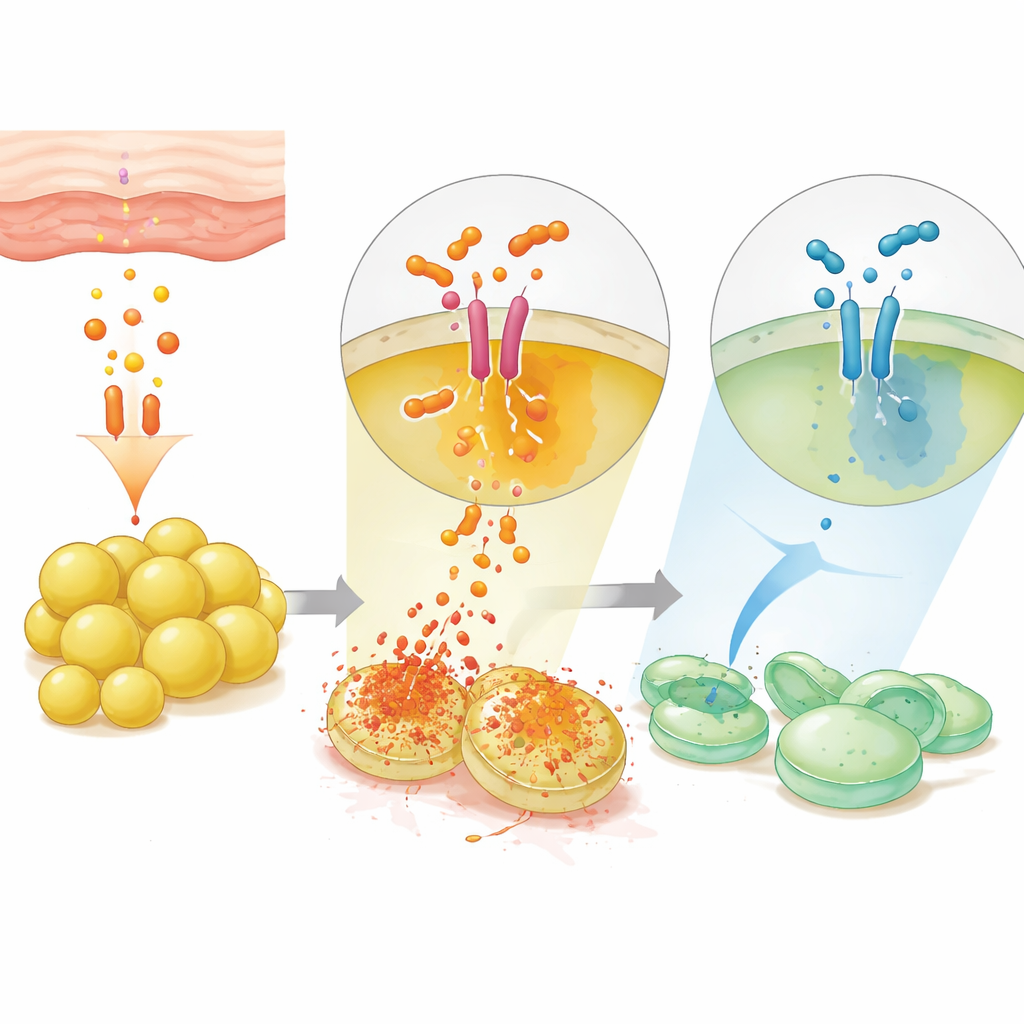
A direct molecular handshake and a built‑in off switch
Surprisingly, testosterone could still activate quorum sensing in mutant bacteria unable to make their own signaling peptides, meaning the hormone can substitute for the usual bacterial trigger. However, this required an intact AgrC sensor on the bacterial surface and its partner AgrA; when either was disrupted, testosterone no longer boosted toxin genes or cell damage. Computer modeling suggested that testosterone nestles into a hydrophobic pocket on AgrC distinct from the usual peptide‑binding site, physically pushing the receptor toward its active shape. The team then flipped the problem around by testing a mirror‑image version of testosterone, called enantiomer‑testosterone (ent‑T). Although chemically similar, ent‑T blocked quorum sensing, reduced toxin production and protected human cells from bacterial damage. Applied to infected mouse skin, ent‑T dampened quorum‑sensing activity in both sexes, acting as a molecular brake on virulence.
What this means for future treatments
In plain terms, the study reveals that S. aureus can literally "smell" testosterone on our skin and uses that signal to decide when to attack. Men’s higher skin androgen levels help explain why they are more prone to severe S. aureus infections. Just as important, the work introduces a new therapeutic idea: instead of killing bacteria outright, we might dial down their aggression by blocking their hormone sensors. Compounds like ent‑T, or strategies that lower skin‑surface androgens, could weaken MRSA and related strains, making infections easier to control and less damaging while sidestepping some of the pressures that drive antibiotic resistance.
Citation: John, M.S., Chinnappan, M., Sturges, C.I. et al. Skin androgens regulate Staphylococcus aureus pathogenicity via quorum sensing. Nat Microbiol 11, 704–717 (2026). https://doi.org/10.1038/s41564-026-02261-2
Keywords: skin hormones, Staphylococcus aureus, quorum sensing, MRSA infection, testosterone signaling