Clear Sky Science · en
Host factors dictate gut microbiome alterations in chronic kidney disease more strongly than kidney function
Why gut microbes matter for kidney health
People living with chronic kidney disease often hear that their “numbers” are getting worse, but less attention is paid to another hidden player: the trillions of microbes in their gut. This study asks a deceptively simple question with big implications: are changes in gut bacteria mainly driven by failing kidneys themselves, or by everyday factors such as diet, medications and how fast food moves through the intestines? The answer could reshape how we prevent harmful toxins from building up in the blood and help doctors design more effective lifestyle-based treatments.
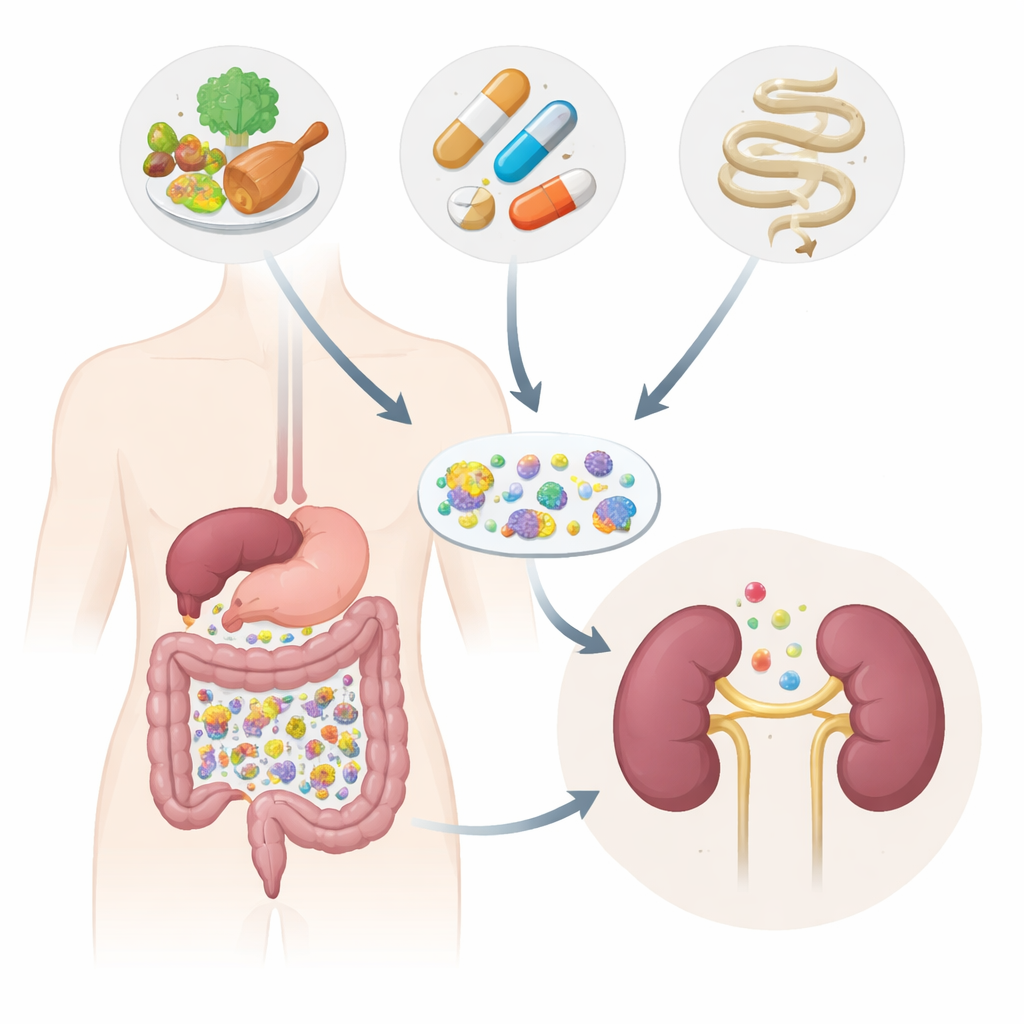
The study and who took part
The researchers followed 130 adults in Belgium: people with different stages of chronic kidney disease, a small group on peritoneal dialysis, and individuals without kidney problems. They collected stool and blood samples, measured many blood chemicals, and carefully recorded medication use and signs of gut inflammation. Instead of just looking at the relative proportions of microbes, they used a more advanced approach that estimates the absolute number of bacterial cells and the genes they carry. They also compared their results with data from more than 4,000 people in other studies to see whether proposed “kidney disease microbes” really hold up across different groups.
Slow digestion and medicines outweigh kidney function
One of the clearest signals did not come from the kidneys at all, but from the gut’s own rhythm. People whose stools were drier—indicating slower movement of food through the intestines—tended to have microbiomes dominated by bacteria that thrive on protein rather than on plant fibers. This shift, called a move from “sugar-loving” to “protein-loving” fermentation, was more strongly tied to intestinal transit time and stool moisture than to kidney filtration rate itself. Medications, such as drugs for diabetes, blood thinners and certain mood-related medicines, also explained a sizable fraction of differences in gut communities. In other words, how fast food passes through the gut and which pills people take often mattered more than how well their kidneys were working.
A special gut pattern in dialysis patients
Patients on peritoneal dialysis showed a particularly disturbed gut ecosystem. They were much more likely to carry a community pattern known from previous work to be linked with inflammation and gut imbalance. These patients had higher levels of calprotectin, a marker of intestinal inflammation, and fewer different bacterial species overall. The findings suggest that kidney replacement therapy, along with the high medication burden it usually entails, may push the gut toward a more inflamed and less diverse state, distinct from that seen in earlier stages of kidney disease.
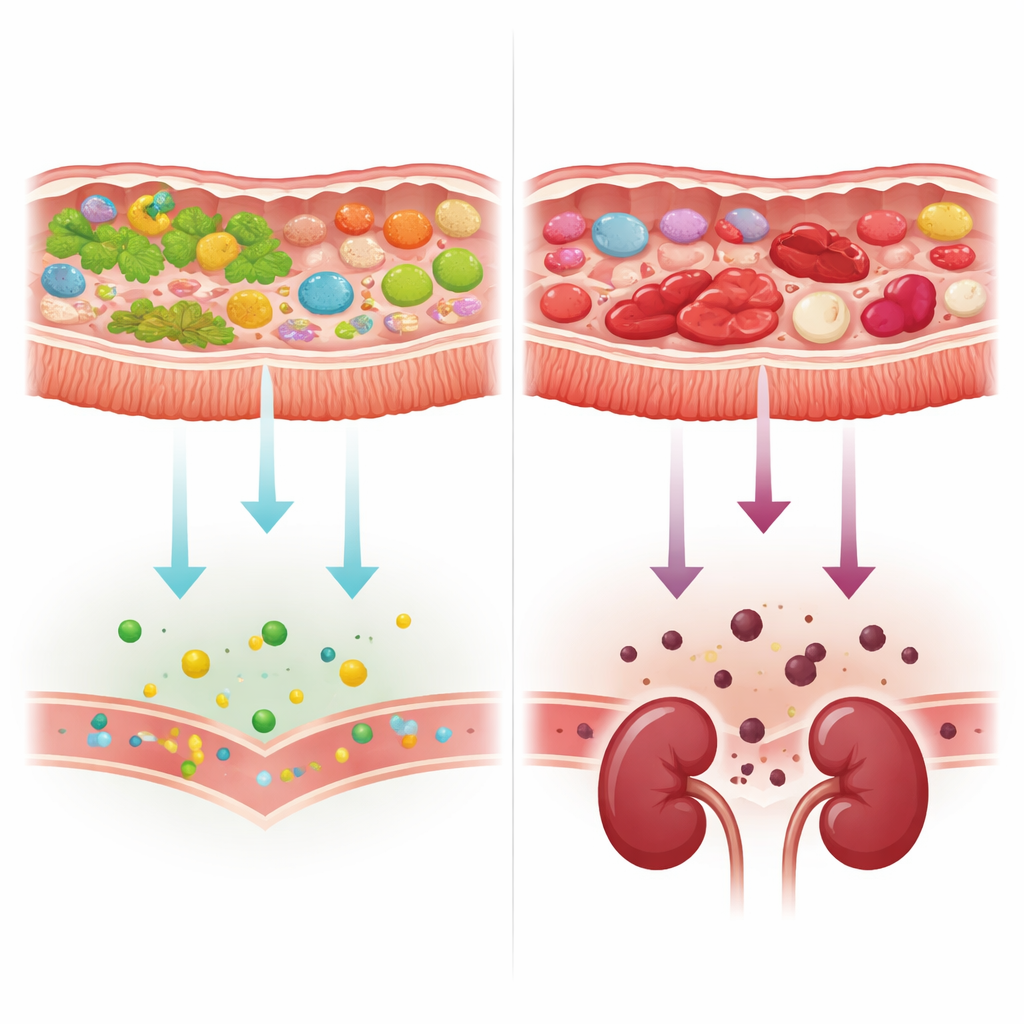
From plant foods to protein and toxin production
As kidney disease advanced, the microbial genes present in stool samples shifted. Microbes gradually lost tools for breaking down plant-derived carbohydrates and showed relatively more machinery for handling animal-derived components and proteins. At the same time, genes involved in making two key gut-derived precursors of kidney-related toxins—p-cresol and indole—became more common in people with poorer kidney function. A narrow set of bacteria, particularly certain strains of Escherichia coli and an Alistipes species, carried many of these genes, while a typically beneficial species, Bifidobacterium adolescentis, was more abundant in people with better kidney function and lacked these toxin-making pathways. However, when the researchers accounted for a simple genetic marker of diet—the balance of plant- versus animal-linked carbohydrate-processing enzymes—the apparent link between kidney function and toxin-related pathways largely disappeared, pointing back to food choices and gut conditions as the main drivers.
Rethinking “kidney microbiome” markers
Over the past decade, many small studies have reported specific bacterial species as hallmarks of chronic kidney disease. When this team systematically re-examined 24 of those proposed markers in their own data and across 11 studies, most associations did not reliably repeat. Once they controlled for stool moisture and other host factors, only three bacterial species remained consistently tied to kidney filtration rate, and none predicted which patients’ kidney function would decline faster over four years. This suggests that earlier work may have overestimated the importance of individual “bad” or “good” bacteria by ignoring how deeply the microbiome is shaped by diet, medicines and gut transit.
What this means for patients and care
The authors conclude that many of the gut changes seen in chronic kidney disease are not direct fingerprints of failing kidneys but side effects of slower bowel movement, altered diets and heavy medication use. These conditions favor microbes that feed on protein and produce more precursors of uremic toxins, which can then accumulate in the blood when kidneys are already struggling. For patients, this means that strategies aimed at softening stools, shortening intestinal transit time and increasing intake of plant-based fibers could be as important as targeting specific microbes. Rather than chasing a single “kidney disease microbiome,” future care may focus on restoring a healthier balance between fiber fermentation and protein breakdown in the gut, potentially easing the toxic load on vulnerable kidneys.
Citation: Krukowski, H., Valkenburg, S., Vich Vila, A. et al. Host factors dictate gut microbiome alterations in chronic kidney disease more strongly than kidney function. Nat Microbiol 11, 664–677 (2026). https://doi.org/10.1038/s41564-026-02259-w
Keywords: chronic kidney disease, gut microbiome, intestinal transit time, dietary fiber, uremic toxins