Clear Sky Science · en
Initial sites of SIV rebound after antiretroviral treatment cessation in rhesus macaques
Why the Virus Comes Back
Modern HIV medicines can drive the virus in blood down to levels so low that standard tests see nothing. Yet when treatment stops, the virus almost always returns. This study in rhesus macaques, infected with a close cousin of HIV called SIV, asks a basic but crucial question: where, exactly, does the very first spark of this viral comeback occur inside the body, and what kinds of tissues fan it into a flame?
Following Viral Barcodes
To track the virus with unusual precision, the researchers used a special SIV in which each virus particle carries a unique genetic "barcode"—a short, harmless sequence that works like a serial number. Twenty-four macaques were infected with a mix of thousands of these barcoded viruses and then quickly started on a strong three-drug antiretroviral regimen. The drugs pushed virus in the blood down to essentially undetectable levels and kept it there for more than a year, mimicking effective long-term HIV therapy in people. Because each barcode marked a distinct viral family, the team could later identify which specific viral lineages woke up when treatment stopped and where in the body that awakening first took hold.
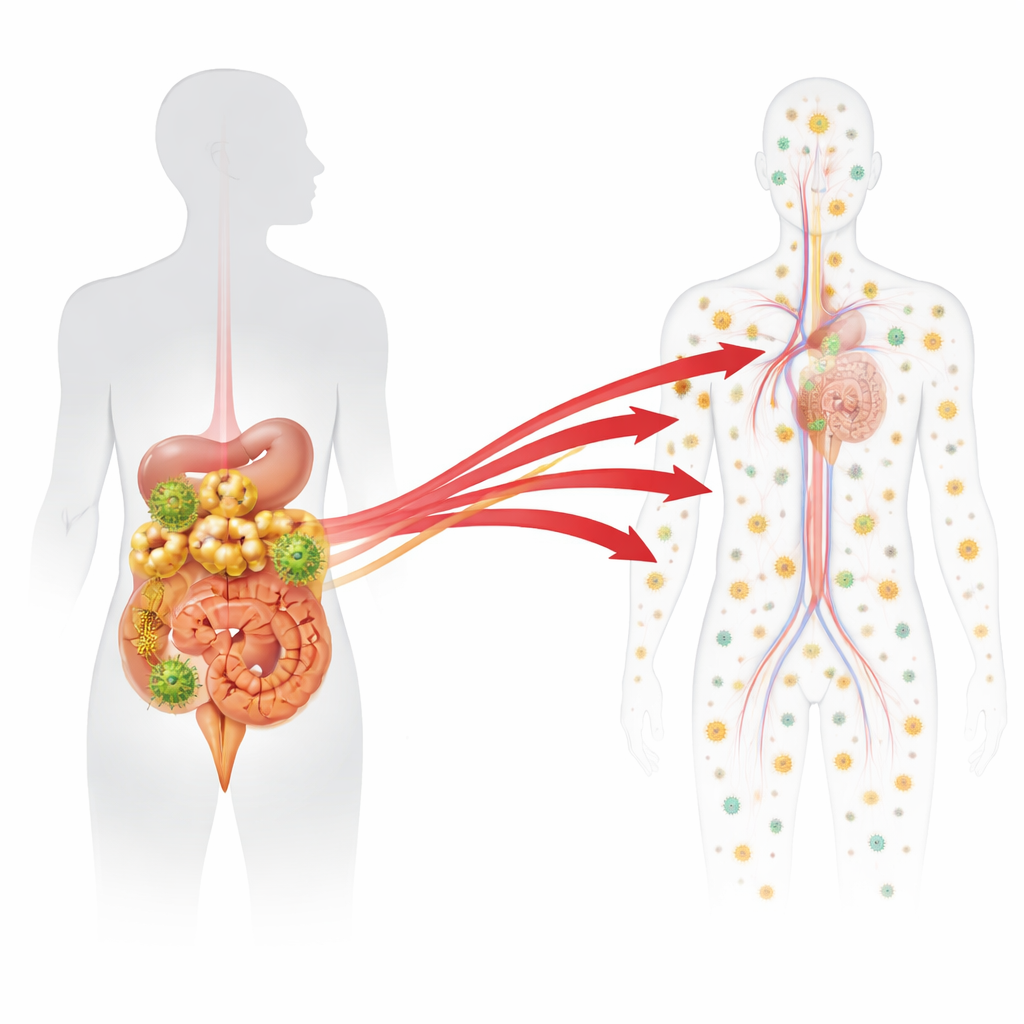
Peering Into Tissues During and After Treatment
At the end of the long treatment period, the macaques were divided into groups. Some were examined while still on therapy to define the normal background pattern of tiny amounts of viral genetic material that persist despite drugs. Others had treatment stopped and were examined either five or seven days later, before or just as virus began to reappear in the blood. From each animal, scientists collected and analysed around 90 tissue samples spanning the gut, many kinds of lymph nodes, blood-forming organs like bone marrow, and non-immune organs such as liver and lung. For every tissue, they measured how much viral DNA (a sign of infected cells) and viral RNA (a sign of active virus production) was present for each barcode. They then used statistical models to mark any barcode whose RNA level in a particular tissue rose well above what would be expected from simple low-level background activity while on treatment.
Pinpointing the First Sparks of Rebound
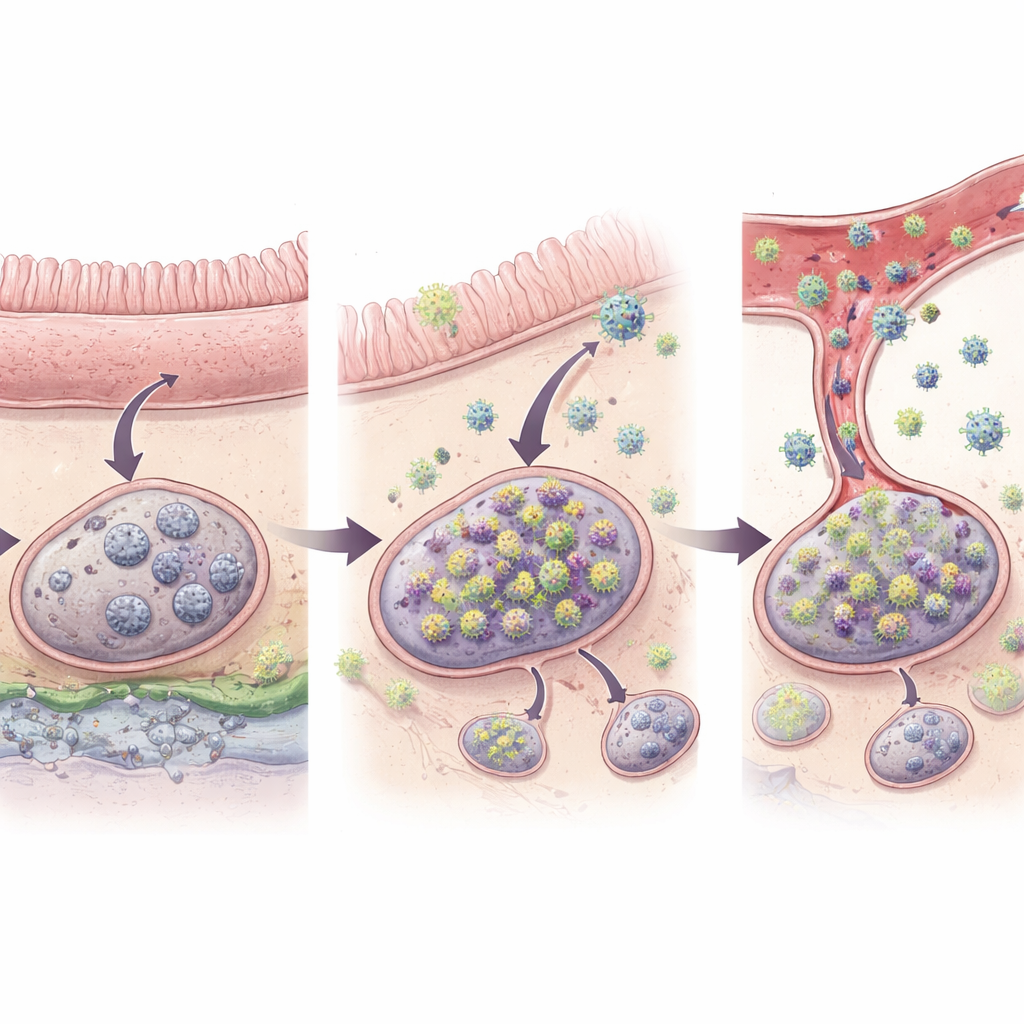
Those unusually active barcodes, which the researchers called "outliers," singled out viral lineages that were likely undergoing fresh bursts of replication and local spread after drug withdrawal. Across all animals, they identified 32 such outlier barcodes. Crucially, when virus did reappear in the blood, about half of the barcodes found in plasma matched these outliers in tissues, directly linking specific tissue sites to the earliest stage of systemic rebound. Most outliers showed up in just a single tissue sample, consistent with a very local starting point, while a smaller fraction had already spread to neighbouring gut segments and their draining lymph nodes. Only in the one animal with clearly higher virus levels in blood did the team see evidence that multiple tissues—including distant ones—had already been reseeded. This suggests a stepwise progression: first a quiet ignition in one spot, then local expansion, then body-wide dissemination.
Why the Gut Matters Most
One finding stood out: nearly all of the early rebound events traced back to the digestive tract and the immune tissues that serve it. Of the 27 barcodes judged to represent genuine origins of rebound, 96% were in the intestines themselves or in gut-associated lymph nodes, such as mesenteric nodes that drain the small and large bowel. Only a single origin site appeared in a lymph node not linked to the gut, and none were identified in organs like liver, lung or bone marrow. Statistical analyses showed that, even after accounting for how many infected cells were present, gut-associated lymphoid tissues were about ten times more likely than other lymphoid sites to give rise to rebounding virus. The gut’s unique environment—constantly exposed to food and microbes and often inflamed in HIV-like infections—may keep infected cells there more active and more easily nudged into producing virus again.
What This Means for Future Cures
Put simply, this work argues that when powerful HIV-like drugs are stopped, the virus almost always restarts from a few hidden reservoirs in and around the gut rather than equally from all corners of the body. Early on, these flare-ups are tiny and confined to a single patch of tissue, but once virus reaches the bloodstream and distant organs, levels rise quickly. For efforts aimed at long-term remission or cure, this means that therapies must reach and work effectively in the gut’s immune tissues. Tools that can visualize or sample these hard-to-access sites in people, or medicines that are specially tuned to act there, could be key to preventing the first sparks of rebound from ever catching fire.
Citation: Keele, B.F., Okoye, A.A., Immonen, T.T. et al. Initial sites of SIV rebound after antiretroviral treatment cessation in rhesus macaques. Nat Microbiol 11, 648–663 (2026). https://doi.org/10.1038/s41564-025-02258-3
Keywords: HIV rebound, viral reservoirs, gut lymphoid tissue, rhesus macaque model, antiretroviral therapy interruption